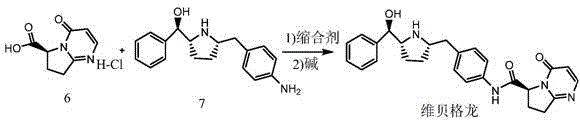
Advanced Synthesis of Vibegron Intermediate Salts for High-Purity API Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes for complex active pharmaceutical ingredients (APIs), particularly for treatments addressing chronic conditions like overactive bladder. Patent CN115850286A introduces a significant technological advancement in the preparation of a key intermediate for Vibegron, a beta-3 adrenergic agonist. This patent discloses a novel method for synthesizing a salt of the compound shown as Formula I, which serves as a critical precursor in the Vibegron supply chain. Unlike traditional approaches that struggle with purification and chiral integrity, this invention utilizes specific acid salts—such as hydrochloride, L-tartrate, or D-mandelate—to overcome solubility and crystallization challenges inherent to the molecule's polar functional groups. 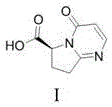 . By transforming the free acid or unstable sodium salt into a well-defined crystalline salt, manufacturers can achieve superior control over impurity profiles and stereochemistry, ensuring a reliable pharmaceutical intermediate supplier can meet the stringent quality demands of global regulatory bodies.
. By transforming the free acid or unstable sodium salt into a well-defined crystalline salt, manufacturers can achieve superior control over impurity profiles and stereochemistry, ensuring a reliable pharmaceutical intermediate supplier can meet the stringent quality demands of global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, specifically those disclosed in international patent WO2013062881A1, rely heavily on the synthesis of a sodium salt intermediate, which presents substantial hurdles for large-scale manufacturing. In these conventional routes, the precursor compound I-17 requires rigorous purification via silica gel column chromatography followed by recrystallization to achieve acceptable purity levels, yet the total recovery rate remains inefficiently low at approximately 68 percent. Furthermore, the subsequent hydrolysis to form the sodium salt (Compound I-12) often yields a product with chiral purity ranging only between 85 percent and 95 percent, necessitating costly and time-consuming chiral separation or enzymatic resolution steps to meet commercial standards. 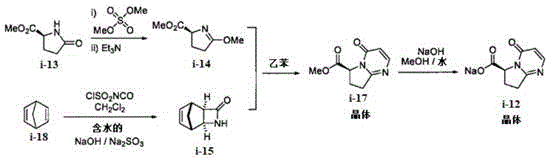 . Additionally, the sodium salt form complicates the downstream condensation reaction, requiring an extra acidification step to liberate the free acid before coupling, thereby increasing reagent costs and waste generation while introducing potential points of failure in the production line.
. Additionally, the sodium salt form complicates the downstream condensation reaction, requiring an extra acidification step to liberate the free acid before coupling, thereby increasing reagent costs and waste generation while introducing potential points of failure in the production line.
The Novel Approach
The innovative strategy presented in CN115850286A fundamentally shifts the paradigm by targeting the direct formation of stable acid salts from Compound 5, bypassing the problematic purification stages associated with the prior art. This method involves a streamlined hydrolysis of Compound 5 in the presence of an alkali, followed immediately by acidification with selected organic or inorganic acids to precipitate the target salt. 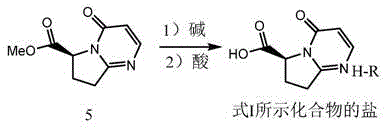 . This approach allows for the effective enrichment of enantiomeric excess (ee%) through simple recrystallization, achieving chiral purities exceeding 97 percent without the need for complex chromatographic separations. Crucially, because the intermediate is isolated as an acid salt rather than a sodium salt, it can be directly utilized in the subsequent condensation reaction with Compound 7 to form Vibegron without additional pH adjustments, significantly simplifying the overall synthetic sequence and enhancing process robustness for cost reduction in API manufacturing.
. This approach allows for the effective enrichment of enantiomeric excess (ee%) through simple recrystallization, achieving chiral purities exceeding 97 percent without the need for complex chromatographic separations. Crucially, because the intermediate is isolated as an acid salt rather than a sodium salt, it can be directly utilized in the subsequent condensation reaction with Compound 7 to form Vibegron without additional pH adjustments, significantly simplifying the overall synthetic sequence and enhancing process robustness for cost reduction in API manufacturing.
Mechanistic Insights into Alkaline Hydrolysis and Salt Crystallization
The core of this technological breakthrough lies in the precise control of the hydrolysis and subsequent salt formation kinetics. The process initiates with the nucleophilic attack of hydroxide ions on the ester moiety of Compound 5, typically conducted in a mixed solvent system of alcohol and water to balance solubility and reaction rate. Maintaining the reaction temperature between 0°C and 15°C is critical to prevent racemization at the chiral center while ensuring complete conversion of the starting material. Following hydrolysis, the careful addition of acid serves a dual purpose: it neutralizes the excess base and protonates the carboxylate anion to form the insoluble acid salt. The choice of acid, such as hydrochloric acid, dictates the crystal lattice energy and solubility profile, enabling the selective precipitation of the desired enantiomer while leaving impurities in the mother liquor. This crystallization-driven purification is far more scalable and economically viable than chromatographic methods, providing a clear pathway for the commercial scale-up of complex pharmaceutical intermediates.
Impurity control is further enhanced by the specific recrystallization protocols detailed in the patent, which utilize solvents like isopropanol to maximize the rejection of structural analogs and by-products. The formation of specific crystal forms, such as Form A of the hydrochloride salt, ensures consistent physical properties including flowability and stability, which are essential for downstream processing. By optimizing the molar ratios of base and acid, and strictly controlling the concentration of the reaction mixture during the workup, the process minimizes the entrapment of inorganic salts and solvent residues. This level of mechanistic understanding allows for the production of high-purity Vibegron intermediates that consistently meet rigorous specifications, thereby reducing lead time for high-purity pharmaceutical intermediates and ensuring a steady supply for final drug substance production.
How to Synthesize Vibegron Intermediate Salt Efficiently
The synthesis of this critical intermediate is designed for operational simplicity and high throughput, making it ideal for contract development and manufacturing organizations (CDMOs) looking to optimize their portfolios. The process begins with the dissolution of Compound 5 in a suitable alcohol-water mixture, followed by the controlled addition of an aqueous alkali solution under cooling to manage the exotherm. Once hydrolysis is confirmed complete via HPLC or TLC monitoring, the reaction mixture is treated with the chosen acid to induce salt formation. . The resulting crude solid is then subjected to a recrystallization step, typically involving heating in isopropanol followed by slow cooling, to yield the final high-purity product ready for the next synthetic transformation.
- Perform alkaline hydrolysis of Compound 5 in an alcohol-water solvent system at 0-15°C using NaOH, LiOH, or KOH.
- Upon completion, add a stoichiometric amount of acid (e.g., HCl, L-tartaric acid) to the reaction mixture to form the corresponding salt precipitate.
- Concentrate the reaction liquid, remove inorganic salts, and recrystallize the crude product from isopropanol to obtain the high-purity salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this salt-based synthesis route offers transformative benefits in terms of cost efficiency and supply reliability. By eliminating the need for silica gel column chromatography and complex chiral resolution steps required in older methods, the overall production cost is significantly reduced through the simplification of unit operations and the minimization of solvent consumption. The ability to achieve high chiral purity through straightforward recrystallization means that raw material costs are optimized, as lower grade starting materials can potentially be upgraded to specification during the process, driving substantial cost savings in the final bill of materials.
- Cost Reduction in Manufacturing: The streamlined process removes expensive and time-consuming purification steps, such as column chromatography, which are bottlenecks in traditional synthesis. By utilizing crystallization as the primary purification tool, the method drastically reduces solvent usage and waste disposal costs, leading to a more economical manufacturing footprint. Furthermore, the elimination of the acidification step prior to the final coupling reaction saves on reagent costs and reduces the complexity of the downstream process, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: The robust nature of the crystallization process ensures consistent batch-to-batch quality, minimizing the risk of production delays caused by failed purification attempts. Since the intermediate is isolated as a stable solid salt, it offers improved shelf-life and handling characteristics compared to oily or hygroscopic free acids, facilitating easier storage and transportation. This stability translates to a more resilient supply chain, capable of meeting Just-In-Time delivery schedules without the buffer stocks often required for less stable intermediates.
- Scalability and Environmental Compliance: The use of common, green solvents like isopropanol and water aligns with modern environmental, health, and safety (EHS) standards, simplifying regulatory compliance and permitting. The process is inherently scalable from kilogram to multi-ton quantities without the need for specialized equipment like preparative HPLC systems, ensuring that supply can be rapidly ramped up to meet market demand. This scalability, combined with reduced waste generation, positions the technology as a sustainable choice for long-term API production strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel intermediate synthesis. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical advantages of the salt formation strategy. .
Q: Why is the salt form of the Vibegron intermediate preferred over the sodium salt?
A: The salt forms (such as hydrochloride or tartrate) exhibit superior crystallinity compared to the sodium salt, allowing for effective purification via recrystallization. This significantly enhances both HPLC purity and chiral purity (ee%), which are critical for the final API quality.
Q: Which acid salt provides the best balance of yield and purity?
A: While various acids like L-tartaric and D-maleic acid are effective, the hydrochloride salt (specifically Crystal Form A) demonstrates the highest yield and chiral purity after recrystallization, making it the most suitable candidate for industrial scale-up.
Q: How does this intermediate simplify the final Vibegron synthesis?
A: Using the acid salt eliminates the need for an additional acidification step prior to the condensation reaction with Compound 7. The salt can react directly in the presence of a condensing agent, streamlining the process and reducing reagent consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vibegron Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the successful commercialization of life-saving medications like Vibegron. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of materials. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral purity and residual solvent limits, guaranteeing that every batch meets the exacting standards required for global pharmaceutical markets.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to delivering excellence in pharmaceutical intermediate manufacturing and supporting your journey from clinical trials to commercial launch.