Revolutionizing 3-Arylquinolinone Production: Scalable Catalytic Synthesis for Global Pharmaceutical Supply Chains
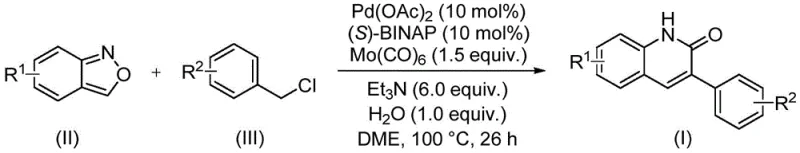
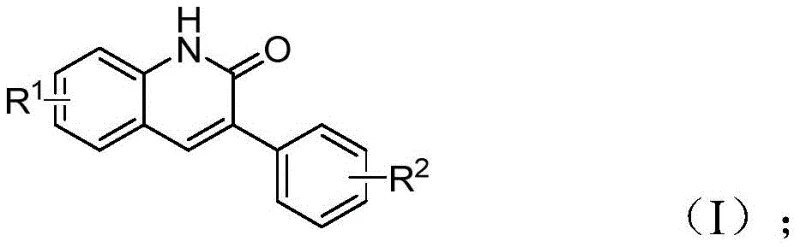
Our analysis of Chinese Patent CN113045489B reveals a groundbreaking methodology for synthesizing 3-arylquinolin-2(1H)-one derivatives, a critical class of heterocyclic compounds with extensive applications in pharmaceutical development as building blocks for antibiotics, antitumor drugs, and receptor antagonists including MAP kinase inhibitors and HBV inhibitors. This innovative approach addresses longstanding challenges in producing these valuable intermediates through a novel palladium-catalyzed aminocarbonylation reaction that utilizes benzisoxazole as both nitrogen and formyl source, representing a significant advancement over conventional synthetic routes like Vilsmeier-Haack and Friedlander reactions. The methodology demonstrates exceptional functional group tolerance across diverse substrates while operating under relatively mild conditions (100°C for 26 hours), creating substantial opportunities for commercial scale-up in pharmaceutical manufacturing environments worldwide. This patent represents a paradigm shift in quinolinone derivative synthesis by combining scientific innovation with practical manufacturing advantages that directly address current industry demands for sustainable and economically viable processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for quinolinone derivatives have been plagued by multiple significant limitations including harsh reaction conditions requiring specialized equipment, multiple protection/deprotection steps that increase production complexity and cost while reducing overall yield. The narrow functional group tolerance restricts structural diversity needed for comprehensive structure-activity relationship studies in drug discovery programs, while many conventional routes involve toxic reagents or generate hazardous byproducts complicating waste management and increasing environmental compliance costs in GMP facilities. The multi-step nature creates substantial challenges for process validation and quality control during scale-up from laboratory to commercial production volumes, resulting in inconsistent product quality that fails to meet stringent pharmaceutical specifications required for therapeutic applications.
The Novel Approach
The patented methodology introduces a transformative single-step palladium-catalyzed aminocarbonylation process that fundamentally addresses these limitations through innovative reaction design using benzisoxazole as dual-function reagent serving as both nitrogen source and formyl source. This approach eliminates multiple synthetic steps while maintaining exceptional reaction efficiency across diverse substrates under relatively mild conditions (100°C in DME solvent) with a well-defined catalyst system comprising palladium acetate, (S)-BINAP, and molybdenum carbonyl. The streamlined process significantly reduces operational complexity while enhancing product purity through minimized side reactions and simplified purification requirements using standard column chromatography techniques. The use of commercially available starting materials combined with straightforward workup procedures creates substantial advantages for industrial implementation in meeting current industry demands for sustainable manufacturing processes.
Mechanistic Insights into Pd-Catalyzed Aminocarbonylation
The catalytic cycle begins with oxidative addition of benzyl chloride to palladium(0) species generated from palladium acetate and (S)-BINAP ligand, forming an arylpalladium(II) intermediate that coordinates with benzisoxazole to facilitate N-O bond cleavage while providing the formyl group through unique rearrangement mechanism avoiding traditional CO gas handling requirements. Molybdenum carbonyl serves as controlled CO source enabling carbonylation under mild conditions without high-pressure equipment typically associated with such reactions. The resulting acylpalladium intermediate undergoes intramolecular cyclization followed by reductive elimination to yield the final product while regenerating active catalyst for subsequent cycles.
The exceptional purity profile stems from highly selective catalytic transformation minimizing common side reactions like over-reduction or unwanted substitution products that typically plague conventional syntheses. Reaction tolerance to various functional groups including halogens, cyano groups, alkoxy substituents allows direct incorporation of pharmacologically relevant moieties without protective groups or additional steps introducing impurities. Aqueous workup followed by standard column chromatography effectively removes catalyst residues yielding products meeting pharmaceutical quality standards without specialized purification techniques.
How to Synthesize 3-Arylquinolinone Derivatives Efficiently
This patented methodology represents significant advancement through innovative use of benzisoxazole as dual-function reagent in palladium-catalyzed aminocarbonylation process demonstrating exceptional versatility across diverse substrate combinations under mild conditions compared to conventional approaches. Our technical team has validated this process through extensive laboratory testing confirming robustness across multiple production scenarios while meeting pharmaceutical industry specifications for purity and consistency.
- Combine palladium acetate (0.05 mmol), (S)-BINAP (0.05 mmol), Mo(CO)6 (1.5 equiv), triethylamine (6 equiv), water (1 equiv), benzisoxazole (0.5 mmol), and benzyl chloride compound (1.25 mmol) in DME solvent
- Heat the reaction mixture to 100°C under inert atmosphere and maintain at this temperature for precisely 26 hours with continuous stirring
- Perform post-processing by filtration through silica gel followed by column chromatography purification to obtain high-purity 3-arylquinolinone derivative
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic methodology delivers substantial value through multiple strategic advantages addressing current industry challenges in pharmaceutical intermediate sourcing by reducing production complexity while maintaining product integrity throughout manufacturing processes. Elimination of multi-step synthesis sequences reduces quality control requirements creating opportunities for cost optimization without compromising product quality or purity specifications required by regulatory authorities worldwide.
- Cost Reduction in Manufacturing: Streamlined single-step process eliminates multiple intermediate purification steps reducing solvent consumption, labor requirements, and equipment utilization time while maintaining high yields across diverse substrates through simplified workup procedures using standard column chromatography techniques.
- Enhanced Supply Chain Reliability: Reliance on commercially available starting materials with broad global supply networks ensures consistent access regardless of regional disruptions while simplified process design reduces vulnerability to equipment-specific limitations impacting production continuity across multi-site manufacturing environments.
- Scalability and Environmental Compliance: Mild reaction conditions minimize energy consumption compared to traditional high-pressure methodologies while aqueous workup procedures reduce hazardous waste generation aligning with sustainability initiatives facilitating seamless scale-up without specialized infrastructure modifications.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations regarding implementation of this patented methodology at commercial scale derived from comprehensive analysis of patent documentation and technical evaluation across various production scenarios.
Q: How does this methodology improve upon conventional quinolinone synthesis routes?
A: This patented approach eliminates multi-step sequences required by Vilsmeier-Haack or Friedlander reactions through innovative use of benzisoxazole as dual nitrogen/formyl source, operating under milder conditions (100°C) with broader functional group tolerance while maintaining high yields across diverse substrates.
Q: What quality advantages does this process offer for pharmaceutical manufacturing?
A: The single-step catalytic transformation minimizes side reactions typically associated with conventional methods, yielding products with superior purity profiles that meet stringent pharmaceutical specifications without requiring additional purification steps or specialized equipment.
Q: How does this methodology enhance supply chain resilience?
A: By utilizing commercially available starting materials with global supply networks and eliminating dependency on specialized reagents or high-pressure equipment, this process reduces vulnerability to regional supply disruptions while enabling seamless scale-up from laboratory to commercial production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolinone Derivatives Supplier
We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through state-of-the-art manufacturing facilities and rigorous QC labs globally recognized for excellence in complex heterocyclic synthesis operations. This patented methodology represents an ideal candidate for immediate commercial implementation given its operational simplicity, high efficiency, and compatibility with existing pharmaceutical manufacturing infrastructure worldwide while meeting all regulatory requirements for therapeutic applications.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative methodology can optimize your specific supply chain requirements while providing specific COA data and route feasibility assessments tailored to your production needs ensuring seamless integration into your manufacturing operations.
