Advanced One-Pot Synthesis of 2,3,5-Trisubstituted Furans for Commercial Scale-Up
The landscape of organic synthesis is continually evolving to meet the rigorous demands of the pharmaceutical and advanced materials sectors, particularly for complex heterocyclic scaffolds. Patent CN114634469A introduces a groundbreaking preparation method for 2,3,5-trisubstituted furan compounds, a structural motif pervasive in bioactive natural products and functional materials. As illustrated in the structural diversity of naturally occurring furans such as Teubrevin G and (+)-Halenaquinone, as well as synthetic oligomers like Oligofuran used in organic semiconductors, the utility of this core structure is immense. 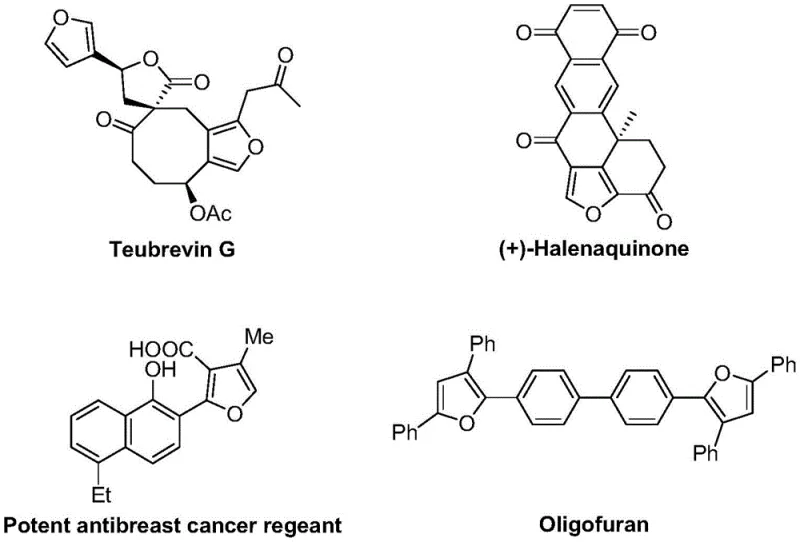 . This patent discloses a highly efficient, one-pot synthetic route that leverages a monovalent copper catalyst to drive the cycloisomerization of cyclopropylidene allenones with substituted alkynones. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this technology represents a significant leap forward in accessing high-purity furan derivatives with streamlined operational complexity.
. This patent discloses a highly efficient, one-pot synthetic route that leverages a monovalent copper catalyst to drive the cycloisomerization of cyclopropylidene allenones with substituted alkynones. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this technology represents a significant leap forward in accessing high-purity furan derivatives with streamlined operational complexity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 2,3,5-trisubstituted furan core has been fraught with synthetic challenges that hinder large-scale production. Traditional literature methods often rely on the activation of hydroxyl or ether groups in alcohols and phenols to induce intramolecular nucleophilic attacks on unsaturated bonds, or the activation of carbonyl groups in aldehydes and ketones. 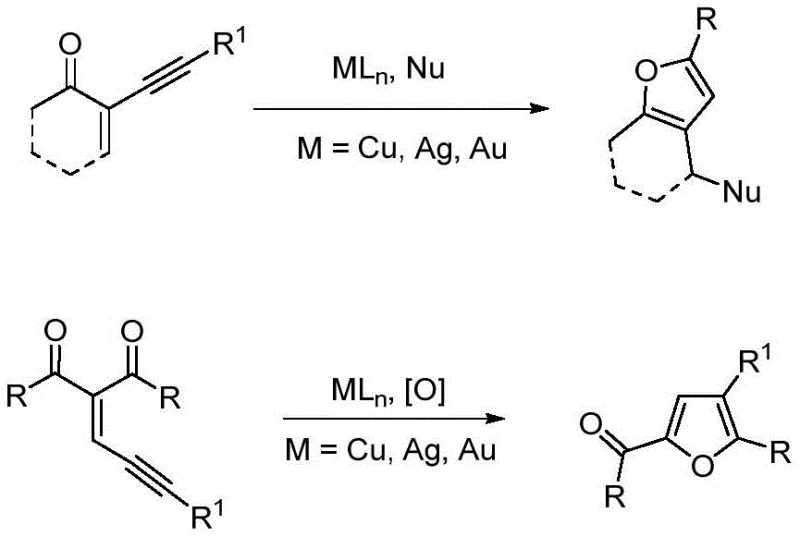 . These conventional pathways frequently necessitate the use of prohibitively expensive catalysts and specialized ligand systems, which drastically inflate the cost of goods sold (COGS). Furthermore, the substrate scope in these legacy methods is often narrowly defined, failing to accommodate diverse functional groups required for modern drug discovery. The multi-step nature of precursor synthesis and the harsh conditions often required for cyclization create significant bottlenecks in supply chain continuity and process safety, making them less attractive for commercial scale-up of complex pharmaceutical intermediates.
. These conventional pathways frequently necessitate the use of prohibitively expensive catalysts and specialized ligand systems, which drastically inflate the cost of goods sold (COGS). Furthermore, the substrate scope in these legacy methods is often narrowly defined, failing to accommodate diverse functional groups required for modern drug discovery. The multi-step nature of precursor synthesis and the harsh conditions often required for cyclization create significant bottlenecks in supply chain continuity and process safety, making them less attractive for commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology described in CN114634469A offers a robust solution by utilizing a direct coupling strategy between cyclopropylidene allenones and substituted alkynones. This novel approach eliminates the need for pre-functionalized substrates and expensive ligand systems, relying instead on a simple monovalent copper salt catalyst. The reaction operates under mild conditions, typically at room temperature (rt) to 50°C, which significantly reduces energy consumption compared to high-temperature thermal cyclizations. The one-pot nature of the reaction means that multiple bond-forming events occur in a single vessel, minimizing solvent usage and waste generation. This operational simplicity translates directly into cost reduction in fine chemical manufacturing, as it reduces the number of unit operations and purification steps required to isolate the final high-purity API intermediate. The broad substrate tolerance allows for the rapid generation of diverse chemical libraries, accelerating the lead optimization phase for medicinal chemistry teams.
Mechanistic Insights into Cu(I)-Catalyzed Cycloisomerization
The mechanistic pathway of this transformation is a fascinating example of cascade reactivity driven by ring strain and metal coordination. It is proposed that the monovalent copper salt first coordinates with the cyclopropylidene allenone, promoting a cycloisomerization reaction that expands the cyclopropyl ring to form a key furanocyclobutane intermediate. This highly reactive species then engages with the terminal alkynone through a highly regioselective Diels-Alder reaction, generating a transient oxanorbornadiene intermediate. The driving force for the final step is the release of ring strain; the oxanorbornadiene intermediate undergoes a retro-Diels-Alder fragmentation reaction, efficiently ejecting the strained components to yield the stable 2,3,5-trisubstituted furan product. 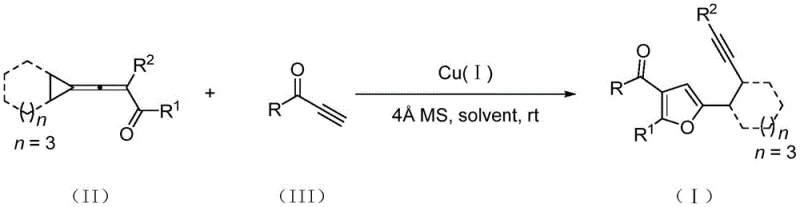 . Understanding this mechanism is critical for impurity control, as the high regioselectivity of the Diels-Alder step ensures that side products arising from alternative cyclization modes are minimized. The use of 4Å molecular sieves in the reaction mixture further aids in maintaining anhydrous conditions, preventing hydrolysis of sensitive intermediates and ensuring consistent batch-to-batch reproducibility.
. Understanding this mechanism is critical for impurity control, as the high regioselectivity of the Diels-Alder step ensures that side products arising from alternative cyclization modes are minimized. The use of 4Å molecular sieves in the reaction mixture further aids in maintaining anhydrous conditions, preventing hydrolysis of sensitive intermediates and ensuring consistent batch-to-batch reproducibility.
How to Synthesize 2,3,5-Trisubstituted Furan Efficiently
Implementing this synthesis requires careful attention to reagent stoichiometry and reaction environment to maximize yield and purity. The protocol dictates that the cyclopropylidene allenone should be used in excess relative to the substituted alkynone, typically in a molar ratio ranging from 1:1 to 2:1, to suppress potential homodimerization of the allenone. The detailed standardized synthesis steps involve charging a reaction vessel with activated molecular sieves, the copper catalyst, and the substrates in a halogenated solvent like chloroform, followed by stirring at ambient temperature.
- Add molecular sieves, monovalent copper salt, cyclopropylidene allenone, and substituted alkynone into an organic solvent such as chloroform.
- Stir the mixture at room temperature (20-50°C) for 10 to 20 hours until the reaction is complete.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to obtain the target 2,3,5-trisubstituted furan compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic advantages beyond mere chemical novelty. The shift towards a one-pot, ligand-free copper catalysis system fundamentally alters the cost structure of producing these valuable intermediates. By removing the dependency on precious metal catalysts and complex phosphine ligands, the raw material costs are significantly reduced, allowing for more competitive pricing in the global market. Additionally, the use of readily available starting materials, such as commercially sourced cyclopropylidene acetylene derivatives and substituted iodobenzenes, enhances supply chain reliability and reduces lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive ligands and the use of base metal copper catalysts result in substantial cost savings per kilogram of product. The mild reaction conditions (room temperature) lower energy overheads, while the simplified workup procedure involving filtration and standard column chromatography reduces labor and solvent costs associated with complex extractions or recrystallizations.
- Enhanced Supply Chain Reliability: The robustness of the reaction across a wide range of electronic and steric environments means that supply disruptions due to specific substrate failures are minimized. The ability to source precursors from multiple vendors without compromising reaction efficiency ensures a resilient supply chain capable of meeting fluctuating demand schedules for clinical and commercial batches.
- Scalability and Environmental Compliance: The one-pot design inherently reduces the environmental footprint by minimizing solvent transfers and waste generation. The process is amenable to scale-up from gram to multi-kilogram quantities without requiring specialized high-pressure or high-temperature equipment, facilitating a smoother transition from process development to commercial production while adhering to strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy for process development teams evaluating this route for their own pipelines.
Q: What catalyst system is utilized in this novel furan synthesis?
A: The process employs a monovalent copper salt, specifically copper tetrafluoroborate tetraacetonitrile (Cu(MeCN)4BF4), which demonstrates high catalytic efficiency without the need for expensive external ligands.
Q: What are the optimal reaction conditions for this transformation?
A: The reaction proceeds efficiently at mild temperatures ranging from 20°C to 50°C (room temperature is preferred) over a period of 10 to 20 hours using halogenated aprotic solvents like chloroform.
Q: Does this method support a broad range of substrate substituents?
A: Yes, the methodology exhibits excellent substrate tolerance, accommodating various groups including phenyl, naphthyl, heterocycles (furan, thiophene), and substituted benzenes with electron-donating or withdrawing groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3,5-Trisubstituted Furan Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and material science applications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify identity and assay. Whether you require custom synthesis of novel furan derivatives or bulk supply of established intermediates, our infrastructure is designed to support your long-term growth.
We invite you to engage with our technical procurement team to discuss how this innovative copper-catalyzed route can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this methodology. We encourage potential partners to contact us directly to obtain specific COA data for relevant compounds and to schedule a consultation for route feasibility assessments tailored to your unique molecular targets.
