Revolutionizing Pharmaceutical Intermediate Production Through Copper-Catalyzed Indole Synthesis at Commercial Scale
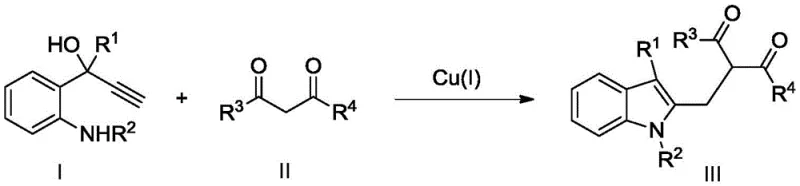
The patent CN109851548B introduces a groundbreaking methodology for synthesizing 2,3-disubstituted indole compounds, representing a significant advancement in the field of organic synthesis for pharmaceutical applications. This innovative approach utilizes monovalent copper catalysts to facilitate cyclization tandem reactions between ortho-propargyl alcohol substituted aniline compounds and dicarbonyl compounds under remarkably mild conditions. The process achieves product yields ranging from 62% to 78% while generating minimal byproducts, addressing critical limitations in traditional synthetic routes. Furthermore, the methodology demonstrates exceptional scalability potential from laboratory to industrial production volumes, making it particularly valuable for manufacturers requiring high-purity intermediates. The environmental benefits of this copper-catalyzed system align with growing industry demands for sustainable manufacturing practices, positioning it as a strategic solution for modern pharmaceutical supply chains. This patent represents a paradigm shift in indole compound synthesis that directly addresses the evolving needs of global pharmaceutical manufacturers seeking both technical excellence and operational efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for producing 2,3-disubstituted indole compounds have historically relied on noble metal catalysts such as gold, silver, or platinum, creating significant economic and operational challenges for pharmaceutical manufacturers. These precious metal systems often require stringent reaction conditions including elevated temperatures and inert atmospheres, substantially increasing energy consumption and operational complexity. The generation of multiple byproducts during these reactions necessitates extensive purification processes that dramatically reduce overall process efficiency and yield valuable material. Furthermore, the high cost of noble metal catalysts represents a substantial financial burden that directly impacts the cost structure of final pharmaceutical products. Supply chain vulnerabilities associated with precious metal sourcing create additional risks for consistent production scheduling and inventory management. The environmental footprint of these conventional methods is also considerable due to the need for specialized waste treatment procedures to handle residual metal contaminants. These combined limitations have long constrained the commercial viability of indole-based pharmaceutical intermediates despite their significant therapeutic potential.
The Novel Approach
The patented methodology overcomes these limitations through an elegant copper-catalyzed cyclization tandem reaction that operates under remarkably mild conditions between 40°C and 80°C without requiring inert atmospheres. By utilizing inexpensive monovalent copper catalysts such as cuprous iodide or copper hexachlorophosphate tetraacetylamide, the process eliminates the need for costly noble metals while maintaining high reaction efficiency. The system demonstrates exceptional selectivity with minimal byproduct formation, directly translating to simplified purification workflows and higher overall yields ranging from 62% to 78%. The flexibility in solvent selection—including readily available options like dichloromethane and chloroform—further enhances operational practicality for manufacturing facilities. This approach significantly reduces both capital expenditure requirements and operational costs while improving environmental sustainability through reduced energy consumption and waste generation. The methodology's compatibility with standard manufacturing equipment enables seamless integration into existing production facilities without requiring specialized infrastructure investments.
Mechanistic Insights into Copper-Catalyzed Cyclization
The reaction mechanism involves a sophisticated copper-mediated cyclization process where monovalent copper activates the alkyne functionality of the ortho-propargyl alcohol substituted aniline compound, facilitating nucleophilic attack by the amine group. This initial cyclization forms a key indole intermediate that subsequently undergoes addition to the dicarbonyl compound through a Michael-type reaction pathway. The copper catalyst plays a dual role in both activating the alkyne and stabilizing the enol intermediate during the tandem reaction sequence. The precise control over reaction parameters—particularly temperature maintenance between 40°C and 80°C—ensures optimal catalyst activity while preventing undesired side reactions. The molar ratio of aniline to dicarbonyl compound (1:5) creates ideal stoichiometric conditions that maximize conversion efficiency while minimizing unreacted starting materials. This mechanistic understanding reveals why the process achieves such high selectivity with minimal byproduct formation compared to traditional approaches.
The exceptional purity profile achieved by this methodology stems from the inherent selectivity of the copper-catalyzed tandem reaction mechanism. The absence of competing reaction pathways that typically generate impurities in noble metal-catalyzed systems results in cleaner reaction profiles that require less intensive purification. The mild reaction conditions prevent thermal degradation of sensitive intermediates, preserving molecular integrity throughout the process. Careful optimization of catalyst loading (0.5-50 mol%) ensures complete conversion without promoting side reactions that could introduce impurities. The solvent system's compatibility with standard chromatographic purification techniques further enhances the ability to achieve stringent purity specifications required for pharmaceutical intermediates. This combination of factors delivers products with consistently high purity levels that meet the rigorous quality standards demanded by global pharmaceutical manufacturers.
How to Synthesize 2,3-Disubstituted Indole Compounds Efficiently
This patented synthetic route represents a significant advancement in the production of complex indole-based pharmaceutical intermediates through its innovative use of copper catalysis under mild reaction conditions. The methodology provides a robust framework for manufacturing high-purity compounds essential for advanced drug development pipelines while addressing critical cost and scalability challenges faced by modern pharmaceutical manufacturers. By eliminating reliance on expensive noble metal catalysts and operating within practical temperature ranges, this process delivers substantial operational advantages without compromising product quality or yield. The detailed standardized synthesis steps outlined below provide a clear pathway for implementation in commercial manufacturing environments, ensuring consistent production outcomes across varying scales. This approach exemplifies how strategic process innovation can transform challenging synthetic targets into commercially viable manufacturing operations.
- Dissolve ortho-propargyl alcohol substituted aniline compound (0.1 mmol) in organic solvent under air atmosphere
- Add dicarbonyl compound (0.5 mmol) and monovalent copper catalyst (0.01 mmol) to the reaction mixture
- Stir at controlled temperature (40-80°C) for specified duration (4-24 hours) before column chromatography purification
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing process directly addresses critical pain points in pharmaceutical supply chains by delivering solutions that enhance both cost efficiency and operational reliability. The elimination of expensive noble metal catalysts creates immediate cost advantages while simultaneously reducing supply chain vulnerabilities associated with precious metal sourcing. The simplified reaction profile minimizes downstream processing requirements, leading to more predictable production timelines and improved resource allocation across manufacturing facilities. These advantages collectively strengthen procurement strategies by providing greater flexibility in raw material sourcing and reducing exposure to volatile market conditions affecting traditional catalyst systems. The methodology's compatibility with standard manufacturing infrastructure further enhances its value proposition by eliminating capital expenditure requirements for specialized equipment.
- Cost Reduction in Manufacturing: The substitution of inexpensive copper catalysts for precious metals creates substantial cost savings throughout the production process by eliminating expensive catalyst procurement and associated purification steps required to remove residual noble metals. This approach significantly reduces raw material expenses while simultaneously lowering waste treatment costs due to minimal byproduct formation. The simplified purification workflow further contributes to operational cost reductions by decreasing solvent consumption and processing time. These combined factors deliver meaningful economic benefits without requiring additional capital investment or process modifications.
- Enhanced Supply Chain Reliability: The use of readily available copper catalysts and common organic solvents substantially improves supply chain resilience by reducing dependence on specialized or constrained raw materials. The process's tolerance for standard atmospheric conditions eliminates the need for specialized inert gas handling systems, enhancing production flexibility across diverse manufacturing environments. This operational robustness ensures consistent production output even during periods of raw material volatility or logistical disruptions. The methodology's compatibility with existing manufacturing infrastructure further strengthens supply chain continuity by enabling rapid implementation without requiring facility modifications.
- Scalability and Environmental Compliance: The mild reaction conditions (40-80°C) and straightforward process design enable seamless scale-up from laboratory to commercial production volumes without requiring specialized equipment modifications. The reduced environmental footprint resulting from lower energy consumption and minimal waste generation aligns with increasingly stringent regulatory requirements for sustainable manufacturing practices. This approach facilitates compliance with global environmental standards while supporting corporate sustainability initiatives through reduced resource consumption and waste generation. The process's inherent simplicity enhances operational flexibility across different production scales while maintaining consistent product quality.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations regarding the implementation of this patented copper-catalyzed synthesis methodology for pharmaceutical intermediate production. These insights are derived directly from the patent documentation and reflect practical considerations for manufacturing operations seeking to adopt this innovative approach. Each response provides actionable information to support informed decision-making regarding process implementation and optimization strategies.
Q: How does this copper-catalyzed method improve upon traditional noble metal approaches?
A: The process eliminates expensive gold/silver catalysts while maintaining high yields (62-78%), significantly reducing raw material costs and simplifying purification by minimizing byproduct formation.
Q: What scalability advantages does this patent offer for pharmaceutical manufacturing?
A: Mild reaction conditions (40-80°C) and solvent flexibility enable seamless transition from laboratory to industrial scale without specialized equipment, ensuring reliable supply chain continuity.
Q: How does the methodology address purity requirements for pharmaceutical intermediates?
A: The near-absence of byproducts and precise control over reaction parameters facilitate stringent purity specifications essential for API intermediate production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-disubstituted indole compounds Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring seamless transition from laboratory development to full-scale manufacturing operations. We maintain stringent purity specifications through rigorous QC labs equipped with advanced analytical capabilities that guarantee consistent product quality meeting global pharmaceutical standards. Our technical expertise in copper-catalyzed methodologies enables precise control over critical quality attributes while optimizing process efficiency for maximum economic benefit. As a trusted partner in complex intermediate synthesis, we provide comprehensive support from route validation through commercial production, leveraging our deep understanding of regulatory requirements and manufacturing best practices.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this patented methodology can optimize your specific manufacturing requirements. Please contact us to obtain detailed COA data and route feasibility assessments tailored to your production needs, enabling informed decisions about implementing this innovative synthesis approach within your supply chain operations.
