Advanced Quinoxothiazole Compound Synthesis: Streamlined Manufacturing with Enhanced Scalability and Purity
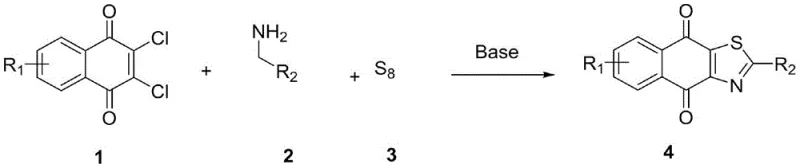
This technical report analyzes Chinese Patent CN116253697A, which discloses a novel one-pot synthesis method for quinoxothiazole compounds using dichloronaphthoquinone and methylamine derivatives as starting materials. The patented process represents a significant advancement in heterocyclic compound manufacturing by eliminating the need for toxic oxidants and noble metal catalysts that have traditionally been required in such syntheses. This innovation addresses critical challenges in pharmaceutical intermediate production where purity, cost efficiency, and environmental sustainability are paramount concerns for global pharmaceutical manufacturers. The method's mild reaction conditions and high substrate tolerance offer substantial advantages over conventional multi-step approaches that often involve hazardous reagents and complex purification procedures. By leveraging simple alkali promotion rather than expensive catalytic systems, this technology provides a more economically viable pathway to these biologically important compounds. The patent demonstrates exceptional versatility across diverse substrate combinations while maintaining excellent yield profiles and product purity standards required by the pharmaceutical industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to quinoxothiazole compounds have historically relied on multi-step procedures that require stoichiometric amounts of toxic oxidizing agents, creating significant environmental and safety concerns for manufacturing facilities. These conventional methods often necessitate expensive noble metal catalysts for arylation steps, substantially increasing production costs while introducing potential metal contamination risks that complicate purification for pharmaceutical applications. The harsh reaction conditions typically employed in existing syntheses frequently lead to poor selectivity and increased formation of undesired byproducts, requiring complex and time-consuming purification processes that reduce overall process efficiency. Furthermore, the limited substrate scope of many established methods restricts their applicability to only specific structural variants, forcing manufacturers to develop customized synthetic routes for different target molecules. The requirement for specialized equipment to handle hazardous reagents adds capital expenditure burdens and operational complexities that are particularly challenging for contract manufacturing organizations serving multiple clients with varying requirements. These cumulative limitations have created persistent challenges in achieving cost-effective, scalable, and environmentally sustainable production of quinoxothiazole-based pharmaceutical intermediates.
The Novel Approach
The patented methodology introduces an elegant solution through an alkali-promoted one-pot reaction that simultaneously addresses multiple limitations of conventional syntheses by utilizing readily available starting materials under mild conditions. This innovative process eliminates the need for external oxidants by leveraging elemental sulfur as both the sulfur source and oxidation agent within the reaction system, significantly enhancing process safety and environmental profile. The use of common alkaline promoters such as sodium carbonate instead of expensive transition metal catalysts reduces raw material costs while eliminating concerns about metal contamination in the final product. The reaction demonstrates remarkable substrate tolerance across diverse functional groups, enabling the synthesis of a wide range of quinoxothiazole derivatives without requiring significant process modifications. With optimized conditions operating at moderate temperatures (80°C–120°C) and standard atmospheric pressure, the method is readily adaptable to existing manufacturing infrastructure without requiring specialized equipment investments. The simplified workup procedure involving basic extraction and chromatography delivers high-purity products suitable for pharmaceutical applications while substantially reducing processing time compared to traditional multi-step approaches.
Mechanistic Insights into Alkali-Promoted One-Pot Synthesis
The reaction mechanism begins with the nucleophilic attack of the amine group from the methylamine derivative on one of the carbonyl groups of dichloronaphthoquinone, forming an intermediate imine species through condensation. This initial step is facilitated by the alkaline environment which deprotonates the amine, enhancing its nucleophilicity while simultaneously promoting chloride displacement from the quinone ring system. The resulting intermediate then undergoes intramolecular cyclization with elemental sulfur, where the base catalyst plays a dual role in activating both the sulfur species and facilitating the elimination steps required for aromatization. The sulfur incorporation occurs through a series of electron transfer processes that enable the formation of the thiazole ring without requiring external oxidants, as the base-promoted conditions facilitate the necessary oxidation state changes internally within the reaction system. This elegant cascade process demonstrates excellent atom economy by utilizing all starting materials efficiently without generating significant stoichiometric byproducts that would require disposal or treatment.
The mechanism inherently controls impurity formation through its high selectivity for the desired cyclization pathway, minimizing side reactions that could lead to impurities problematic for pharmaceutical applications. The mild reaction conditions prevent decomposition pathways that might occur under more aggressive synthetic approaches, while the one-pot nature eliminates intermediate isolation steps that could introduce contaminants. The alkaline environment helps neutralize any acidic byproducts that might form during the reaction, preventing potential degradation pathways that could compromise product quality. The solvent system (preferably DMSO) provides optimal solubility for all reaction components while maintaining stability throughout the process window, further contributing to consistent product quality. This combination of factors results in a process that delivers high-purity quinoxothiazole compounds with minimal impurities that would require extensive purification before pharmaceutical use.
How to Synthesize Quinoxothiazole Compounds Efficiently
This patented methodology represents a significant advancement in quinoxothiazole synthesis through its innovative one-pot approach that eliminates multiple processing steps while maintaining excellent yield and purity profiles. The process leverages simple alkaline promotion rather than expensive catalytic systems, making it particularly attractive for large-scale manufacturing operations seeking cost-effective solutions without compromising quality standards. By utilizing readily available starting materials under mild reaction conditions, this method offers substantial operational advantages over conventional multi-step syntheses that require hazardous reagents and complex purification procedures. The following standardized synthesis procedure provides detailed guidance for implementing this technology in industrial settings while ensuring consistent product quality and process reliability.
- Prepare the reaction mixture by combining dichloronaphthoquinone, methylamine derivative, and elemental sulfur in DMSO solvent with sodium carbonate as the base promoter at optimized molar ratios.
- Heat the reaction mixture to 100°C under nitrogen atmosphere for four hours to ensure complete conversion while maintaining mild reaction conditions that prevent decomposition.
- Perform post-reaction processing by cooling to room temperature, extracting with ethyl acetate-water mixture, drying over anhydrous sodium sulfate, and purifying via column chromatography using petroleum ether/dichloromethane eluent.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis method delivers substantial commercial benefits that directly address critical pain points faced by procurement and supply chain professionals in pharmaceutical manufacturing organizations. By eliminating reliance on expensive noble metal catalysts and toxic oxidants, the process significantly reduces raw material costs while simultaneously improving supply chain resilience through the use of more readily available starting materials. The simplified reaction sequence reduces processing time and equipment requirements, enabling faster turnaround times without compromising product quality or regulatory compliance standards required for pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and toxic oxidants substantially reduces raw material expenses while avoiding costly metal removal steps required in traditional syntheses. The simplified one-pot procedure minimizes solvent usage and processing time compared to multi-step approaches, resulting in significant operational cost savings throughout the manufacturing process without requiring major capital investments in new equipment.
- Enhanced Supply Chain Reliability: The use of stable, commercially available starting materials with long shelf lives reduces vulnerability to supply chain disruptions commonly associated with specialized reagents required in conventional methods. The process's robustness across various substrate types allows for greater flexibility in raw material sourcing while maintaining consistent product quality, providing procurement teams with more reliable supply options and reduced risk of production delays.
- Scalability and Environmental Compliance: The mild reaction conditions and straightforward workup procedure enable seamless scale-up from laboratory to commercial production volumes without requiring significant process re-engineering. The elimination of hazardous reagents substantially reduces environmental impact while simplifying waste treatment requirements, aligning with increasingly stringent regulatory standards for sustainable manufacturing practices in the pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address common concerns from technical procurement teams regarding implementation of this innovative synthesis method for quinoxothiazole compounds. These answers are based on detailed analysis of the patent documentation and reflect practical considerations for manufacturing scale-up and quality assurance protocols.
Q: How does this method eliminate the need for toxic oxidants compared to conventional approaches?
A: The alkali-promoted one-pot reaction utilizes elemental sulfur as both sulfur source and oxidation agent within the system, where base catalysts simultaneously facilitate cyclization and oxidation steps without requiring external toxic oxidants typically needed in traditional syntheses.
Q: What are the key advantages of this process for supply chain reliability?
A: The process employs stable, commercially available starting materials with long shelf lives under mild reaction conditions, reducing vulnerability to supply chain disruptions while maintaining consistent product quality across diverse substrate types.
Q: How does this method ensure high purity without complex purification steps?
A: The reaction's high selectivity minimizes byproduct formation through controlled cyclization pathways, while straightforward workup procedures involving simple extraction deliver pharmaceutical-grade purity without requiring specialized purification equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoxothiazole Compound Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex heterocyclic compounds while maintaining stringent purity specifications required by global pharmaceutical clients. As a specialized CDMO with dedicated R&D facilities and rigorous QC labs, we have successfully implemented this patented methodology to produce high-purity quinoxothiazole intermediates with exceptional batch-to-batch consistency. Our technical team has optimized this process for industrial-scale manufacturing while preserving all the advantages described in the patent documentation, ensuring reliable supply of these critical building blocks for drug development programs worldwide.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis approach can benefit your specific manufacturing requirements. Contact us today to obtain specific COA data and route feasibility assessments tailored to your production needs.
