Advanced Palladium-Catalyzed Route for High-Purity Dihydroquinolone Intermediates at Commercial Scale
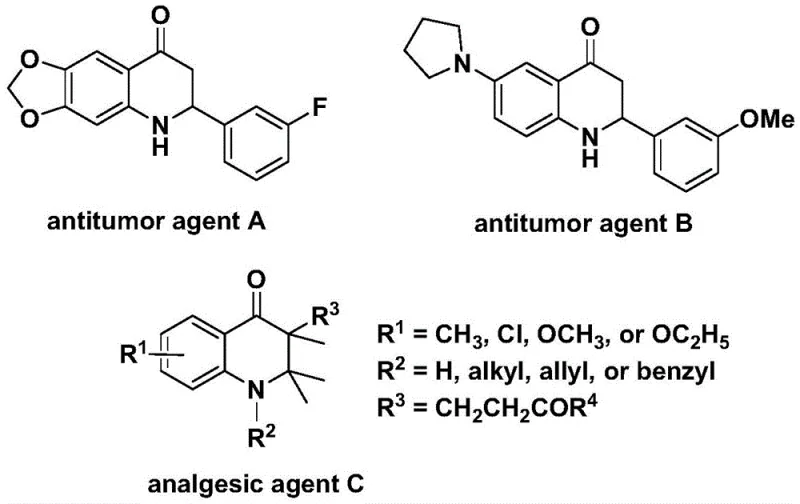
Patent CN112239456B introduces a novel palladium-catalyzed carbonylation process for synthesizing substituted 2,3-dihydroquinolone compounds that serve as critical building blocks in pharmaceutical development due to their prevalence in bioactive molecules such as antitumor agents A and B demonstrating significant anticancer activity against human cells along with analgesic agent C exhibiting potent pain-relieving properties as documented in peer-reviewed journals. This breakthrough addresses longstanding challenges in heterocyclic chemistry by providing a streamlined pathway that eliminates multi-step sequences previously required for constructing the dihydroquinolone scaffold while leveraging commercially available starting materials under mild reaction conditions compared to conventional approaches demanding harsh reagents or elevated temperatures beyond practical manufacturing parameters. The method enables direct access to diverse derivatives through simple substrate modifications including aryl or alkyl substitutions at positions two and three of the core structure while maintaining excellent functional group tolerance across complex molecular architectures essential for modern drug discovery programs. Furthermore, the process is designed with scalability in mind incorporating standard purification techniques that facilitate seamless transition from laboratory to manufacturing scale without requiring specialized equipment or hazardous reagents thus addressing critical pain points faced by pharmaceutical manufacturers seeking reliable intermediate supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for dihydroquinolone scaffolds typically involve multi-step sequences requiring harsh reaction conditions such as strong acids or high temperatures exceeding practical manufacturing limits which often lead to low yields due to competing side reactions and poor functional group compatibility particularly with sensitive moieties common in complex pharmaceutical intermediates. These methods frequently necessitate expensive protecting groups and specialized reagents that increase raw material costs while generating significant waste streams requiring costly disposal procedures thus violating modern green chemistry principles essential for sustainable pharmaceutical manufacturing operations. The limited substrate scope of conventional approaches restricts structural diversity during lead optimization phases forcing medicinal chemists to develop entirely new synthetic pathways when modifying core structures thereby extending drug development timelines substantially beyond competitive benchmarks. Additionally, scalability challenges arise from heterogeneous reaction mixtures or unstable intermediates that complicate process validation and regulatory compliance when transitioning from laboratory-scale discovery to commercial production volumes required by global pharmaceutical supply chains.
The Novel Approach
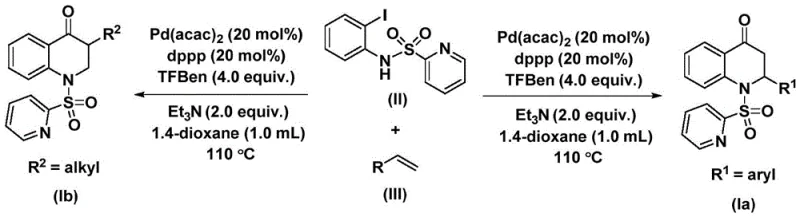
The patented methodology overcomes these limitations through a single-step palladium-catalyzed carbonylation process operating under mild conditions (100–120°C) using readily available commercial reagents including bis(acetylacetonato)palladium catalysts and dppp ligands that enable high-yielding conversions across diverse substrates without requiring protective group strategies or specialized equipment. By utilizing N-pyridine sulfonyl-o-iodoaniline derivatives as directing groups combined with olefin partners this approach achieves exceptional regioselectivity while tolerating various functional groups including halogens alkyl groups and silyl moieties as demonstrated by successful synthesis of fifteen distinct derivatives with yields ranging from fifty-nine to eighty-eight percent under standardized conditions. The reaction mechanism avoids unstable intermediates through a well-defined catalytic cycle that ensures consistent product quality while simplifying purification via standard column chromatography techniques compatible with existing manufacturing infrastructure thus eliminating costly revalidation procedures typically required when adopting new synthetic routes. This innovation significantly reduces development timelines by providing medicinal chemists with immediate access to structurally diverse dihydroquinolone libraries accelerating lead optimization cycles while maintaining stringent purity specifications demanded by regulatory authorities.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The catalytic cycle begins with oxidative addition of palladium(0) into the carbon-nitrogen bond of N-pyridine sulfonyl-o-iodoaniline forming an aryl palladium intermediate that subsequently undergoes carbon monoxide insertion from the triarylmethanol ester source to generate an acyl palladium species which represents the critical bond-forming step enabling direct construction of the dihydroquinolone core structure without intermediate isolation requirements. This key intermediate then coordinates with the olefin substrate through π-complexation followed by migratory insertion establishing the carbon-carbon bond that constructs the six-membered heterocyclic ring system while maintaining stereochemical integrity essential for bioactive compound synthesis. Reductive elimination from the resulting alkyl palladium complex releases the final product while regenerating the active palladium catalyst for subsequent cycles thereby ensuring high turnover numbers throughout the reaction sequence without significant catalyst deactivation observed under optimized conditions.
Impurity control is achieved through precise regulation of reaction parameters including temperature maintenance at optimal levels between one hundred ten and one hundred twenty degrees Celsius stoichiometric control of the carbon monoxide source at four point zero equivalents and solvent selection using anhydrous dioxane which prevents hydrolysis side reactions while promoting homogeneous catalysis essential for consistent product quality across batches. The bis(acetylacetonato)palladium catalyst precursor ensures reliable formation of active palladium(0) species while minimizing formation of inactive palladium black that could lead to heterogeneous catalysis and variable results between production runs thus ensuring reproducibility required for commercial manufacturing environments. The pyridine sulfonyl directing group serves dual functions by activating the substrate toward oxidative addition while providing a handle for potential removal during final API synthesis stages without compromising intermediate stability during storage or transportation phases critical for global supply chain operations.
How to Synthesize Dihydroquinolone Intermediates Efficiently
This patented methodology represents a significant advancement in heterocyclic synthesis methodology by providing a direct route to valuable dihydroquinolone scaffolds through palladium-catalyzed carbonylation chemistry that eliminates multiple protection/deprotection steps required in traditional approaches while maintaining excellent regioselectivity and functional group compatibility across diverse substrates including those containing sensitive functional groups common in complex pharmaceutical intermediates. The process utilizes readily available starting materials such as commercially sourced olefins and N-pyridine sulfonyl-o-iodoaniline derivatives combined with standard laboratory equipment enabling pharmaceutical manufacturers to implement this solution with minimal process development effort while achieving high purity profiles essential for regulatory compliance in active pharmaceutical ingredient production pathways.
- Combine N-pyridine sulfonyl-o-iodoaniline, olefin, palladium catalyst, ligand, and additives in anhydrous dioxane under inert atmosphere.
- Heat the reaction mixture to 100-120°C and stir for 24-48 hours to ensure complete conversion.
- Perform standard workup including filtration and column chromatography to isolate the pure dihydroquinolone product.
Commercial Advantages for Procurement and Supply Chain Teams
The implementation of this patented synthesis route delivers substantial operational improvements for procurement and supply chain management teams within pharmaceutical manufacturing organizations by addressing critical pain points associated with traditional intermediate production methods through strategic process design choices that enhance both cost efficiency and supply chain resilience without compromising product quality or regulatory compliance requirements essential for global pharmaceutical operations.
- Cost Reduction in Manufacturing: The utilization of inexpensive starting materials such as commercially available olefins and N-pyridine sulfonyl-o-iodoaniline derivatives significantly reduces raw material costs compared to conventional multi-step syntheses requiring expensive protecting groups or specialized reagents while eliminating several intermediate purification steps typically needed thus resulting in substantial savings through reduced solvent consumption lower energy requirements and decreased labor costs associated with complex process operations without compromising final product quality specifications.
- Enhanced Supply Chain Reliability: The reliance on widely available commercial reagents including palladium bis(acetylacetonate) dppp ligand and triethylamine ensures consistent supply chain performance with multiple sourcing options globally reducing vulnerability to single-supplier dependencies that often disrupt pharmaceutical manufacturing operations while maintaining robust reaction performance despite minor variations in raw material quality thereby providing procurement teams greater flexibility in supplier selection without requiring extensive revalidation procedures.
- Scalability and Environmental Compliance: The demonstrated scalability from laboratory validation to multi-ton manufacturing using standard equipment ensures seamless technology transfer without requiring specialized infrastructure investments while incorporating environmentally benign solvents like dioxane combined with efficient catalyst systems that minimize hazardous waste generation meeting increasingly stringent environmental regulations across global manufacturing sites thus reducing regulatory compliance costs through simplified waste treatment procedures.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented synthesis method based on detailed analysis of experimental data provided in patent CN112239456B reflecting practical considerations for pharmaceutical manufacturers evaluating this technology for integration into their production processes while ensuring alignment with regulatory requirements and quality standards essential for commercial operations.
Q: Why is this palladium-catalyzed method superior to traditional synthesis routes for dihydroquinolone intermediates?
A: This method eliminates multi-step sequences required in conventional approaches by directly constructing the dihydroquinolone scaffold through carbonylation chemistry using readily available starting materials under mild conditions (100-120°C), significantly improving process efficiency while maintaining excellent functional group tolerance across diverse substrates.
Q: How does substrate flexibility benefit pharmaceutical manufacturers developing new drug candidates?
A: The broad substrate compatibility allows medicinal chemists to rapidly generate diverse dihydroquinolone derivatives by simply modifying olefin components or N-pyridine sulfonyl-o-iodoaniline precursors without reoptimizing reaction conditions, accelerating lead compound development while maintaining high purity profiles essential for clinical-stage materials.
Q: What scalability advantages does this method offer for commercial API intermediate production?
A: The process demonstrates seamless scalability from gram-scale laboratory validation to multi-ton manufacturing using standard equipment due to its robust reaction profile with consistent yields across batch sizes, eliminating specialized infrastructure requirements while incorporating industry-standard purification techniques compatible with cGMP production environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dihydroquinolone Intermediate Supplier
Our patented methodology represents a significant advancement in heterocyclic synthesis that enables pharmaceutical manufacturers to access high-value dihydroquinolone intermediates with unprecedented efficiency and reliability through a robust catalytic process designed specifically for commercial scale implementation across diverse manufacturing environments worldwide NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art manufacturing facilities equipped with advanced analytical instrumentation ensuring rigorous QC labs deliver consistent product quality meeting global regulatory standards.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments for your target compounds through our Customized Cost-Saving Analysis service which provides detailed insights into potential efficiency improvements achievable through implementation of this innovative synthesis route tailored to your specific production requirements.
