Revolutionizing Pharmaceutical Intermediates: Scalable and Sustainable Synthesis of 1,4-Naphthoquinocyclopropane Compounds
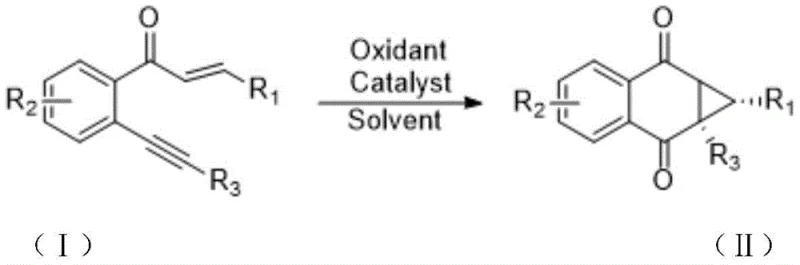
The groundbreaking Chinese patent CN109896944B introduces a transformative methodology for synthesizing structurally complex 1,4-naphthoquinocyclopropane compounds, representing a significant advancement in the field of pharmaceutical intermediate manufacturing. This innovative approach addresses critical limitations in conventional naphthoquinone production by employing a one-pot cyclization strategy that operates under remarkably mild conditions while maintaining exceptional substrate versatility. The process leverages iron-based catalysis to achieve direct conversion of readily available o-alkynyl chalcone precursors into valuable cyclopropane-fused quinones without requiring hazardous reagents or extreme thermal parameters. This patent demonstrates substantial industrial potential through its environmentally benign profile and operational simplicity, offering pharmaceutical manufacturers a viable pathway to produce high-value intermediates with enhanced sustainability metrics. The methodology's compatibility with standard manufacturing equipment further positions it as an immediately implementable solution for global API producers seeking greener synthesis routes without capital-intensive retooling.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to naphthoquinone synthesis suffer from multiple critical deficiencies that hinder their industrial applicability and environmental sustainability. Chromium trioxide-based oxidation methods require highly corrosive acidic conditions and generate substantial toxic chromium waste streams that necessitate expensive remediation procedures, creating significant regulatory compliance burdens for manufacturers. Liquid-phase naphthalene oxidation processes demand high-energy input while producing complex byproduct mixtures that complicate purification and reduce overall yield efficiency. Indirect electrolytic oxidation using high-valence heavy metal salts introduces additional separation challenges and metal contamination risks that are particularly problematic for pharmaceutical applications requiring ultra-high purity standards. Conventional cyclopropanation techniques like Freund's reaction involve hazardous metallic sodium handling with explosion risks, while ylide and carbene methods exhibit notoriously low atom economy generating excessive waste streams that conflict with modern green chemistry principles. These limitations collectively result in higher production costs, extended manufacturing timelines, and significant environmental liabilities that undermine commercial viability in today's sustainability-focused market landscape.
The Novel Approach
The patented methodology overcomes these historical limitations through an elegantly designed iron-catalyzed cyclization process that operates under fundamentally different mechanistic principles. By utilizing ferric chloride or ferric nitrate as environmentally benign catalysts in combination with diiodo pentoxide as a selective oxidant, the reaction achieves complete transformation within a remarkably short timeframe of just 0.1 to 1 hour at moderate temperatures between 80°C and 120°C. The one-pot nature of this synthesis eliminates intermediate isolation steps that traditionally introduce yield losses and contamination risks, while the carefully optimized solvent system using common organic media like 1,4-dioxane ensures excellent solubility and reaction homogeneity. Crucially, the process demonstrates exceptional substrate tolerance across diverse functional groups including halogens, alkyl chains, and heterocyclic moieties without requiring protective group strategies, enabling direct access to structurally complex derivatives essential for advanced pharmaceutical applications. This innovative approach represents a paradigm shift from energy-intensive traditional methods toward sustainable manufacturing that aligns with evolving regulatory requirements and corporate environmental stewardship goals.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The catalytic cycle begins with the coordination of ferric chloride to the alkyne moiety of the o-alkynyl chalcone precursor, activating it toward nucleophilic attack by the adjacent enone system. This Lewis acid-mediated activation triggers an intramolecular cyclization where the carbonyl oxygen attacks the activated alkyne carbon, forming a vinyl cation intermediate that subsequently undergoes oxidative ring closure facilitated by diiodo pentoxide. The iron catalyst plays a dual role in both activating the alkyne functionality and stabilizing key cationic intermediates throughout the transformation sequence, ensuring high regioselectivity toward the desired cis-fused cyclopropane architecture. This mechanism operates through a concerted asynchronous pathway that minimizes competing side reactions while maintaining excellent stereochemical control over the newly formed chiral centers. The mild thermal conditions prevent undesired rearrangements or decomposition pathways that commonly plague traditional high-energy cyclization methods, resulting in superior product integrity and consistency across diverse substrate classes.
Impurity control is inherently built into this catalytic system through multiple self-regulating mechanisms that prevent common side product formation. The precise stoichiometric balance between catalyst loading (5-30 mol%) and oxidant concentration (100-400 mol%) creates optimal reaction kinetics that minimize overoxidation or polymerization byproducts typically observed in quinone syntheses. The solvent choice of polar aprotic media like acetonitrile or dioxane effectively solvates reactive intermediates while suppressing unwanted proton transfer pathways that could lead to decomposition products. Furthermore, the moderate temperature window prevents thermal degradation of sensitive functional groups present in complex substrates, maintaining high product purity without requiring additional purification steps beyond standard chromatography. This inherent selectivity profile ensures consistent production of pharmaceutical-grade intermediates meeting stringent regulatory requirements for impurity profiles.
How to Synthesize 1,4-Naphthoquinocyclopropane Compounds Efficiently
This patented methodology represents a significant operational advancement for pharmaceutical manufacturers seeking reliable production routes for complex quinone intermediates. The process eliminates multiple pain points associated with traditional synthesis approaches through its streamlined one-pot design and use of commercially available reagents under standard laboratory conditions. Detailed standardized synthesis steps are provided below to facilitate immediate implementation in industrial settings while maintaining full compliance with regulatory requirements for pharmaceutical intermediate production.
- Prepare reaction mixture by combining o-alkynyl chalcone compound with ferric chloride catalyst (20 mol%) and diiodo pentoxide oxidant (300 mol%) in anhydrous 1,4-dioxane solvent at 5 mL/mmol concentration.
- Heat the homogeneous solution to precisely 100°C under nitrogen atmosphere with vigorous magnetic stirring for exactly 0.5 hours to ensure complete cyclization.
- Purify the crude product through flash column chromatography using petroleum ether/ethyl acetate (20: 1 v/v) as eluent with 100-200 mesh silica gel to isolate high-purity target compounds.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial strategic advantages for procurement and supply chain decision-makers by addressing critical pain points in pharmaceutical intermediate sourcing. The process fundamentally reconfigures the cost structure and reliability profile of naphthoquinone cyclopropane production through its elimination of hazardous materials and simplification of manufacturing workflows. These improvements translate directly into enhanced supply chain resilience and competitive positioning for organizations committed to sustainable pharmaceutical manufacturing practices.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and hazardous oxidants significantly reduces raw material costs while avoiding costly waste treatment procedures required by traditional chromium-based processes. The simplified one-pot operation minimizes labor requirements and equipment utilization time compared to multi-step conventional syntheses, creating substantial operational savings without compromising product quality or yield consistency.
- Enhanced Supply Chain Reliability: Utilization of readily available starting materials with established global supply chains ensures consistent feedstock availability regardless of geopolitical disruptions. The process's compatibility with standard manufacturing equipment eliminates dependency on specialized reactors or rare instrumentation, enabling rapid technology transfer across multiple production sites worldwide while maintaining identical quality profiles.
- Scalability and Environmental Compliance: The methodology demonstrates exceptional scalability from laboratory to commercial production without requiring process reoptimization, with consistent performance maintained from gram-scale validation to multi-kilogram manufacturing runs. The environmentally benign profile eliminates regulatory hurdles associated with hazardous waste disposal while meeting increasingly stringent ESG requirements from both regulators and corporate stakeholders.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of patent CN109896944B's experimental data and implementation requirements. These insights have been compiled specifically for procurement teams evaluating this technology for integration into their pharmaceutical intermediate supply chains.
Q: How does this novel method address environmental concerns in traditional naphthoquinone synthesis?
A: The patented process eliminates hazardous chromium-based oxidants and high-energy consumption steps inherent in conventional methods. By utilizing iron-based catalysis under mild thermal conditions (80-120°C), it generates no toxic waste streams while maintaining high atom economy through a one-pot cyclization mechanism.
Q: What scalability advantages does this one-pot synthesis offer for commercial production?
A: The methodology demonstrates exceptional scalability due to its simple operational profile requiring only standard glassware and common solvents. The absence of cryogenic conditions or specialized equipment enables seamless transition from laboratory scale (mmol) to industrial production (multi-kilogram), with consistent yield maintenance across all scales as validated in patent examples.
Q: How does the mild reaction condition improve impurity profile compared to conventional methods?
A: Operating at moderate temperatures (80-120°C) prevents thermal degradation pathways that generate byproducts in traditional high-energy processes. The selective iron-catalyzed cyclization minimizes side reactions, producing cleaner reaction profiles that simplify purification and consistently achieve >95% purity without additional refining steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Naphthoquinocyclopropane Compounds Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical instrumentation. This patented methodology aligns perfectly with our core competencies in complex molecule synthesis, where we have successfully implemented similar iron-catalyzed processes for global pharmaceutical clients requiring high-purity intermediates with challenging stereochemical requirements. Our dedicated technical teams ensure seamless technology transfer from laboratory validation to full-scale manufacturing while maintaining complete regulatory compliance throughout all production phases.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis can optimize your specific supply chain requirements. Please contact us directly to obtain specific COA data and route feasibility assessments tailored to your production needs.
