Advanced Asymmetric Hydrogenation for Scalable Tetrahydro-13-Methyl Berberine Derivative Production
Introduction to Patent CN113980017A
The pharmaceutical industry continuously seeks robust synthetic routes for bioactive alkaloids, particularly those with potent anticancer and antimalarial properties. Patent CN113980017A, published on January 28, 2022, introduces a groundbreaking methodology for the synthesis of C-13 methyl-substituted tetrahydro-13-methyl berberine derivatives. This innovation addresses the critical bottlenecks of traditional extraction and multi-step chemical synthesis by employing a highly efficient asymmetric hydrogenation strategy. By utilizing 13-methyl berberine alkaloids as direct precursors, the process achieves yields greater than 90% with exceptional stereocontrol, marking a significant leap forward in the manufacturing of these valuable pharmaceutical intermediates. For R&D directors and procurement specialists, this technology represents a viable pathway to secure high-purity materials while drastically simplifying the supply chain.
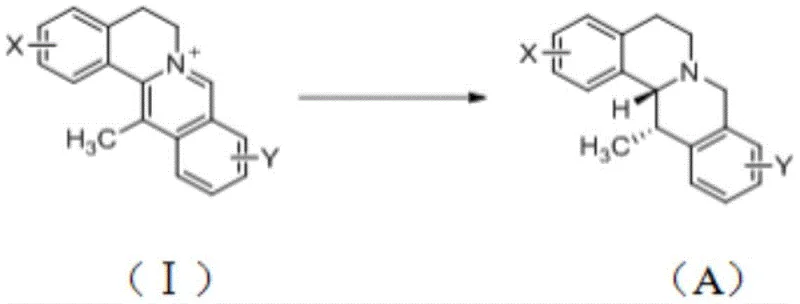
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of (13S,13aR)-5,8,13,13a-tetrahydro-6H-isoquinolino[3,2-a]isoquinoline derivatives has been fraught with inefficiencies. Traditional reliance on extraction from natural sources like Coptis chinensis Franch is inherently unstable, subject to seasonal variations, geographical limitations, and low content in plant tissues, failing to meet the surging global demand. Furthermore, prior chemical synthesis routes, such as the condensation of imines with o-carboxyphthalic anhydride followed by complex configuration conversions and reductions, involve excessive steps and harsh conditions. As illustrated in earlier literature methods, these pathways often require expensive transition metal catalysts in stoichiometric amounts and generate significant waste, leading to low overall yields and prohibitive production costs that hinder commercial viability.
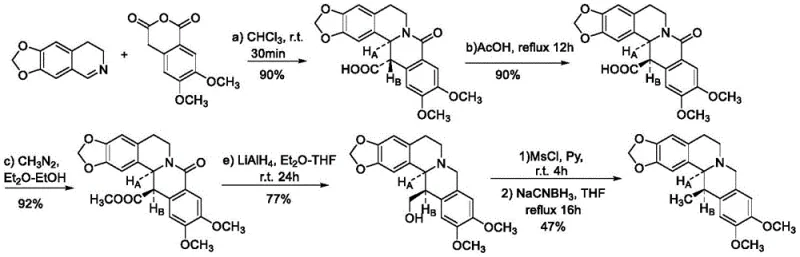
The Novel Approach
In stark contrast, the method disclosed in CN113980017A streamlines the production into a single, catalytic hydrogenation step. By directly reducing the C=N double bond of readily available 13-methyl berberine alkaloids, the process eliminates the need for laborious protection-deprotection sequences and intermediate isolations. This direct approach not only simplifies the operational workflow but also significantly enhances atom economy. The use of a chiral metal catalyst system allows for precise control over the stereocenter at the C-13 position, ensuring the formation of the biologically active (13S,13aR) isomer with high enantiomeric excess. This shift from multi-step linear synthesis to a convergent catalytic process fundamentally alters the cost structure, making large-scale manufacturing economically feasible for the first time.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Hydrogenation
The core of this technological breakthrough lies in the sophisticated interplay between the metal catalyst and the chiral ligand environment. The reaction typically employs an iridium precursor, such as [(1,5-cyclooctadiene)iridium(I) chloride] dimer, which complexes with bulky, electron-rich chiral phosphine ligands. The patent specifically identifies ligands like (S)-SEGPHOS, (R)-SYNPHOS, and various Josiphos derivatives as highly effective. These ligands create a chiral pocket around the metal center that directs the approach of the hydrogen molecule and the substrate, ensuring that hydride transfer occurs exclusively from one face of the planar iminium ion intermediate. This steric guidance is crucial for achieving the observed high enantioselectivity, often exceeding 90% ee, which is vital for pharmaceutical applications where isomeric purity dictates biological activity and safety profiles.
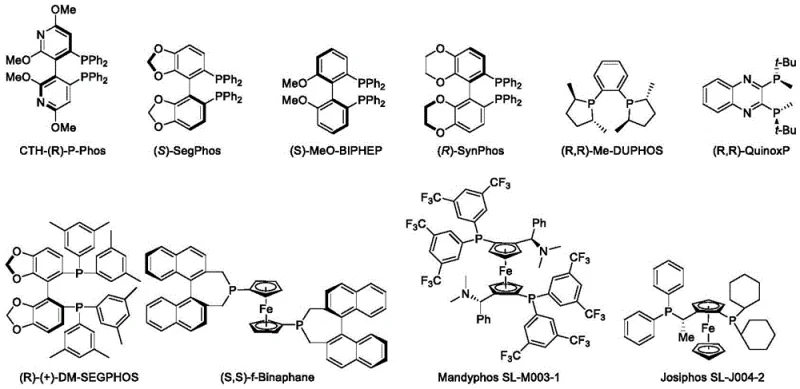
Beyond the catalyst system, the reaction environment plays a pivotal role in stabilizing the active catalytic species and facilitating proton transfer. The inclusion of inorganic salt additives, such as potassium bromide or lithium iodide, alongside acid additives like glacial acetic acid, serves to modulate the electronic properties of the catalyst and activate the substrate. The acid additive likely protonates the nitrogen atom of the berberine alkaloid, generating a more electrophilic iminium species that is more susceptible to nucleophilic attack by the metal-hydride complex. This synergistic effect between the metal-ligand complex and the additives allows the reaction to proceed under relatively mild temperatures (preferably 60°C) and pressures (preferably 8 MPa), balancing reaction rate with selectivity to minimize side reactions and impurity formation.
How to Synthesize Tetrahydro-13-Methyl Berberine Derivative Efficiently
The implementation of this synthesis route requires careful attention to catalyst activation and reaction parameters to maximize yield and purity. The process begins with the pre-complexation of the metal catalyst and chiral ligand in an anhydrous organic solvent under an inert atmosphere to prevent catalyst deactivation by oxygen or moisture. Once the active catalytic species is formed, the substrate and additives are introduced, and the system is pressurized with hydrogen. The reaction is then heated to facilitate the reduction, followed by a standard workup involving quenching with base and extraction. For detailed operational parameters and specific embodiment data, please refer to the standardized synthesis guide below.
- Complex a metal catalyst (e.g., Iridium dimer) with a chiral ligand (e.g., SEGPHOS) in an organic solvent under inert gas protection.
- Add the 13-methyl berberine alkaloid substrate along with inorganic salt additives (e.g., KBr) and acid additives (e.g., acetic acid).
- Conduct hydrogenation reduction under high pressure (preferably 8 MPa) and elevated temperature (preferably 60°C) to achieve >90% yield.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this hydrogenation technology offers transformative benefits regarding cost stability and supply security. By shifting away from agriculture-dependent extraction and complex multi-step syntheses, manufacturers can decouple production from seasonal harvest cycles and raw material volatility. The simplified one-step process reduces the number of unit operations, thereby lowering labor costs, energy consumption, and equipment occupancy time. Furthermore, the high yield (>90%) minimizes raw material waste, directly contributing to a lower cost of goods sold (COGS) and enabling more competitive pricing strategies in the global marketplace for these high-value intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric reagents and the reduction of synthetic steps significantly lower the overall production cost. By avoiding the need for multiple metal catalysts and excessive acid catalysis required in older methods, the process achieves substantial cost savings through improved material efficiency and reduced waste disposal expenses.
- Enhanced Supply Chain Reliability: Utilizing stable, commercially available 13-methyl berberine alkaloids as starting materials ensures a consistent feedstock supply. Unlike plant extraction which is vulnerable to climate change and crop failures, this chemical synthesis route provides a reliable and predictable production schedule, mitigating the risk of supply disruptions for downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The process is designed for scalability, utilizing standard high-pressure hydrogenation reactors common in fine chemical plants. The simplified workflow generates less chemical waste and avoids the use of hazardous reagents like diazomethane found in previous routes, aligning with modern green chemistry principles and easing regulatory compliance burdens for environmental health and safety teams.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on catalyst selection, reaction conditions, and product specifications for potential partners and licensees evaluating this technology for their production pipelines.
Q: What are the key advantages of this hydrogenation method over traditional extraction?
A: Unlike plant extraction which is limited by seasonal availability and low concentration, this synthetic method offers consistent supply, higher purity, and yields exceeding 90%, independent of agricultural factors.
Q: Which chiral ligands are most effective for this transformation?
A: The patent highlights several effective ligands including (S)-SEGPHOS, (R)-SYNPHOS, and Josiphos variants, with (S)-SEGPHOS being particularly preferred for achieving high enantiomeric excess (ee).
Q: Is this process scalable for industrial manufacturing?
A: Yes, the process utilizes standard high-pressure hydrogenation equipment and commercially available reagents, making it highly suitable for commercial scale-up from kilogram to tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydro-13-Methyl Berberine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this asymmetric hydrogenation technology to revolutionize the supply of anticancer and antimalarial intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory bench to full-scale manufacturing. Our state-of-the-art facilities are equipped with high-pressure hydrogenation capabilities and rigorous QC labs dedicated to maintaining stringent purity specifications, guaranteeing that every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this innovative synthesis route for your specific drug development programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this streamlined process can optimize your budget. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a sustainable and cost-effective supply chain for your critical alkaloid derivatives.
