Advanced Synthesis of 1-Carbazole-Boronic Acid Pinacol Ester via Amide-Directed Borylation for Commercial Scale-Up
Introduction to Next-Generation Carbazole Boronation Technology
The landscape of organic synthesis for high-value heterocyclic intermediates is undergoing a significant transformation, driven by the urgent need for sustainable and cost-effective manufacturing processes. Patent CN113831360A introduces a groundbreaking methodology for the synthesis of 1-carbazole-boronic acid pinacol ester, a critical building block widely utilized in the fabrication of organic light-emitting diodes (OLEDs) and pharmaceutical agents. This innovation addresses the longstanding limitations of conventional routes by employing a strategic amide ortho-oriented boronation technique. By leveraging the directing capability of an N-acyl group, the process achieves exceptional regioselectivity at the C1 position of the carbazole scaffold without relying on scarce transition metals. For global supply chain leaders and R&D directors, this represents a pivotal shift towards more robust and scalable chemical manufacturing, ensuring a steady supply of high-purity intermediates essential for next-generation electronic materials and therapeutic compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-carbazole-boronic acid derivatives has been plagued by significant technical and economic hurdles that impede large-scale production. Traditional approaches often rely on precious metal catalysis, such as iridium or ruthenium complexes, which not only inflate raw material costs but also introduce severe purification challenges due to the difficulty of removing trace metal residues to parts-per-million levels. Alternative routes utilizing 1-bromocarbazole as a starting material necessitate the use of hazardous organolithium reagents at cryogenic temperatures, posing substantial safety risks and energy burdens. Furthermore, palladium-catalyzed Miyaura borylation, while effective, suffers from the high cost of both the catalyst and the halogenated starting material. These legacy methods frequently result in moderate yields ranging from 34% to 81% and often require labor-intensive column chromatography for purification, generating excessive solvent waste and extending production lead times considerably.
The Novel Approach
In stark contrast to these established but inefficient protocols, the novel approach detailed in the patent utilizes a clever two-step sequence starting from inexpensive and readily available carbazole. The first step involves an N-acylation reaction that serves a dual purpose: it protects the reactive nitrogen center and installs a powerful directing group for the subsequent electrophilic substitution. This strategic modification enables highly selective boronation at the ortho-position using boron trihalides, completely bypassing the need for expensive iridium or palladium catalysts. The process operates under mild thermal conditions and utilizes common organic solvents, facilitating a straightforward workup procedure that relies on crystallization rather than chromatography. This paradigm shift not only drastically simplifies the operational workflow but also enhances the overall economic viability of producing this key intermediate for industrial applications.
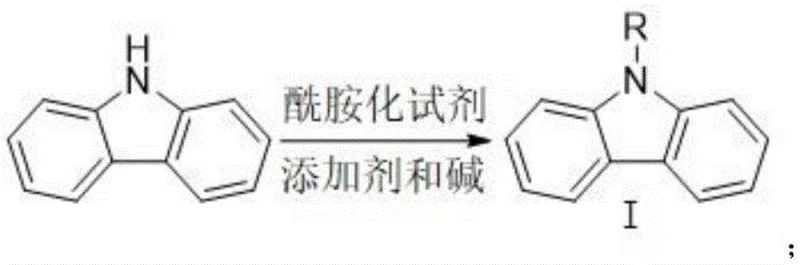
Mechanistic Insights into Amide Ortho-Oriented Boronation
The core mechanistic advantage of this synthesis lies in the electronic and steric influence of the N-acyl protecting group installed during the initial amidation phase. In unsubstituted carbazole, the N-H bond can interfere with catalytic cycles or lead to non-selective side reactions. By converting the amine into an amide, the nitrogen lone pair is delocalized into the carbonyl system, which modulates the electron density of the aromatic rings. More critically, the bulky acyl group acts as a proximal directing group that guides the electrophilic boron species specifically to the C1 position through a coordinated transition state. This intramolecular coordination ensures that the boronation occurs with high fidelity at the desired site, minimizing the formation of regioisomers that would otherwise complicate downstream processing. The use of boron trihalides, such as boron tribromide or boron trichloride, provides a potent electrophilic boron source that reacts efficiently with the activated aromatic system under controlled conditions.
Following the successful introduction of the boron moiety, the process seamlessly transitions into esterification and deprotection. The addition of pinacol in the presence of a base facilitates the formation of the stable pinacol boronate ester, which is the preferred form for storage and subsequent cross-coupling reactions. Simultaneously, the reaction conditions promote the removal of the acyl directing group, regenerating the free N-H carbazole structure in the final product. This telescoped or sequential strategy eliminates the need for isolating sensitive boronic acid intermediates, which are prone to protodeboronation or oxidation. The result is a streamlined pathway that maximizes atom economy and minimizes waste generation, aligning perfectly with the principles of green chemistry while delivering a product with purity exceeding 99% through simple recrystallization techniques.
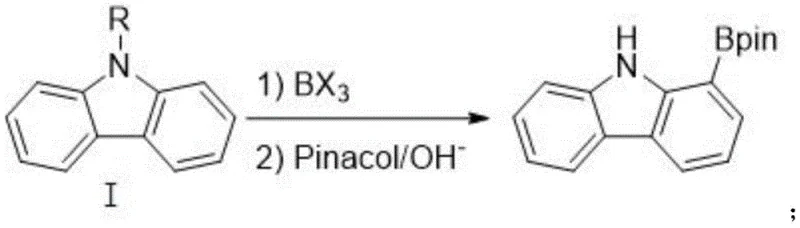
How to Synthesize 1-Carbazole-Boronic Acid Pinacol Ester Efficiently
The practical execution of this synthesis is designed for ease of operation in standard chemical manufacturing facilities, requiring no specialized high-pressure or cryogenic equipment. The protocol begins with the activation of carbazole using an amidation reagent like pivaloyl chloride in the presence of a nucleophilic catalyst such as DMAP and a tertiary amine base. Once the N-acyl intermediate is secured, it is subjected to boronation using a boron trihalide solution, followed by quenching and esterification with pinacol. The entire sequence is optimized to proceed at temperatures ranging from -10°C to ambient conditions, ensuring safety and reproducibility. Detailed standardized synthetic steps see the guide below.
- Perform amidation reaction on carbazole with an amidation reagent (e.g., pivaloyl chloride) using DMAP catalyst and base to obtain N-acylcarbazole intermediate.
- React the N-acylcarbazole intermediate with boron trihalide (BX3) in an organic solvent to effect ortho-boronation.
- Add pinacol and base to the reaction mixture to perform esterification and deprotection, yielding the final 1-carbazole-boronic acid pinacol ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers profound strategic benefits that extend far beyond simple laboratory curiosity. The elimination of noble metal catalysts such as iridium and palladium removes a major source of cost volatility and supply risk, as the prices of these metals are subject to intense geopolitical and market fluctuations. Furthermore, the reliance on commodity chemicals like carbazole and pivaloyl chloride ensures a stable and diversified supply base, reducing the likelihood of production stoppages due to raw material shortages. The simplified purification process, which eschews column chromatography in favor of crystallization, significantly reduces solvent consumption and waste disposal costs, contributing to a lower overall cost of goods sold (COGS) and a smaller environmental footprint. These factors collectively enhance the resilience and profitability of the supply chain for downstream manufacturers.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the complete avoidance of expensive transition metal catalysts and ligands, which traditionally account for a significant portion of raw material expenses in fine chemical synthesis. By substituting these with affordable organic reagents and Lewis acids, the direct material costs are substantially lowered. Additionally, the high selectivity of the reaction minimizes the formation of byproducts, thereby improving the effective yield and reducing the loss of valuable starting materials. The ability to purify the final product through crystallization rather than chromatography further drives down operational expenditures by saving on silica gel, solvents, and labor hours associated with complex purification workflows.
- Enhanced Supply Chain Reliability: Dependence on specialized catalysts often creates single points of failure in the supply chain, where delays from a single vendor can halt entire production lines. This new method mitigates such risks by utilizing widely available reagents that can be sourced from multiple global suppliers. The robustness of the reaction conditions, which do not require extreme temperatures or pressures, also means that the process can be easily transferred between different manufacturing sites without extensive re-validation. This flexibility ensures consistent delivery schedules and allows for rapid scaling of production volumes to meet fluctuating market demands without compromising on quality or lead times.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process is inherently safer and cleaner than its predecessors. The absence of pyrophoric reagents like n-butyllithium eliminates the need for specialized handling equipment and reduces the risk of fire or explosion incidents. Moreover, the reduction in solvent usage and the elimination of heavy metal waste streams simplify compliance with increasingly stringent environmental regulations regarding wastewater treatment and hazardous waste disposal. This makes the technology particularly attractive for large-scale commercial production, where sustainability metrics are becoming critical criteria for vendor selection and long-term partnership agreements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific advantages and operational details outlined in the patent documentation to provide clarity for potential partners. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: Why is the amide-directed borylation method superior to traditional iridium-catalyzed C-H activation?
A: Traditional iridium-catalyzed methods often suffer from low yields (e.g., 34-47%) and require expensive noble metal catalysts that are difficult to remove to ppm levels required for electronics. The amide-directed method uses cheap carbazole, avoids noble metals entirely, and achieves higher selectivity and yields up to 89.6% with simpler purification.
Q: Does this synthesis route require cryogenic conditions like lithiation methods?
A: No. Unlike lithiation routes that require n-BuLi at ultralow temperatures (e.g., -78°C), this patented process operates under mild conditions, typically between -10°C and room temperature. This significantly reduces energy consumption and safety risks associated with handling pyrophoric reagents on a large scale.
Q: How is product purity managed without column chromatography?
A: The high regioselectivity of the amide directing group ensures that the boronation occurs almost exclusively at the C1 position. This intrinsic selectivity allows for purification via simple washing, concentration, and crystallization steps, avoiding the need for costly and solvent-intensive column chromatography while achieving purity levels greater than 99%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Carbazole-Boronic Acid Pinacol Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final products, whether they are life-saving medicines or cutting-edge display technologies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 1-carbazole-boronic acid pinacol ester meets the highest industry standards. Our expertise in implementing advanced synthetic routes allows us to offer competitive pricing without compromising on the quality or reliability that your operations depend upon.
We invite you to collaborate with us to leverage this innovative synthesis technology for your specific applications. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your current supply chain structure. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that demonstrate how our optimized manufacturing processes can drive value and efficiency for your organization. Let us be your trusted partner in navigating the complexities of fine chemical sourcing and development.
