Advanced Grignard-Based Synthesis of Enilconazole for Industrial Scale-Up
The global demand for high-efficiency, broad-spectrum fungicides continues to drive innovation in agrochemical synthesis, particularly for critical active ingredients like Enilconazole (Imazalil). A recent technological breakthrough detailed in patent CN114409600A introduces a streamlined, two-step synthetic methodology that fundamentally reshapes the production landscape for this vital compound. Unlike legacy processes that suffer from excessive step counts and hazardous reagent usage, this novel approach leverages a robust Grignard reaction followed by a precise nucleophilic substitution to achieve exceptional results. The patent data indicates that this method consistently delivers yields exceeding 80% and product purity surpassing 98%, addressing the dual imperatives of economic efficiency and stringent quality control required by modern pharmaceutical and agrochemical standards. For industry stakeholders, this represents a pivotal shift towards more sustainable and reliable manufacturing paradigms.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Enilconazole has been plagued by convoluted reaction pathways that introduce significant operational risks and cost inefficiencies. As illustrated in prior art such as World Patent WO2000038521, traditional routes often commence with 2,4-dichloroacetophenone, necessitating a sequential series of bromination, amination, reduction, and etherification steps. 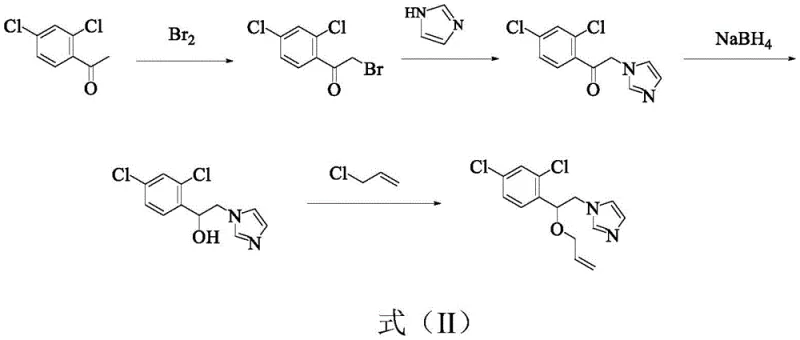 This multi-stage progression not only extends the production timeline but also relies on dangerous reagents like elemental bromine and sodium borohydride, which pose severe safety hazards and complicate waste management protocols. Furthermore, alternative methods involving cyclic ether intermediates or chloroketone reductions often struggle with suboptimal yields and difficult purification processes, creating bottlenecks that hinder the ability of a reliable agrochemical intermediate supplier to meet fluctuating market demands without inflating costs.
This multi-stage progression not only extends the production timeline but also relies on dangerous reagents like elemental bromine and sodium borohydride, which pose severe safety hazards and complicate waste management protocols. Furthermore, alternative methods involving cyclic ether intermediates or chloroketone reductions often struggle with suboptimal yields and difficult purification processes, creating bottlenecks that hinder the ability of a reliable agrochemical intermediate supplier to meet fluctuating market demands without inflating costs.
The Novel Approach
In stark contrast, the methodology disclosed in CN114409600A simplifies the entire value chain by condensing the synthesis into two highly efficient stages. The core innovation lies in the direct construction of the carbon skeleton via a Grignard addition, bypassing the need for pre-functionalized ketones or complex etherifications found in older patents. 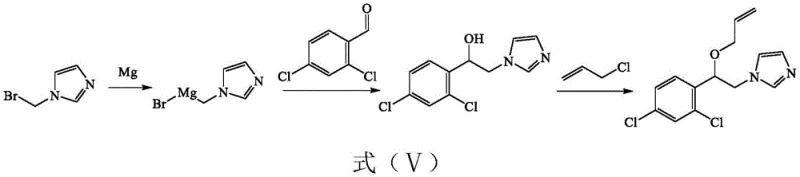 This streamlined pathway utilizes 2-(bromomethyl)imidazole and 2,4-dichlorobenzaldehyde as primary building blocks, reacting them under controlled conditions to form the key alcohol intermediate, which is subsequently etherified with allyl chloride. By eliminating unnecessary transformation steps and hazardous oxidizing or reducing agents, this novel approach drastically reduces the environmental footprint and operational complexity, offering a clear pathway for cost reduction in fungicide manufacturing while simultaneously enhancing the safety profile of the production facility.
This streamlined pathway utilizes 2-(bromomethyl)imidazole and 2,4-dichlorobenzaldehyde as primary building blocks, reacting them under controlled conditions to form the key alcohol intermediate, which is subsequently etherified with allyl chloride. By eliminating unnecessary transformation steps and hazardous oxidizing or reducing agents, this novel approach drastically reduces the environmental footprint and operational complexity, offering a clear pathway for cost reduction in fungicide manufacturing while simultaneously enhancing the safety profile of the production facility.
Mechanistic Insights into Grignard Addition and Nucleophilic Substitution
The chemical elegance of this new process is rooted in the precise execution of the Grignard reaction, which serves as the foundation for constructing the chiral center and the core molecular architecture of Enilconazole. The mechanism initiates with the formation of ((1H-imidazol-1-yl)methyl)magnesium bromide, generated by reacting 2-(bromomethyl)imidazole with magnesium turnings in anhydrous tetrahydrofuran (THF) at elevated temperatures between 60°C and 70°C. This organometallic species is then carefully introduced to a solution of 2,4-dichlorobenzaldehyde maintained at cryogenic temperatures ranging from -10°C to 0°C. This strict thermal control is critical to suppress side reactions and ensure the nucleophilic attack occurs selectively at the carbonyl carbon, yielding the intermediate 2-(2,4-dichlorophenyl)-1H-imidazole-1-ethanol with high fidelity. The subsequent quenching and extraction phases are optimized to recover the crude alcohol with minimal loss, setting the stage for the final transformation.
Following the successful formation of the alcohol intermediate, the process transitions to a nucleophilic substitution reaction to install the allyl ether moiety, which is essential for the biological activity of the final fungicide. In this step, the hydroxyl group of the intermediate acts as a nucleophile, attacking allyl chloride in the presence of an acid-binding agent such as triethylamine, DIPEA, or N,N-dimethylacetamide. The reaction is conducted at moderate temperatures of 60°C to 80°C, facilitating the displacement of the chloride ion and the formation of the stable ether bond. Crucially, the patent highlights that this specific sequence minimizes the formation of regioisomers or over-alkylated byproducts, which are common impurities in less optimized syntheses. The resulting crude product is then subjected to a straightforward recrystallization process, typically using ethanol or acetone, which effectively removes trace impurities and delivers the final high-purity Enilconazole with a melting point consistent with pharmacopeial standards.
How to Synthesize Enilconazole Efficiently
Implementing this advanced synthetic route requires adherence to strict operational parameters to maximize yield and safety, particularly regarding moisture control and temperature regulation during the Grignard formation. The patent outlines a scalable protocol that begins with the activation of magnesium and the careful generation of the organomagnesium reagent under an inert nitrogen atmosphere to prevent premature decomposition. Following the addition of the aldehyde and the subsequent workup, the intermediate alcohol is isolated and immediately subjected to the etherification step without the need for extensive intermediate purification, thereby saving time and solvent resources.
- Prepare ((1H-imidazol-1-yl)methyl)magnesium bromide by reacting 2-(bromomethyl)imidazole with magnesium in THF at 60-70°C under nitrogen protection.
- Add 2,4-dichlorobenzaldehyde to the Grignard reagent at -10 to 0°C, then react at 0-10°C to form 2-(2,4-dichlorophenyl)-1H-imidazole-1-ethanol.
- Perform nucleophilic substitution by reacting the intermediate with allyl chloride and an acid binding agent (e.g., triethylamine) at 60-80°C, followed by recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Grignard-based synthesis offers tangible strategic benefits that extend far beyond simple chemical yield improvements. The primary advantage lies in the substantial simplification of the raw material portfolio; by shifting away from complex, multi-step precursors to commodity chemicals like 2,4-dichlorobenzaldehyde and allyl chloride, manufacturers can significantly mitigate supply risk and volatility. This transition ensures a more resilient supply chain, as the sourcing of these fundamental building blocks is less susceptible to the bottlenecks that often plague specialized intermediate markets. Furthermore, the elimination of hazardous reagents such as elemental bromine and sodium borohydride reduces the regulatory burden and insurance costs associated with handling dangerous goods, contributing to a leaner and more compliant operational model.
- Cost Reduction in Manufacturing: The streamlined two-step process inherently lowers production costs by reducing the total number of unit operations, which directly translates to decreased labor, energy, and solvent consumption. By avoiding the use of expensive and hazardous reducing agents like sodium borohydride, the process eliminates the need for specialized waste treatment protocols required for boron-containing effluents. Additionally, the high yield reported in the patent means that less raw material is wasted per kilogram of finished product, optimizing the overall material balance and driving down the cost of goods sold (COGS) without compromising on quality specifications.
- Enhanced Supply Chain Reliability: The reliance on widely available and commercially mature starting materials ensures that production schedules are not held hostage by the availability of niche reagents. This stability is crucial for maintaining continuous operation in large-scale facilities, where any interruption in the supply of a single specialized precursor can halt the entire production line. The robustness of the Grignard reaction conditions also allows for greater flexibility in batch sizing, enabling manufacturers to respond more agilely to sudden spikes in demand for agrochemical products during peak planting seasons.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns perfectly with green chemistry principles by minimizing waste generation and avoiding the use of heavy metals or toxic halogens in the final steps. The simplicity of the purification process, which relies on standard recrystallization rather than complex chromatography, makes the technology highly scalable from pilot plant trials to multi-ton commercial production. This ease of scale-up ensures that the commercial scale-up of complex agrochemical intermediates can be achieved rapidly, allowing companies to bring products to market faster while meeting increasingly stringent environmental regulations regarding industrial emissions and waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the experimental data and claims presented in the patent documentation. These insights are intended to clarify the operational feasibility and quality outcomes associated with this new manufacturing route for decision-makers evaluating potential technology transfers or procurement partnerships.
Q: What are the key advantages of the Grignard-based synthesis route for Enilconazole?
A: The Grignard-based route described in patent CN114409600A offers a significantly shorter reaction sequence compared to traditional methods, eliminating hazardous reagents like bromine and sodium borohydride. It achieves yields exceeding 80% and purity greater than 98%, ensuring high operational safety and cost efficiency for large-scale production.
Q: How does this new method improve supply chain reliability for fungicide manufacturers?
A: By utilizing readily available and inexpensive starting materials such as 2-(bromomethyl)imidazole and 2,4-dichlorobenzaldehyde, the process reduces dependency on complex or scarce intermediates. The simplified two-step workflow minimizes processing time and equipment requirements, enhancing overall supply continuity.
Q: Is this synthesis method suitable for commercial scale-up?
A: Yes, the patent explicitly states the method is designed for large-scale industrial application. The mild reaction conditions, high yield, and straightforward purification via recrystallization make it highly scalable from pilot batches to multi-ton commercial production without compromising quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Enilconazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to more efficient synthetic routes is critical for maintaining competitiveness in the global agrochemical market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN114409600A are fully realized in practical, industrial settings. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Enilconazole meets the highest international standards for efficacy and safety. We are committed to leveraging our technical expertise to optimize this Grignard-based process, delivering a product that combines superior quality with unmatched supply reliability.
We invite forward-thinking partners to collaborate with us to explore the full potential of this advanced synthesis technology. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, allowing you to make informed decisions that will strengthen your supply chain and enhance your product portfolio with high-quality, cost-effective fungicide solutions.
