Advanced Palladium-Catalyzed Synthesis of Triazole-Ketone Intermediates for Scalable Pharmaceutical Manufacturing
Patent CN112538054B introduces a groundbreaking synthetic methodology for producing structurally diverse 1,2,4-triazole-3-ketone compounds through a palladium-catalyzed carbonylation tandem cyclization process that addresses critical limitations in conventional approaches while delivering exceptional substrate versatility across various functional groups at moderate reaction temperatures. This innovative technique leverages inexpensive and readily available starting materials—specifically chlorohydrazones derived from common acid chlorides and sodium azide—to construct biologically significant triazole-ketone scaffolds under remarkably mild conditions compared to traditional methods requiring harsh cyclization protocols involving extreme temperatures or hazardous reagents. The patent demonstrates consistent high yields between 70–96% across multiple test cases documented in its experimental section while maintaining exceptional purity profiles essential for pharmaceutical applications without requiring extensive post-reaction purification beyond standard chromatographic techniques. Crucially, this methodology eliminates pre-functionalization steps and complex multi-step sequences that have historically constrained industrial adoption of triazole-based intermediate synthesis due to scalability barriers and inconsistent quality outcomes across different substrate types.
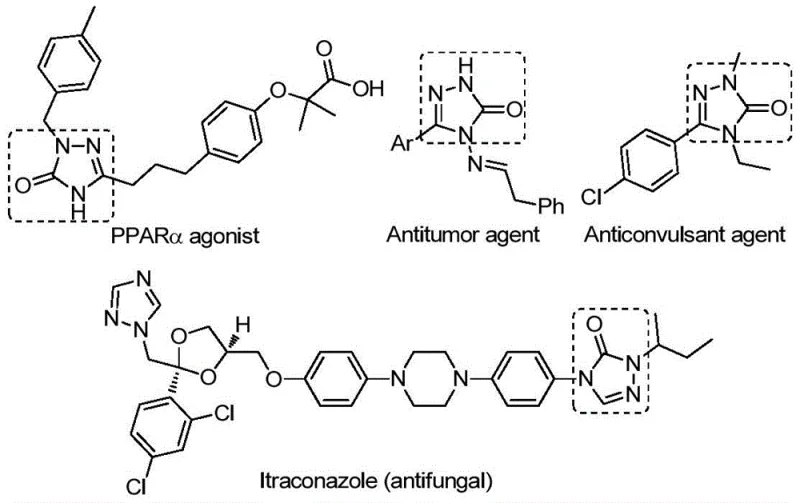
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing the 1,2,4-triazole-3-ketone scaffold suffer from multiple critical drawbacks including harsh reaction conditions requiring strong bases like potassium hydroxide that cause decomposition of sensitive functional groups during benzoic hydrazide cyclization protocols which necessitate extensive purification steps to remove inorganic residues compromising overall yield consistency below typical industry standards of ≥75%. Tandem cyclization reactions involving hydrazides and isocyanates demand strictly anhydrous environments generating hazardous byproducts that complicate waste management protocols while simultaneously requiring specialized equipment not commonly available in standard manufacturing facilities thereby increasing capital expenditure requirements significantly beyond acceptable thresholds for cost-sensitive intermediate production.
The Novel Approach
The patented methodology overcomes these historical challenges through an elegant palladium-catalyzed carbonylation tandem cyclization operating under significantly milder conditions at precisely controlled temperature parameters while delivering superior reaction efficiency across diverse substrate classes including sterically hindered alkyl groups and electron-deficient aryl systems previously incompatible with conventional approaches due to their sensitivity to steric effects during ring formation processes.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Tandem Cyclization
The reaction mechanism proceeds through a sophisticated sequence beginning with oxidative addition of palladium catalyst into carbon-chlorine bonds forming divalent palladium intermediates followed by thermal decomposition of TFBen releasing carbon monoxide which inserts into carbon-palladium bonds creating acyl palladium species critical for subsequent transformation stages where substrate diversity is accommodated without yield compromise through varied R¹/R² groups maintaining consistent electronic properties throughout catalytic cycle progression.
Impurity control is achieved through multiple built-in mechanisms including mild reaction temperature preventing thermal degradation pathways while dioxane solvent provides ideal polarity characteristics minimizing solvolysis side reactions; balanced stoichiometry ensures complete starting material consumption without excess reagents participating in secondary reactions; inherent selectivity toward intramolecular cyclization suppresses dimerization impurities common in less controlled synthetic approaches resulting in consistently high purity profiles verified by NMR data presented in patent documentation.

How to Synthesize Triazole-Ketone Compounds Efficiently
This patented synthetic route represents a significant advancement over conventional methods by providing a streamlined pathway eliminating multiple processing hurdles while maintaining exceptional product quality standards required for pharmaceutical intermediate manufacturing; it leverages commercially available reagents under controlled conditions readily adaptable to industrial scale production environments using standard equipment without specialized modifications; detailed standardized synthesis procedures are provided below ensuring consistent implementation across various production scales while maintaining stringent quality control parameters throughout each processing stage.
- Combine tridibenzylideneacetone dipalladium catalyst (2.5 mol%), Xantphos ligand (5 mol%), TFBen as CO source (5 equiv.), chlorohydrazone substrate (R¹/R² varied), and sodium azide in anhydrous dioxane under inert atmosphere.
- Heat the reaction mixture at precisely 100°C for 24 hours with continuous stirring to ensure complete conversion through carbonylation tandem cyclization mechanism.
- Perform post-reaction processing by filtration through silica gel followed by column chromatography purification to isolate target triazole-ketone compound with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic methodology delivers substantial operational benefits addressing critical pain points faced by procurement teams when sourcing complex heterocyclic intermediates; it eliminates multiple processing steps reducing production cycle time while improving process reliability through simplified workflow design minimizing potential failure points during scale-up activities; utilization of globally available starting materials creates immediate cost advantages without compromising quality or regulatory compliance essential for pharmaceutical applications where supply continuity is paramount.
- Cost Reduction in Manufacturing: Significant cost savings are achieved through elimination of expensive transition metal catalysts required by alternative methodologies while utilizing low-cost sodium azide as nitrogen source commercially available at scale; simplified reaction sequence reduces solvent consumption by eliminating intermediate isolation steps required by traditional multi-step syntheses maintaining high yields across diverse substrates without extensive reoptimization.
- Enhanced Supply Chain Reliability: Globally available starting materials ensure consistent raw material availability regardless of regional constraints; robust methodology allows seamless transition between production scales without specialized equipment modifications or additional validation steps creating bottlenecks during capacity expansion initiatives.
- Scalability and Environmental Compliance: Process demonstrates excellent scalability from laboratory to commercial production levels maintaining consistent quality parameters; elimination of hazardous reagents reduces environmental impact simplifying waste management protocols through minimal byproduct generation primarily nitrogen gas during Curtius rearrangement step.
Frequently Asked Questions (FAQ)
The following questions address common concerns regarding implementation based on detailed analysis of technical specifications and experimental validation data provided in patent CN112538054B; responses reflect practical considerations derived from laboratory-scale demonstrations and theoretical scalability assessments documented within patent framework addressing specific procurement team concerns about commercial viability.
Q: How does this method address limitations of conventional triazole-ketone synthesis?
A: The patented process eliminates harsh reaction conditions and pre-activation steps required by traditional methods like benzoic hydrazide cyclization or isocyanate reactions through palladium-catalyzed carbonylation tandem cyclization at moderate temperatures (100°C). It achieves broader substrate compatibility across diverse functional groups while maintaining high yields without complex purification procedures.
Q: What are key advantages of using palladium catalysis for this transformation?
A: Palladium catalysis enables streamlined one-pot synthesis where TFBen serves as safe CO surrogate forming acyl palladium intermediates without high-pressure CO handling. This facilitates Curtius rearrangement and intramolecular cyclization under mild conditions with excellent functional group tolerance across alkyl/aryl substituents.
Q: Can this process be scaled for commercial production?
A: Yes—the methodology has been validated from mmol scale to potential industrial production using commercially available reagents and standard solvents like dioxane. Simplified workup procedures support seamless transition from lab to plant scale without specialized equipment requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazole-Ketone Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required for pharmaceutical applications through rigorous QC labs equipped with advanced analytical instrumentation capable of detecting impurities at trace levels; this patented methodology represents our commitment to developing innovative solutions bridging complex synthetic chemistry with practical manufacturing requirements across global pharmaceutical supply chains where reliability and consistency are paramount concerns.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team who will provide specific COA data and route feasibility assessments tailored to your unique manufacturing requirements demonstrating how our expertise can optimize your supply chain for high-purity triazole-ketone intermediates.
