Advanced One-Step Synthesis of Trisubstituted Pyridazine Derivatives for Commercial Scale-Up
Introduction to Next-Generation Pyridazine Synthesis
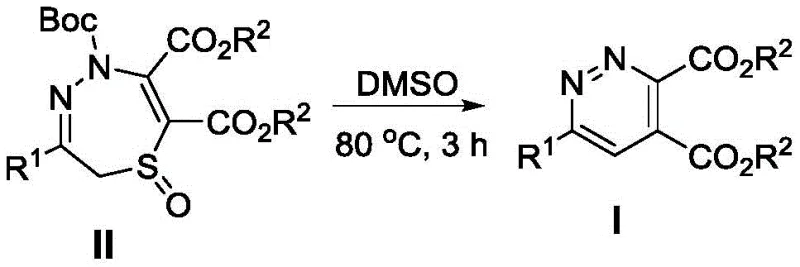
The landscape of heterocyclic chemistry is constantly evolving, driven by the relentless demand for more efficient and sustainable pathways to access bioactive scaffolds. Patent CN110240568B introduces a groundbreaking methodology for the preparation of trisubstituted pyridazine derivatives, a class of compounds that serves as a critical backbone in modern medicinal chemistry and agrochemical development. This innovation addresses long-standing challenges in the field by offering a concise, one-step synthetic route that bypasses the need for complex catalytic systems. The core of this technology lies in the utilization of 2,5-dihydro-1,4,5-thiadiazepine 1-oxides as versatile precursors, which undergo a remarkable thermal transformation to yield the desired pyridazine core with high structural fidelity.
For R&D directors and process chemists, the significance of this patent cannot be overstated, as it provides a robust alternative to traditional multi-step syntheses that often suffer from low atom economy and harsh reaction conditions. By leveraging dimethyl sulfoxide (DMSO) not merely as a solvent but as a key participant in the reaction medium, the method achieves excellent conversion rates under relatively mild thermal conditions. This technical advancement positions the technology as a vital tool for the reliable pharmaceutical intermediate supplier seeking to optimize their portfolio of heterocyclic building blocks. The ability to generate diverse substitution patterns on the pyridazine ring through simple modification of the starting material further enhances the utility of this approach for lead optimization campaigns.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the pyridazine nucleus has been a formidable challenge for organic chemists, often necessitating the use of hazardous hydrazine derivatives or relying on transition metal-catalyzed cyclizations that introduce significant purification burdens. Conventional routes frequently involve multiple synthetic steps, each carrying the risk of yield erosion and the accumulation of difficult-to-remove impurities. Furthermore, many established protocols require stringent exclusion of moisture and oxygen, demanding specialized equipment such as gloveboxes or Schlenk lines, which drastically increases the operational complexity and capital expenditure for manufacturing facilities. The reliance on precious metal catalysts also raises concerns regarding residual metal levels in the final active pharmaceutical ingredient (API), necessitating costly downstream purification processes to meet regulatory standards.
The Novel Approach
In stark contrast, the methodology disclosed in the patent data presents a paradigm shift towards simplicity and efficiency. The novel approach utilizes a thermodynamic ring contraction strategy where the precursor compound II is simply dissolved in DMSO and heated to 80°C for 3 hours. This single-step transformation elegantly constructs the aromatic pyridazine system while extruding the sulfur and nitrogen oxide components in a controlled manner. As illustrated in the reaction scheme, this process eliminates the need for any external reagents other than the solvent and heat source, representing a significant reduction in chemical waste and raw material costs. The operational simplicity is further highlighted by the fact that the reaction proceeds smoothly without the need for inert gas protection, making it exceptionally attractive for cost reduction in API manufacturing where safety and throughput are paramount concerns.
Mechanistic Insights into Thermal Ring Contraction
The mechanistic pathway underlying this transformation is a fascinating example of thermodynamic control driving the formation of a stable aromatic system. The reaction initiates with the solvation of the 2,5-dihydro-1,4,5-thiadiazepine 1-oxide precursor in the polar aprotic solvent DMSO, which stabilizes the transition states involved in the bond reorganization. Upon heating to 80°C, the molecule undergoes a concerted rearrangement that facilitates the cleavage of the sulfur-nitrogen bonds and the simultaneous formation of the new carbon-nitrogen double bond required for aromatization. This driving force is rooted in the immense stability gained by forming the fully conjugated pyridazine ring, which acts as a thermodynamic sink pulling the equilibrium towards the product side. The absence of radical initiators or metal catalysts suggests a pericyclic or ionic mechanism that is inherently cleaner and less prone to generating complex byproduct profiles often seen in radical-mediated processes.
From an impurity control perspective, this metal-free mechanism offers distinct advantages for the production of high-purity pyridazine derivatives. Without the introduction of transition metals, the risk of metal-catalyzed side reactions such as homocoupling or over-oxidation is virtually eliminated. The primary byproducts of this reaction are likely small, volatile molecules or polar species that can be easily separated during the aqueous workup and subsequent silica gel chromatography. The patent data indicates that a wide range of substituents, including electron-donating groups like methoxy and electron-withdrawing groups like trifluoromethyl, are well-tolerated under these conditions. This broad substrate scope confirms the robustness of the mechanistic pathway, ensuring that the electronic nature of the R1 and R2 groups does not significantly impede the ring contraction process, thereby providing a reliable platform for synthesizing diverse chemical libraries.
How to Synthesize Trisubstituted Pyridazine Derivatives Efficiently
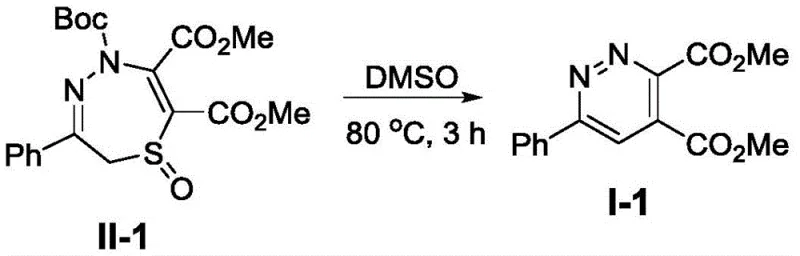
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific parameters to ensure optimal yield and reproducibility. The protocol dictates dissolving the starting material, such as Compound II-1, in DMSO to a precise concentration of 0.1M, which balances reaction kinetics with heat transfer efficiency. The mixture is then subjected to thermal treatment at 80°C for a duration of 3 hours, a timeframe that has been empirically determined to maximize conversion while minimizing potential degradation. Following the reaction, the workup procedure is remarkably straightforward, involving a simple aqueous quench followed by extraction with dichloromethane. The detailed standardized synthesis steps below outline the exact quantities and procedural nuances required to replicate the high yields reported in the patent examples, serving as a foundational guide for process development teams.
- Dissolve the precursor compound II (2,5-dihydro-1,4,5-thiadiazepine 1-oxide derivative) in dimethyl sulfoxide (DMSO) to achieve a concentration of approximately 0.1M.
- Heat the reaction mixture to 80°C and maintain this temperature for 3 hours to facilitate the thermal ring contraction and aromatization without requiring inert gas protection.
- Upon completion, cool to room temperature, extract with dichloromethane after water addition, dry the organic phase, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates directly into tangible operational benefits and risk mitigation. The elimination of expensive transition metal catalysts removes a significant variable from the raw material cost structure, while also simplifying the supply chain by reducing dependency on specialty chemical vendors who provide these sensitive reagents. Furthermore, the robustness of the reaction conditions means that the process is less susceptible to batch failures caused by minor fluctuations in atmospheric conditions, thereby enhancing overall supply chain reliability. The simplified workup procedure, which avoids complex distillation or crystallization steps often required to remove metal residues, significantly reduces the processing time and energy consumption per kilogram of product. These factors collectively contribute to a more resilient and cost-effective manufacturing model that can better withstand market volatility.
- Cost Reduction in Manufacturing: The most immediate financial impact of this technology stems from the complete removal of transition metal catalysts from the synthetic sequence. In traditional pharmaceutical manufacturing, the cost of palladium or copper catalysts, coupled with the expensive scavengers required to remove them to ppm levels, constitutes a substantial portion of the COGS. By utilizing a thermal, metal-free pathway, manufacturers can bypass these costs entirely. Additionally, the use of DMSO, a commodity solvent with a low price point, further drives down the raw material expenses. The high yields reported across various substrates mean that less starting material is wasted, improving the overall mass balance and reducing the cost of goods sold significantly without compromising on quality.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the scarcity of specialized reagents or the logistical challenges of transporting air-sensitive materials. This novel method mitigates these risks by relying on stable, shelf-stable starting materials and common solvents that are readily available from multiple global suppliers. The fact that the reaction does not require inert gas protection simplifies the logistics of raw material handling and storage, as there is no need for specialized cylinders or gas delivery infrastructure. This ease of sourcing ensures that production schedules can be maintained consistently, reducing the lead time for high-purity pharmaceutical intermediates and allowing for more agile responses to market demand fluctuations.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often reveals hidden complexities, but this thermal ring contraction is inherently scalable due to its simplicity. The absence of exothermic catalyst activation steps reduces the risk of thermal runaway, a critical safety consideration in large-scale reactors. Moreover, the environmental profile of this process is superior to conventional methods; it generates less hazardous waste since there are no heavy metal contaminants to dispose of. The solvent system is manageable, and the overall E-factor of the process is improved by the high atom economy of the one-step transformation. This alignment with green chemistry principles facilitates easier regulatory approval and supports corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pyridazine synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this route into existing manufacturing pipelines or for initiating new development projects focused on pyridazine-based therapeutics.
Q: Does this synthesis method require transition metal catalysts?
A: No, the patented process described in CN110240568B is entirely metal-free. It relies on thermal activation in DMSO, which eliminates the risk of heavy metal contamination often associated with traditional cross-coupling or cyclization methods.
Q: What are the typical reaction conditions for this transformation?
A: The reaction operates under mild thermodynamic conditions, specifically heating the substrate in DMSO at 80°C for 3 hours. Notably, the process does not require stringent inert atmosphere protection, simplifying operational requirements.
Q: Is this method suitable for large-scale manufacturing of pyridazine intermediates?
A: Yes, the simplicity of the workup procedure (aqueous quench and extraction) combined with the absence of expensive catalysts and inert gas requirements makes this route highly amenable to commercial scale-up and cost-effective production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trisubstituted Pyridazine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of innovative synthetic methodologies like the one described in CN110240568B for accelerating drug discovery and development. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such promising laboratory protocols into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of trisubstituted pyridazine derivatives meets the highest industry standards, providing you with a secure foundation for your downstream applications.
We invite you to collaborate with us to leverage this advanced chemistry for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with us, you gain access to our deep process knowledge and supply chain network, allowing you to focus on your core competencies while we handle the complexities of chemical manufacturing. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how we can drive value and efficiency into your supply chain.
