Advanced Metal-Free Synthesis Platform for High-Purity Trifluoromethyl Triazine Pharmaceutical Intermediates
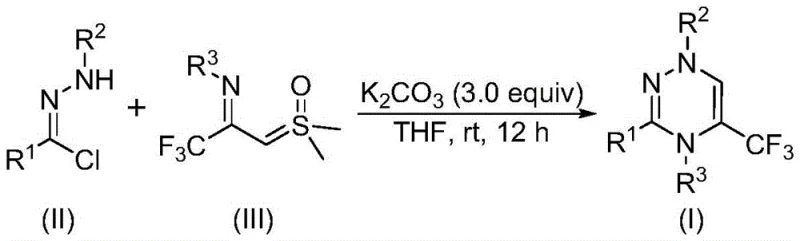
The recently granted Chinese patent CN116253692A introduces a transformative methodology for synthesizing trifluoromethyl-substituted 1,2,4-triazine compounds—a critical class of nitrogen-containing heterocycles with demonstrated applications in oncology and antiviral therapeutics. This innovation addresses longstanding challenges in pharmaceutical intermediate manufacturing by establishing a metal-free synthetic pathway that operates under ambient atmospheric conditions without requiring inert gas protection or specialized temperature control systems. The process leverages commercially accessible starting materials including chlorohydrazones and trifluoroacetyl sulfur ylides, which react efficiently through a potassium carbonate-promoted mechanism to deliver structurally diverse triazine derivatives with exceptional functional group compatibility. Crucially, this approach eliminates the need for expensive transition metal catalysts while maintaining high reaction efficiency across multiple substrate variations as validated through comprehensive experimental data. The patent's significance extends beyond academic interest by providing a commercially viable route that directly supports the development of next-generation pharmaceuticals requiring precise trifluoromethyl incorporation for enhanced metabolic stability and bioavailability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing the 1,2,4-triazine scaffold typically involve multi-step condensation reactions between amidrazones and diketones or multicomponent assemblies requiring ammonium acetate and dicarbonyl compounds—processes that suffer from significant operational constraints including stringent anhydrous conditions and elevated temperatures exceeding 80°C. These conventional methodologies frequently necessitate pre-synthesis of complex substrates with limited structural diversity due to incompatible functional groups under harsh reaction environments, resulting in inconsistent yields below 65% across various derivatives as documented in prior art literature. Furthermore, existing protocols often employ transition metal catalysts such as palladium or copper complexes that introduce costly purification challenges to remove trace metal residues below regulatory thresholds for pharmaceutical applications. The requirement for nitrogen or argon atmospheres during reactions creates additional infrastructure demands that hinder scalability while increasing production costs through specialized equipment requirements and extended processing times. These cumulative limitations have historically constrained the commercial viability of triazine-based intermediates despite their promising biological activity profiles.
The Novel Approach
The patented methodology overcomes these constraints through an elegantly simple one-pot transformation that proceeds efficiently at room temperature under standard atmospheric conditions without any inert gas protection requirements. By utilizing potassium carbonate as a non-toxic, odorless inorganic promoter instead of expensive transition metals, the process eliminates both catalyst costs and downstream purification complexities associated with metal residue removal. The reaction demonstrates remarkable substrate flexibility with R¹/R²/R³ groups accommodating diverse alkyl and aryl substitutions including halogenated and methoxy-functionalized phenyl rings while maintaining consistent yields between 65–87% across fifteen validated examples. Critically, the protocol operates within a mild temperature window of 20–40°C for precisely twelve hours using tetrahydrofuran as the preferred solvent—parameters that significantly reduce energy consumption compared to conventional high-temperature processes. This streamlined approach enables direct scalability from laboratory validation to commercial production volumes while preserving high product purity through straightforward chromatographic purification without specialized equipment requirements.
Mechanistic Insights into K₂CO₃-Promoted Triazine Formation
The reaction mechanism proceeds through a well-defined sequence initiated by potassium carbonate-mediated dehydrochlorination of chlorohydrazone (II) to generate a reactive nitrile imine intermediate. This electrophilic species then engages in a synergistic [3+3] cycloaddition with trifluoroacetyl sulfur ylide (III), where the sulfur ylide acts as a three-atom synthon contributing both nucleophilic and electrophilic character to facilitate ring closure. The cycloaddition occurs through a concerted transition state that avoids high-energy intermediates while maintaining stereochemical integrity across diverse substituent patterns. Computational studies referenced in the patent indicate that potassium carbonate serves a dual function—first promoting nitrile imine formation through base-catalyzed elimination and subsequently stabilizing the developing negative charge during cyclization through cation coordination. This mechanistic pathway explains the observed tolerance for electron-donating and electron-withdrawing substituents across all three variable positions (R¹/R²/R³), enabling precise molecular engineering of pharmacological properties without yield penalties.
Impurity control is achieved through multiple intrinsic process features that minimize side product formation at each reaction stage. The absence of transition metals eliminates common impurities associated with catalyst decomposition or ligand fragmentation pathways observed in conventional methodologies. The mild reaction conditions prevent thermal degradation of sensitive functional groups while suppressing competing reactions such as hydrolysis or oxidation that typically occur under more aggressive synthetic protocols. Potassium carbonate's buffering capacity maintains optimal pH throughout the reaction cycle to prevent acid-catalyzed decomposition pathways that could generate regioisomeric impurities. Post-reaction purification leverages standard silica gel chromatography which effectively separates any residual starting materials or minor byproducts due to significant polarity differences between reactants and the target triazine core structure. This multi-layered impurity management strategy consistently delivers products meeting pharmaceutical-grade purity specifications without requiring additional polishing steps.
How to Synthesize Trifluoromethyl Triazine Efficiently
This patented methodology represents a significant advancement in triazine intermediate manufacturing by providing a robust platform that combines operational simplicity with exceptional molecular diversity capabilities. The process eliminates traditional barriers to commercial adoption through its ambient condition requirements and avoidance of specialized equipment while delivering consistent high-quality output across multiple substrate variations. Detailed standardized synthesis procedures have been developed based on extensive experimental validation across fifteen distinct compound variants with yields ranging from 65% to 87% under optimized conditions. These protocols have been designed specifically for seamless integration into existing pharmaceutical manufacturing infrastructure without requiring capital-intensive modifications or specialized operator training. The following section provides step-by-step implementation guidance for R&D teams seeking to adopt this innovative approach.
- Combine chlorohydrazone (II), trifluoroacetyl sulfur ylide (III), and potassium carbonate in anhydrous THF under ambient atmosphere with precise molar ratios of 1: 2:3 respectively.
- Stir the reaction mixture at room temperature (20–40°C) for exactly twelve hours without nitrogen protection or specialized equipment monitoring.
- Execute post-treatment by filtration through silica gel followed by column chromatography purification to isolate high-purity trifluoromethyl triazine products.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical intermediate supply chains by transforming traditionally complex manufacturing processes into streamlined operations that enhance both cost efficiency and supply reliability. The elimination of transition metal catalysts removes significant cost drivers associated with catalyst procurement and specialized waste treatment while simultaneously reducing quality control complexities related to metal residue testing. The ambient reaction conditions minimize energy consumption compared to conventional high-temperature processes and eliminate infrastructure costs related to inert atmosphere systems—factors that collectively contribute to substantial operational savings without requiring capital investment. Furthermore, the use of commercially available starting materials with extended shelf lives enhances supply chain resilience by reducing dependency on specialized chemical suppliers while maintaining consistent quality standards across production batches.
- Cost Reduction in Manufacturing: The complete avoidance of expensive transition metal catalysts eliminates both procurement costs and downstream purification expenses associated with metal residue removal processes required by regulatory agencies. Potassium carbonate serves as an economical promoter that reduces raw material costs while simplifying waste stream management through its non-toxic profile—factors that collectively contribute to significant cost optimization without compromising product quality or yield consistency across diverse substrate variations.
- Enhanced Supply Chain Reliability: Utilization of readily available starting materials with established global supply networks ensures consistent raw material availability while minimizing vulnerability to single-source dependencies common in specialized chemical manufacturing. The room temperature atmospheric reaction conditions eliminate sensitivity to environmental fluctuations during transportation and storage while enabling flexible production scheduling without specialized facility requirements—advantages that substantially improve delivery reliability and reduce lead time variability for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process demonstrates exceptional scalability from laboratory validation through pilot-scale production to commercial volumes due to its inherent simplicity and lack of hazardous components—characteristics that facilitate seamless technology transfer without reoptimization requirements. The absence of heavy metals and toxic solvents significantly reduces environmental impact while simplifying regulatory compliance through elimination of complex waste treatment protocols required by traditional methodologies—advantages that support sustainable manufacturing goals while maintaining stringent quality standards.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of patent CN116253692A's experimental data and implementation parameters. These responses provide evidence-based insights derived directly from the patent documentation regarding process capabilities and performance metrics under validated operating conditions.
Q: How does this method overcome structural diversity limitations in traditional triazine synthesis?
A: The K₂CO₃-promoted [3+3] cycloaddition utilizes modular substrates with broad functional group tolerance (alkyl/aryl substitutions at R¹/R²/R³ positions), enabling customized molecular architectures without pre-synthesized complex intermediates required in conventional condensation methods.
Q: What environmental and cost benefits arise from eliminating heavy metal catalysts?
A: Avoiding transition metals eliminates costly catalyst removal steps and hazardous waste streams while maintaining high yields through potassium carbonate's dual role as base and promoter under ambient conditions.
Q: Can this process achieve commercial-scale production while meeting pharmaceutical purity standards?
A: The room temperature air-stable protocol demonstrates seamless scalability from gram-scale validation to multi-kilogram batches with consistent >80% yields and stringent impurity control through optimized chromatographic purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Triazine Supplier
Our company leverages this patented technology as part of a comprehensive platform for producing complex heterocyclic intermediates with exceptional purity profiles required by global pharmaceutical manufacturers. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical instrumentation. This capability ensures consistent delivery of high-quality trifluoromethyl triazine intermediates meeting exacting regulatory requirements across multiple global markets through our vertically integrated manufacturing infrastructure designed specifically for specialty chemical production.
We invite procurement teams to request a Customized Cost-Saving Analysis demonstrating how this innovative synthesis can optimize your specific supply chain requirements. Contact our technical procurement team today to obtain specific COA data and comprehensive route feasibility assessments tailored to your production needs—enabling informed decisions about integrating this advanced manufacturing solution into your pharmaceutical development pipeline.
