Advanced Green Synthesis of Chromanone Compounds for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and cost-effective pathways for synthesizing complex heterocyclic scaffolds. A significant breakthrough in this domain is documented in patent CN111943924B, which details a novel synthetic method for chromanone compounds. This technology represents a paradigm shift from traditional noble metal catalysis to a more economical iron-based system. By leveraging a unique combination of persulfate oxidants and ferric chloride catalysts within a deep eutectic solvent (DES) matrix, this method achieves high-efficiency cyclization under mild aerobic conditions. For R&D directors and procurement strategists, this innovation offers a compelling alternative to legacy processes that are often burdened by high catalyst costs and stringent environmental regulations. The ability to synthesize these biologically active cores without expensive silver catalysts marks a critical advancement in green chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chromanone derivatives has relied heavily on transition metal catalysis, particularly utilizing silver salts. As noted in prior art references such as Chem. Commun. 2016 and Adv. Synth. Catal. 2017, these conventional routes typically require stoichiometric or near-stoichiometric amounts of expensive silver catalysts. Furthermore, these reactions are often conducted in volatile organic solvents that are difficult to recover and recycle, leading to substantial waste generation and increased disposal costs. The reliance on precious metals not only inflates the raw material expenditure but also introduces risks related to metal residue contamination in the final API intermediates. Additionally, many of these traditional protocols demand rigorous exclusion of oxygen, necessitating complex inert atmosphere setups that increase operational overhead and limit scalability in standard manufacturing facilities.
The Novel Approach
The methodology disclosed in CN111943924B fundamentally re-engineers this synthetic landscape by replacing the costly silver system with an inexpensive iron-based catalytic couple. This new approach utilizes readily available potassium or sodium persulfate alongside ferric chloride hexahydrate to drive the radical cyclization process. Crucially, the reaction medium is a deep eutectic solvent formed from choline chloride and urea, which serves as both a green solvent and a potential co-catalyst. This system operates effectively under simple air conditions at moderate temperatures ranging from 60°C to 80°C. The elimination of precious metals and the adoption of a recyclable solvent system drastically simplify the workup procedure and reduce the environmental footprint. This transition from a high-cost, high-waste model to a low-cost, circular economy model provides a robust foundation for the commercial manufacturing of high-purity chromanone intermediates.
Mechanistic Insights into Iron-Catalyzed Radical Cyclization
The core of this synthetic innovation lies in its radical-mediated mechanism, which proceeds efficiently without the need for noble metals. The reaction initiates with the activation of the sodium benzenesulfinate by the persulfate and ferric chloride system within the deep eutectic solvent. This interaction generates a sulfinic acid radical species, which is the key reactive intermediate. This radical subsequently undergoes addition to the alkene double bond present in the 2-(allyloxy)benzaldehyde substrate. 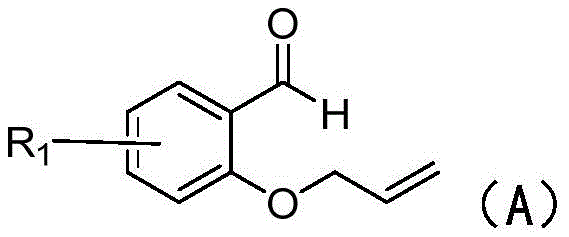 Following this addition, a carbon-centered radical is formed, which then undergoes an intramolecular cyclization with the pendant aldehyde group. This cyclization step constructs the chromanone ring system, generating an oxygen-centered radical that is finally quenched by hydrogen abstraction to yield the target ketone product. The mildness of this radical pathway ensures high chemoselectivity, minimizing the formation of side products that often plague harsher oxidative conditions.
Following this addition, a carbon-centered radical is formed, which then undergoes an intramolecular cyclization with the pendant aldehyde group. This cyclization step constructs the chromanone ring system, generating an oxygen-centered radical that is finally quenched by hydrogen abstraction to yield the target ketone product. The mildness of this radical pathway ensures high chemoselectivity, minimizing the formation of side products that often plague harsher oxidative conditions.
From an impurity control perspective, the use of a deep eutectic solvent plays a pivotal role in stabilizing the radical intermediates and suppressing unwanted side reactions. The hydrogen-bonding network of the choline chloride-urea mixture likely modulates the reactivity of the iron species, preventing over-oxidation or polymerization of the sensitive allyl groups. This results in a cleaner reaction profile, which is essential for pharmaceutical applications where strict impurity thresholds must be met. The ability to tolerate various substituents on the aromatic ring, such as bromo, fluoro, or methyl groups, further demonstrates the robustness of this mechanistic pathway. 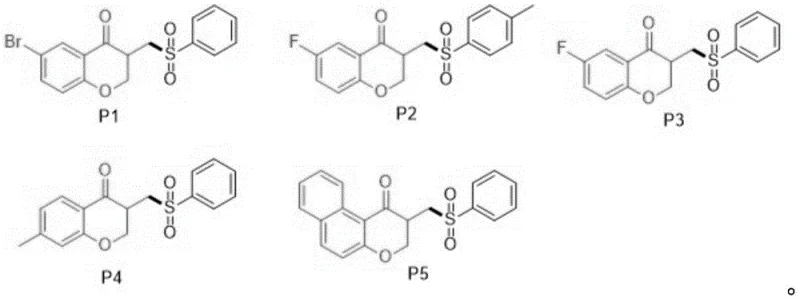 The structural diversity shown in the product library confirms that the electronic nature of the substituents does not significantly hinder the radical cyclization, allowing for the synthesis of a wide range of analogues suitable for SAR studies.
The structural diversity shown in the product library confirms that the electronic nature of the substituents does not significantly hinder the radical cyclization, allowing for the synthesis of a wide range of analogues suitable for SAR studies.
How to Synthesize Chromanone Compounds Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires precise adherence to the optimized reaction parameters defined in the patent. The process is designed to be operationally simple, avoiding the need for specialized equipment like gloveboxes or high-pressure reactors. The key to success lies in the preparation of the deep eutectic solvent and the accurate dosing of the oxidant-to-catalyst ratio. The following guide outlines the standardized protocol derived from the patent examples, ensuring reproducibility and high yield. For detailed step-by-step instructions including specific quantities and purification methods, please refer to the technical guide below.
- Charge the reactor with 2-(allyloxy)benzaldehyde derivatives or 2-(allyloxy)naphthaldehyde and sodium benzenesulfinate compounds.
- Add the catalyst system consisting of persulfate and ferric chloride hexahydrate into the deep eutectic solvent mixture.
- Heat the reaction mixture at 60-80°C under air conditions for 6-10 hours, then isolate the product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of adopting this technology are profound. The primary driver for cost reduction is the complete elimination of silver catalysts, which are subject to volatile market pricing and supply constraints. By switching to iron salts and persulfates, the raw material cost per kilogram of product is significantly lowered. Furthermore, the recyclability of the deep eutectic solvent means that solvent purchase and waste disposal costs are drastically reduced over the lifecycle of the production campaign. This creates a more predictable and stable cost structure, shielding the supply chain from fluctuations in precious metal markets and organic solvent availability.
- Cost Reduction in Manufacturing: The substitution of expensive silver catalysts with abundant iron salts directly translates to lower direct material costs. Additionally, the ability to recycle the reaction solvent multiple times without significant loss of efficiency reduces the consumption of bulk chemicals. This dual saving mechanism—lower catalyst cost and lower solvent cost—results in substantial overall manufacturing savings, making the final intermediates more price-competitive in the global market.
- Enhanced Supply Chain Reliability: The reagents required for this process, such as ferric chloride, potassium persulfate, choline chloride, and urea, are commodity chemicals produced on a massive global scale. Unlike specialized ligands or noble metals that may have limited suppliers, these materials are readily available from multiple sources. This diversification of the supply base mitigates the risk of shortages and ensures continuous production capability, which is critical for meeting the demanding delivery schedules of pharmaceutical clients.
- Scalability and Environmental Compliance: The reaction operates under air atmosphere and moderate temperatures, removing the need for complex inert gas infrastructure and extreme heating or cooling systems. This simplicity facilitates easier scale-up from gram to tonne scale. Moreover, the use of a biodegradable deep eutectic solvent aligns with increasingly stringent environmental regulations regarding VOC emissions and hazardous waste. This compliance reduces the regulatory burden and potential fines associated with traditional organic solvent usage, future-proofing the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method. They are derived from the specific advantages and operational details outlined in the patent documentation. Understanding these aspects helps stakeholders evaluate the feasibility of integrating this technology into their existing supply chains.
Q: Why is the new iron-catalyzed method superior to traditional silver-catalyzed synthesis?
A: Traditional methods rely on expensive silver catalysts and non-recyclable organic solvents, leading to high production costs and environmental concerns. The new method utilizes inexpensive iron salts and recyclable deep eutectic solvents, significantly reducing raw material costs and waste generation while maintaining high yields.
Q: Can the deep eutectic solvent be reused in large-scale manufacturing?
A: Yes, the deep eutectic solvent system composed of choline chloride and urea demonstrates excellent stability and recyclability. Patent data indicates the solvent can be recycled multiple times without significant loss in catalytic efficiency, which is crucial for sustainable industrial operations.
Q: What are the safety advantages of performing this reaction under air conditions?
A: Operating under ambient air conditions eliminates the need for complex inert gas protection systems (such as nitrogen or argon purging). This simplifies the reactor setup, reduces operational complexity, and enhances overall process safety for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chromanone Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed synthesis for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific requirements of deep eutectic solvent systems and radical chemistry, ensuring that the transition from lab to plant is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chromanone compounds meets the highest quality standards required for downstream drug development.
We invite you to collaborate with us to leverage this cost-effective and green technology for your projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can optimize your supply chain and accelerate the delivery of life-saving medicines to the market.
