Scalable Synthesis of Trans-4-N-Boc-Aminocyclohexane Carboxylic Acid via Lewis Acid Catalysis
Scalable Synthesis of Trans-4-N-Boc-Aminocyclohexane Carboxylic Acid via Lewis Acid Catalysis
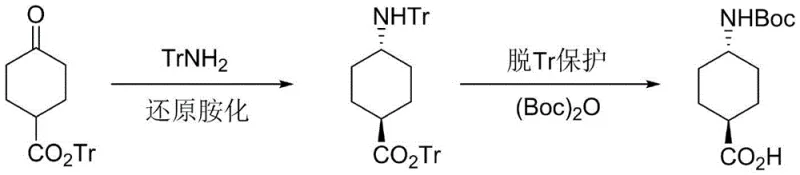
The pharmaceutical industry constantly seeks robust and scalable routes for critical intermediates, particularly those used in the synthesis of complex therapeutic agents like matrix metalloproteinase inhibitors. Patent CN109824545B discloses a highly efficient preparation method for trans-4-N-Boc-aminocyclohexane carboxylic acid, a key building block in medicinal chemistry. This novel approach utilizes 4-oxocyclohexanecarboxylic acid triphenylmethyl ester as the starting material, undergoing a strategic two-step transformation involving selective reductive amination followed by deprotection and amino protection. The significance of this technology lies in its ability to overcome the stereochemical challenges associated with traditional synthesis methods, delivering the desired trans-isomer with exceptional purity exceeding 99 percent. For R&D teams and procurement specialists alike, this represents a substantial advancement in process reliability and cost-effectiveness for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cyclohexane-based amino acids has relied heavily on the catalytic hydrogenation of aromatic precursors, such as 4-aminobenzoic acid. While conceptually straightforward, this conventional pathway suffers from significant drawbacks that hinder industrial efficiency. The hydrogenation process, typically employing platinum oxide (PtO2) catalysts at elevated temperatures, often results in a thermodynamic mixture of cis and trans isomers. Literature indicates that the ratio of cis to trans products can be as unfavorable as 16:49, necessitating complex and yield-loss-inducing separation steps to isolate the desired trans-configuration. Furthermore, the overall yield of these traditional routes is frequently suboptimal, reported around 66 percent, which translates to higher raw material consumption and increased waste generation. These inefficiencies create bottlenecks in the supply chain, driving up costs and extending lead times for downstream API manufacturing.
The Novel Approach
In stark contrast, the methodology outlined in patent CN109824545B introduces a paradigm shift by employing a Lewis acid-catalyzed reductive amination strategy. Instead of reducing an aromatic ring, this route starts with a functionalized cyclohexanone derivative, specifically 4-oxocyclohexanecarboxylic acid triphenylmethyl ester. By reacting this ketone with triphenylmethylamine in the presence of a Lewis acid and a reducing agent, the process achieves high stereoselectivity for the trans-isomer directly. The bulky trityl groups on both the amine and the ester moiety likely exert significant steric influence, directing the incoming hydride to favor the trans-configuration. This inherent selectivity minimizes the formation of the cis-impurity, thereby simplifying the purification process to a straightforward recrystallization. The result is a streamlined workflow that bypasses the isomer separation nightmare of hydrogenation, offering a cleaner, more direct path to the target molecule.
Mechanistic Insights into Lewis Acid-Catalyzed Reductive Amination
The core of this innovative synthesis lies in the precise control of stereochemistry during the reductive amination step. The reaction mechanism involves the initial formation of an imine or iminium ion intermediate between the ketone substrate and triphenylmethylamine. The addition of a Lewis acid, such as titanium tetrachloride (TiCl4), zinc chloride (ZnCl2), or tetraisopropyl titanate, plays a dual role: it activates the carbonyl oxygen towards nucleophilic attack by the amine and stabilizes the resulting iminium species. This activation lowers the energy barrier for imine formation and facilitates the subsequent reduction by hydride sources like sodium borohydride, sodium cyanoborohydride, or sodium triacetoxyborohydride. The steric bulk of the trityl protecting group is crucial here; it forces the cyclohexane ring into a conformation where the incoming hydride attacks from the less hindered face, predominantly yielding the trans-amine. Experimental data from the patent demonstrates that optimizing the molar ratios of the Lewis acid and reducing agent is critical, with ratios ranging from 1:1.2 to 1:2.0 proving effective in maximizing conversion while maintaining high trans-selectivity.
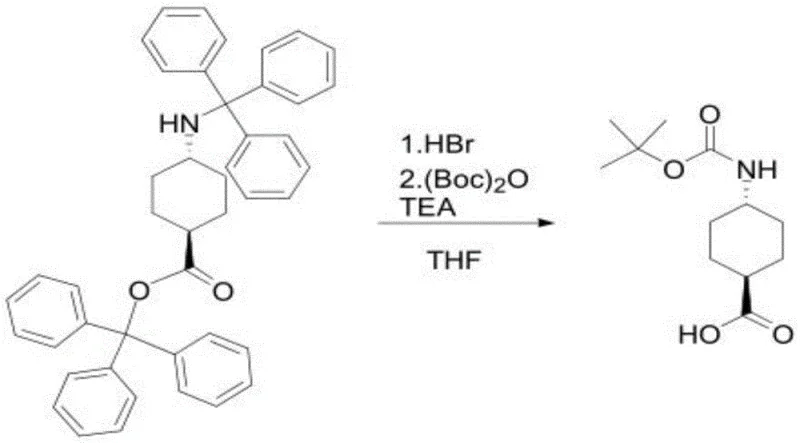
Following the reductive amination, the second phase of the synthesis involves the removal of the trityl protecting groups and the installation of the Boc group. The trityl group is acid-labile, allowing for its cleavage under mild acidic conditions using hydrobromic acid (HBr) or hydrochloric acid (HCl). This deprotection step generates the free amine as a salt, typically the hydrochloride. Subsequently, the amine is protected with di-tert-butyl dicarbonate ((Boc)2O) under basic conditions, utilizing bases like triethylamine or pyridine. This sequence ensures that the sensitive amino acid functionality is preserved while providing a stable, protected intermediate suitable for further peptide coupling or drug synthesis. The use of THF or DCM as solvents in this stage ensures good solubility and reaction homogeneity, contributing to the high final purity of over 99 percent observed in the examples.
How to Synthesize Trans-4-N-Boc-Aminocyclohexane Carboxylic Acid Efficiently
Implementing this synthesis requires careful attention to reaction conditions, particularly temperature control and stoichiometry, to replicate the high yields and purity reported in the patent. The process is divided into two distinct stages: the stereoselective reductive amination to form the bis-trityl intermediate, and the subsequent deprotection-protection sequence to yield the final Boc-amino acid. Operators must ensure an inert atmosphere, typically nitrogen, is maintained throughout the reductive amination to prevent side reactions. The choice of solvent, whether ethanol, methanol, or acetonitrile, can influence the reaction rate and selectivity, with ethanol often providing a good balance of solubility and cost. Detailed standard operating procedures for each step, including specific quenching and extraction protocols, are essential for consistent batch-to-batch quality.
- Perform reductive amination of 4-oxocyclohexanecarboxylic acid triphenylmethyl ester with triphenylmethylamine using a Lewis acid catalyst and reducing agent.
- Purify the resulting trans-intermediate via recrystallization to achieve high stereochemical purity.
- Remove the trityl protecting group under acidic conditions and subsequently protect the amine with Boc anhydride under basic conditions.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The elimination of precious metal catalysts like platinum, which are subject to volatile market pricing and supply constraints, represents a significant strategic advantage. By shifting to base metal Lewis acids and common hydride reagents, manufacturers can drastically reduce raw material costs and mitigate the risk of supply disruptions. Furthermore, the simplified purification process, which relies on recrystallization rather than complex chromatographic separations, reduces solvent consumption and processing time. This operational efficiency translates directly into lower manufacturing costs and a more competitive price point for the final intermediate, enabling better margin management for downstream API producers.
- Cost Reduction in Manufacturing: The replacement of expensive platinum catalysts with abundant Lewis acids like titanium tetrachloride or zinc chloride fundamentally alters the cost structure of the synthesis. Additionally, the high stereoselectivity reduces the loss of material associated with separating cis/trans isomers, effectively increasing the atom economy of the process. The ability to use standard reducing agents and solvents further contributes to substantial cost savings, making the overall production economics far more favorable compared to traditional hydrogenation routes.
- Enhanced Supply Chain Reliability: The starting materials for this process, including 4-oxocyclohexanecarboxylic acid derivatives and triphenylmethylamine, are commercially available and stable, ensuring a reliable supply chain. Unlike processes dependent on specialized catalysts or high-pressure hydrogenation equipment, this method utilizes standard glass-lined or stainless steel reactors found in most fine chemical facilities. This compatibility with existing infrastructure reduces the barrier to entry for contract manufacturing organizations, thereby increasing the number of potential suppliers and enhancing supply security.
- Scalability and Environmental Compliance: The patent explicitly demonstrates the scalability of this method, with successful runs conducted on a multi-kilogram scale (e.g., 3.86 kg of starting material) yielding consistent results. The use of recyclable solvents like ethanol and ethyl acetate, combined with the absence of heavy metal waste streams, aligns well with modern environmental regulations. This green chemistry profile simplifies waste treatment and disposal, reducing the environmental footprint and regulatory burden associated with large-scale production.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its implementation. The following questions address common concerns regarding reaction conditions, catalyst selection, and product quality, drawing directly from the experimental data provided in the patent documentation. These insights are intended to clarify the feasibility of the process for various production scales and to highlight the robustness of the methodology under different operational parameters.
Q: What is the primary advantage of this synthesis route over traditional hydrogenation methods?
A: The primary advantage is the significantly improved stereoselectivity for the trans-isomer. Traditional hydrogenation of 4-aminobenzoic acid often yields a mixture of cis and trans isomers with difficult separation, whereas this Lewis acid catalyzed reductive amination inherently favors the trans-configuration, simplifying purification and boosting overall yield.
Q: Which Lewis acids are suitable for this reductive amination process?
A: The patent specifies several effective Lewis acids including titanium tetrachloride (TiCl4), zinc chloride (ZnCl2), and tetraisopropyl titanate. These catalysts activate the carbonyl group for imine formation and subsequent reduction, ensuring high conversion rates and selectivity.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process has been demonstrated on a multi-kilogram scale (e.g., 3.86 kg starting material) with consistent high purity (>99%) and yield. The use of common solvents like ethanol and THF, along with standard workup procedures, makes it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-4-N-Boc-Aminocyclohexane Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our technical team has thoroughly analyzed the pathway described in patent CN109824545B and possesses the expertise to execute this Lewis acid-catalyzed reductive amination with precision. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of trans-4-N-Boc-aminocyclohexane carboxylic acid meets the highest industry standards for pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. By leveraging our process development capabilities, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to bring your next generation of therapeutics to market faster and more efficiently.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
