Advanced Copper-Catalyzed Synthesis of Imidazolium Salts for Industrial Scale-Up
The chemical industry is constantly seeking more efficient pathways to access functionalized heterocyclic scaffolds, particularly imidazolium onium salts, which serve as critical precursors for N-heterocyclic carbenes (NHCs) and advanced ionic liquids. A pivotal advancement in this domain is documented in patent CN102863387A, which discloses a novel synthetic methodology that overcomes the historical limitations of traditional alkylation and condensation strategies. This technology leverages the reactivity of diaryliodonium salts coupled with imidazole derivatives under metal catalysis to generate a diverse array of N3-aryl substituted imidazolium salts. For R&D directors and procurement specialists evaluating supply chains for high-performance materials, this patent represents a significant shift towards more atom-economical and operationally simple processes. The ability to synthesize these salts with high purity and yield under relatively mild conditions addresses key bottlenecks in the production of electrolytes, organocatalysts, and specialized polymer additives. By adopting this methodology, manufacturers can secure a more robust supply of these high-value intermediates while minimizing the environmental footprint associated with legacy synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of imidazolium onium salts has relied heavily on two primary strategies, both of which suffer from significant drawbacks that hinder industrial scalability and cost-effectiveness. The first conventional approach involves nucleophilic substitution, typically starting from an imidazole ring that undergoes Ullman coupling to form a 1-substituted imidazole, followed by direct quaternization. This multi-step sequence is inherently inefficient; it is largely restricted to reactions with primary halogenated hydrocarbons, as secondary or tertiary halides tend to undergo elimination reactions rather than the desired substitution. Furthermore, this method struggles to introduce chiral groups or aryl substituents directly onto the nitrogen atom, limiting the structural diversity of the final products. The second traditional strategy employs condensation reactions using methine species with two leaving groups and 1,2-diimines. While this allows for some variation in substituents, it is plagued by lengthy synthetic steps, relatively low overall yields, and the requirement for harsh reaction conditions that demand specialized equipment and rigorous safety protocols. These factors collectively render conventional methods less attractive for the commercial scale-up of complex imidazolium derivatives required in modern electronic and pharmaceutical applications.
The Novel Approach
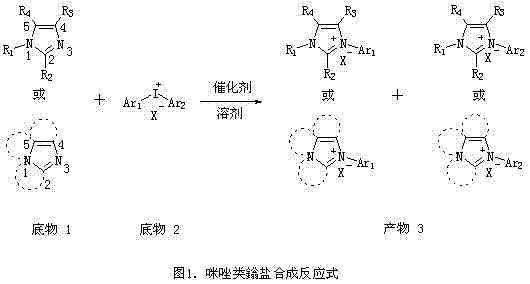
In stark contrast to these legacy techniques, the methodology outlined in CN102863387A introduces a direct and highly efficient coupling reaction between imidazole derivatives and iodonium salts. This innovative route bypasses the need for pre-functionalization or harsh condensation conditions, enabling the direct introduction of aryl groups at the N3 position of the imidazole ring. The process utilizes readily available diaryliodonium salts as electrophilic coupling partners, which react with various substituted imidazoles in the presence of a metal catalyst. This approach is characterized by its remarkable universality, successfully synthesizing novel imidazolium salts that were previously difficult or impossible to access via traditional nucleophilic substitution. The reaction conditions are notably mild, typically operating at temperatures around 100°C, and the procedure is operationally simple, requiring only standard mixing and heating followed by straightforward purification. This paradigm shift not only expands the chemical space accessible to researchers but also offers a practical solution for the industrial manufacturing of high-purity imidazolium salts with reduced operational complexity.
Mechanistic Insights into Copper-Catalyzed N-Arylation
The core of this synthetic breakthrough lies in the metal-catalyzed activation of the iodonium salt, which facilitates the transfer of an aryl group to the nitrogen atom of the imidazole derivative. Experimental data within the patent highlights the critical role of the catalyst choice, with Group IB metal salts demonstrating superior performance compared to other transition metals. Specifically, copper(II) acetate monohydrate (Cu(OAc)2·H2O) acts as a highly effective catalyst, likely functioning through a mechanism involving oxidative addition of the iodonium salt to the metal center, followed by coordination of the imidazole nitrogen and subsequent reductive elimination to form the N-C bond. The patent data explicitly compares various metal salts, showing that while magnesium, zinc, titanium, chromium, manganese, and rhodium salts can catalyze the reaction, they result in significantly lower yields ranging from 4% to 15%. In contrast, the optimized copper catalyst system achieves yields as high as 97% for model substrates like 1,3-diphenylimidazolium tetrafluoroborate. This mechanistic efficiency ensures that the reaction proceeds with high selectivity, minimizing the formation of side products and simplifying the downstream purification process, which is a crucial consideration for maintaining high purity specifications in fine chemical manufacturing.
Furthermore, the reaction exhibits a high degree of tolerance towards different substituents on both the imidazole ring and the iodonium salt, allowing for the synthesis of a broad library of derivatives. The patent details successful reactions with various substituted imidazoles, including those with alkyl, alkenyl, alkynyl, and fused-ring structures, as well as iodonium salts bearing different counterions such as tetrafluoroborate, tosylate, and nitrate. The choice of solvent also plays a pivotal role in the reaction kinetics and yield; aprotic polar solvents like N,N-dimethylformamide (DMF) are preferred, providing a conducive environment for the ionic intermediates involved in the catalytic cycle. The molar ratio of reactants is another key parameter, with a ratio of 1:1.5 (imidazole to iodonium salt) identified as optimal for balancing conversion and cost. Understanding these mechanistic nuances allows process chemists to fine-tune the reaction parameters for specific target molecules, ensuring consistent quality and reproducibility across different batches, which is essential for meeting the stringent regulatory requirements of the pharmaceutical and electronic material sectors.
How to Synthesize Imidazolium Onium Salts Efficiently
Implementing this novel synthetic route in a laboratory or pilot plant setting requires careful attention to the specific reaction parameters identified in the patent to maximize yield and purity. The process begins with the precise weighing and charging of the imidazole derivative and the diaryliodonium salt into a suitable reactor, followed by the addition of the copper catalyst and the chosen solvent. The mixture is then heated to the specified temperature, typically around 100°C, and maintained under stirring for a duration of approximately 4 hours to ensure complete consumption of the starting materials. Post-reaction workup involves the removal of the solvent, often under reduced pressure, followed by purification of the crude solid residue. The patent suggests that column chromatography or recrystallization can be effectively employed to isolate the final product with high purity, as evidenced by the detailed characterization data including NMR and HRMS provided for multiple examples. For detailed standardized synthesis steps, please refer to the guide below.
- Charge the reactor with imidazole derivatives, diaryliodonium salts, a Group IB metal catalyst such as copper acetate, and an aprotic solvent like DMF.
- Heat the reaction mixture to approximately 100°C and maintain stirring for about 4 hours to ensure complete conversion.
- Remove the solvent post-reaction and purify the crude residue via column chromatography or recrystallization to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this copper-catalyzed iodonium coupling strategy offers substantial benefits for procurement managers and supply chain heads looking to optimize costs and ensure supply continuity. The primary advantage lies in the significant simplification of the manufacturing process, which eliminates the need for multi-step sequences and harsh reaction conditions associated with conventional methods. By reducing the number of unit operations and avoiding the use of difficult-to-handle reagents like secondary or tertiary alkyl halides, the overall production cost is drastically lowered. This process intensification translates directly into improved margin potential for suppliers and more competitive pricing for end-users. Additionally, the use of readily available starting materials such as simple imidazole derivatives and commercially sourced iodonium salts enhances supply chain reliability, reducing the risk of bottlenecks caused by scarce or specialized reagents. The mild reaction conditions also imply lower energy consumption and reduced wear on processing equipment, contributing to long-term operational sustainability and lower capital expenditure requirements for scaling up production capacity.
- Cost Reduction in Manufacturing: The elimination of complex multi-step synthesis routes and the use of a highly efficient copper catalyst significantly reduce the overall cost of goods sold. By avoiding expensive transition metals like rhodium or palladium and utilizing base metal catalysts that are effective at low loadings, the raw material costs are minimized. Furthermore, the high yields achieved, often exceeding 90% for optimized substrates, mean less waste generation and higher throughput per batch, which drives down the unit cost of the final imidazolium salt product. The simplified workup procedure, which avoids complex extraction or distillation steps, further reduces labor and utility costs, making this route economically superior to traditional condensation or nucleophilic substitution methods.
- Enhanced Supply Chain Reliability: The reliance on stable and commercially available iodonium salts and imidazole derivatives ensures a robust supply chain that is less susceptible to disruptions. Unlike methods requiring sensitive organometallic reagents or unstable intermediates, the reagents in this process are shelf-stable and easy to transport and store. This stability allows for better inventory management and reduces the lead time for high-purity imidazolium salts, enabling suppliers to respond more quickly to fluctuating market demands. The versatility of the method also means that a single production line can be adapted to manufacture a wide variety of derivatives by simply changing the substrate, providing flexibility to meet diverse customer specifications without the need for dedicated infrastructure for each product variant.
- Scalability and Environmental Compliance: The mild reaction conditions and the use of common solvents like DMF make this process highly scalable from gram to ton quantities without significant re-engineering. The reduced generation of hazardous byproducts and the avoidance of harsh acids or bases align with modern green chemistry principles, facilitating easier compliance with environmental regulations. The ability to recycle solvents and the high atom economy of the coupling reaction contribute to a lower environmental footprint, which is increasingly important for corporate sustainability goals. This scalability ensures that the supply of these critical intermediates can grow in tandem with the expanding markets for ionic liquids and NHC-based catalysts, securing long-term availability for downstream applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of imidazolium onium salts using this novel methodology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing manufacturing workflows or for sourcing high-quality intermediates for R&D projects. The insights provided here cover catalyst selection, reaction optimization, and product purification, offering a comprehensive overview of the process capabilities.
Q: What are the advantages of using iodonium salts over alkyl halides for imidazolium synthesis?
A: Unlike traditional alkyl halides which are limited to primary substrates and often require harsh conditions, iodonium salts allow for the direct introduction of aryl groups at the N3 position under mild conditions with significantly higher yields and broader substrate scope.
Q: Which catalyst system provides the optimal yield for this transformation?
A: Experimental data indicates that Group IB metal salts, specifically copper(II) acetate monohydrate (Cu(OAc)2·H2O), provide superior catalytic activity compared to magnesium, zinc, or titanium salts, achieving yields up to 97%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes readily available starting materials, operates at moderate temperatures around 100°C, and involves simple workup procedures like solvent removal and recrystallization, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidazolium Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed synthesis route for producing high-performance imidazolium salts. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including detailed impurity profiling via HPLC and NMR, to meet the exacting standards of the pharmaceutical and electronic materials industries. We are committed to leveraging advanced synthetic technologies like the one described in CN102863387A to deliver cost-effective and sustainable solutions for complex chemical challenges.
We invite global partners to collaborate with us to explore the full potential of these novel imidazolium derivatives for your specific applications. Whether you require custom synthesis for early-stage drug discovery or bulk manufacturing for industrial catalysts, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your project needs. We encourage you to reach out to our technical procurement team to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for efficiency, quality, and reliability.
