Advanced Synthesis of Z-4-Beta-Bromovinyl Benzoate for Commercial Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes for specialized intermediates that balance high stereochemical fidelity with economic viability. Patent CN101284782A introduces a significant advancement in the preparation of (Z)-4-(beta-bromovinyl) benzoate, a critical building block for drug discovery and polymer science. This technology addresses the longstanding challenges associated with synthesizing beta-halogenated olefins, offering a streamlined esterification pathway that bypasses the need for complex organometallic catalysis. By leveraging a DCC-mediated coupling strategy, this method ensures the preservation of the sensitive Z-configuration while utilizing readily available starting materials. For R&D directors and procurement specialists, understanding the nuances of this patent is essential for securing a reliable supply chain of high-purity pharmaceutical intermediates. The following analysis dissects the technical merits and commercial implications of this innovative synthesis protocol.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of stereospecific (Z)-beta-bromostyrene derivatives has relied heavily on transition metal-catalyzed coupling reactions or reduction processes that introduce significant operational burdens. As illustrated in prior art, methods often employ palladium catalysts such as Pd(PPh3)4 alongside toxic tributyltin hydride reagents to achieve stereoselective reduction of dibromoalkenes. 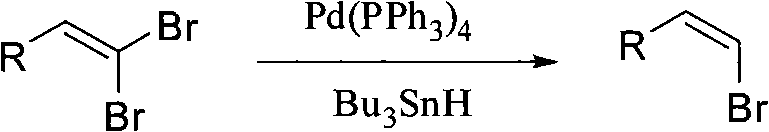 These conventional pathways are fraught with disadvantages, including the high cost of precious metal catalysts and the severe environmental and safety hazards associated with organotin byproducts. Furthermore, the removal of trace heavy metals from the final product to meet pharmaceutical standards requires additional, costly purification steps that reduce overall process efficiency. Alternative routes involving metal-halogen exchange or Julia olefination often suffer from harsh reaction conditions, poor stereoselectivity, or the requirement for expensive and unstable precursors. These factors collectively limit the industrial scalability of traditional methods, creating bottlenecks for manufacturers seeking cost reduction in pharmaceutical intermediate manufacturing.
These conventional pathways are fraught with disadvantages, including the high cost of precious metal catalysts and the severe environmental and safety hazards associated with organotin byproducts. Furthermore, the removal of trace heavy metals from the final product to meet pharmaceutical standards requires additional, costly purification steps that reduce overall process efficiency. Alternative routes involving metal-halogen exchange or Julia olefination often suffer from harsh reaction conditions, poor stereoselectivity, or the requirement for expensive and unstable precursors. These factors collectively limit the industrial scalability of traditional methods, creating bottlenecks for manufacturers seeking cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the methodology disclosed in CN101284782A utilizes a direct esterification strategy that operates under mild, ambient conditions. This novel approach starts with (Z)-4-(beta-bromovinyl) benzoic acid as a stable synthetic building block, reacting it with various monohydric alcohols in the presence of N,N'-dicyclohexylcarbodiimide (DCC) and a catalytic amount of 4-dimethylaminopyridine (DMAP). The reaction proceeds smoothly in benzene solvent at room temperature, eliminating the need for extreme temperatures or pressures that could compromise the stereochemical integrity of the vinyl bromide moiety. By avoiding transition metals entirely, this route inherently produces a cleaner crude product profile, simplifying downstream processing and waste management. The versatility of this method allows for the facile incorporation of different alcohol groups, enabling the rapid generation of diverse ester derivatives for structure-activity relationship studies. This shift from complex organometallic chemistry to straightforward condensation represents a paradigm shift in how high-value halogenated intermediates can be produced commercially.
Mechanistic Insights into DCC/DMAP Mediated Esterification
The core of this technological breakthrough lies in the efficient activation of the carboxylic acid group without affecting the sensitive carbon-bromine double bond. The reaction mechanism involves the formation of an O-acylisourea intermediate upon the interaction between the benzoic acid derivative and DCC. This activated species is highly susceptible to nucleophilic attack by the alcohol, a process that is significantly accelerated by the nucleophilic catalyst DMAP.  DMAP acts by forming a more reactive acylpyridinium intermediate, which lowers the activation energy for the esterification step, allowing the reaction to reach completion within 15 to 24 hours at room temperature. Crucially, the mildness of these reagents ensures that the Z-geometry of the beta-bromovinyl group remains intact throughout the transformation, preventing isomerization to the thermodynamically more stable E-isomer. This mechanistic pathway is particularly advantageous for maintaining the specific spatial arrangement required for subsequent cross-coupling reactions like Suzuki or Heck couplings in downstream drug synthesis.
DMAP acts by forming a more reactive acylpyridinium intermediate, which lowers the activation energy for the esterification step, allowing the reaction to reach completion within 15 to 24 hours at room temperature. Crucially, the mildness of these reagents ensures that the Z-geometry of the beta-bromovinyl group remains intact throughout the transformation, preventing isomerization to the thermodynamically more stable E-isomer. This mechanistic pathway is particularly advantageous for maintaining the specific spatial arrangement required for subsequent cross-coupling reactions like Suzuki or Heck couplings in downstream drug synthesis.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed routes. The primary byproduct of the reaction is dicyclohexylurea (DCU), which is insoluble in many organic solvents and can be easily removed by filtration or during the aqueous workup phase. Unlike palladium or tin residues which require specialized scavenging resins or extensive chromatography to meet strict ppm limits, DCU removal is straightforward and cost-effective. The absence of metal catalysts also eliminates the risk of metal-induced side reactions, such as homocoupling of the vinyl bromide, which can plague transition metal methodologies. The patent specifies precise molar ratios, such as a DCC to acid ratio of 1 to 1.2:1, optimizing reagent consumption while ensuring complete conversion. This level of control over the reaction stoichiometry contributes to a consistent impurity profile, a critical factor for regulatory compliance in the production of high-purity OLED material or pharmaceutical intermediates.
How to Synthesize (Z)-4-(beta-bromovinyl) benzoate Efficiently
Implementing this synthesis route requires careful attention to reagent quality and reaction monitoring to maximize yield and purity. The process begins with the dissolution of the acid substrate and alcohol in benzene, followed by the sequential addition of the catalyst and coupling agent. Maintaining the specified molar ratios is vital; for instance, using a slight excess of DCC ensures full activation of the acid, while controlling the amount of DMAP prevents potential side reactions.
- Prepare the reaction mixture by adding benzene solvent, Z-4-(beta-bromovinyl) benzoic acid, monohydric alcohol, and DMAP into a flask.
- Stir magnetically at room temperature for 5 to 20 minutes, then add DCC and continue reacting for 15 to 24 hours.
- Remove benzene under reduced pressure and purify the residue via column chromatography using ethyl acetate and petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this esterification-based methodology offers tangible benefits that extend beyond mere chemical elegance. The elimination of expensive transition metal catalysts and toxic tin reagents directly translates to substantial cost savings in raw material procurement and waste disposal. By removing the dependency on precious metals like palladium, manufacturers can insulate their production costs from the volatile fluctuations of the commodities market. Additionally, the simplified purification process reduces the consumption of silica gel and solvents typically required for heavy metal scavenging, further driving down the cost of goods sold. The use of common laboratory reagents like DCC and DMAP ensures that supply chains remain robust and resilient, as these chemicals are widely available from multiple global vendors. This accessibility mitigates the risk of supply disruptions that often accompany specialized organometallic catalysts.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the avoidance of noble metals and the simplification of downstream processing. Without the need for expensive metal scavengers or complex extraction protocols to remove tin residues, the operational expenditure per kilogram of product is significantly reduced. The reaction proceeds at room temperature, which eliminates the energy costs associated with heating or cooling large-scale reactors, contributing to a lower carbon footprint and reduced utility bills. Furthermore, the high yields reported in the patent examples indicate efficient atom economy, minimizing the waste of valuable starting materials. These factors combine to create a highly competitive cost structure for the commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical intermediates is paramount for continuous pharmaceutical production. This method relies on stable, shelf-stable reagents that do not require special storage conditions such as inert atmosphere gloveboxes or cryogenic freezing, unlike many organometallic catalysts. The robustness of the reaction conditions means that production can be scaled up with minimal risk of batch failure due to sensitive catalyst deactivation. By diversifying the chemical inputs away from single-source specialty catalysts, procurement teams can negotiate better terms and ensure continuity of supply. This stability is crucial for reducing lead time for high-purity pharmaceutical intermediates, allowing for more accurate forecasting and inventory management.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden complexities, but this esterification route is inherently scalable due to its mild exothermic profile and lack of hazardous gas evolution. The primary byproduct, DCU, is non-toxic and easily managed, simplifying compliance with increasingly stringent environmental regulations regarding heavy metal discharge. The ability to run the reaction at ambient pressure removes the need for specialized high-pressure equipment, lowering the capital expenditure required for plant expansion. Moreover, the high selectivity of the reaction minimizes the formation of difficult-to-separate isomers, ensuring that the final product consistently meets stringent purity specifications without extensive reprocessing. This alignment with green chemistry principles enhances the sustainability profile of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation to assist decision-makers in evaluating feasibility.
Q: What are the advantages of this esterification method over traditional palladium-catalyzed routes?
A: Unlike conventional methods requiring expensive palladium catalysts and toxic tin reagents, this process utilizes mild DCC/DMAP conditions at room temperature, significantly reducing heavy metal contamination risks and operational costs.
Q: Does this synthesis method maintain the stereoselectivity of the Z-isomer?
A: Yes, the patent data confirms that the esterification proceeds without isomerization, preserving the critical Z-configuration of the beta-bromovinyl group which is essential for downstream coupling reactions.
Q: What purification steps are required to achieve pharmaceutical grade purity?
A: The process involves removing the benzene solvent under reduced pressure followed by column chromatographic separation using specific ratios of ethyl acetate and petroleum ether to isolate the high-purity product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (Z)-4-(beta-bromovinyl) benzoate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development pipelines. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of (Z)-4-(beta-bromovinyl) benzoate meets the exacting standards required for pharmaceutical applications. Our commitment to quality assurance means that you can rely on us for consistent material that supports your regulatory filings and clinical trials without delay.
We invite you to leverage our expertise to optimize your supply chain and reduce overall project costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals. Let us partner with you to bring your next generation of therapeutics to market faster and more efficiently.
