Advanced Sulfonic Acid Catalysis for Directional Benzoyl Migration in Pharmaceutical Intermediates
The landscape of organic synthesis, particularly within the realm of complex natural product and pharmaceutical intermediate manufacturing, is constantly evolving to meet the demands for higher efficiency and sustainability. A significant breakthrough in this domain is documented in patent CN110724173B, which discloses a novel method for the directional migration of hydroxyl m-benzoyl groups based on sulfonic acid catalysis. This technology addresses a critical bottleneck in the total synthesis of carbohydrates and related polyol structures, where the precise positioning of protecting groups is paramount for downstream biological activity and chemical reactivity. By leveraging the unique ability of sulfonic acids to promote the formation of cyclic orthoester intermediates, this invention enables the high regioselective transfer of acyl functional groups under remarkably mild conditions. The strategic advantage lies in its atom economy and operational simplicity, offering a robust alternative to traditional methods that often rely on harsh reagents or expensive transition metals. For industrial partners seeking a reliable pharmaceutical intermediate supplier, understanding this catalytic innovation is key to optimizing production workflows for high-value API precursors.
The Limitations of Conventional Methods vs. The Novel Approach
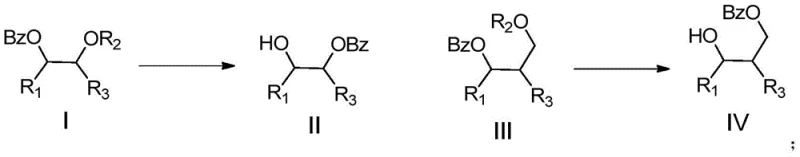
The Limitations of Conventional Methods
Historically, the manipulation of acyl groups within poly-functionalized molecules has been fraught with challenges regarding selectivity and condition severity. Conventional acyl migration strategies often depend on radical induction pathways or anionic mechanisms that require stringent control over reaction parameters to avoid side reactions. For instance, early methodologies utilizing silver tetrafluoroborate (AgBF4) or Lewis acids like zinc triflate necessitated the use of precious metals, which not only escalated raw material costs but also introduced significant complications in post-reaction purification due to heavy metal residue concerns. Furthermore, traditional approaches frequently struggled with poor regiocontrol, leading to mixtures of isomers that required laborious chromatographic separation, thereby reducing overall yield and throughput. In the context of sensitive carbohydrate scaffolds, the harsh conditions associated with these legacy methods often resulted in the degradation of the sugar backbone or the unintended cleavage of other acid-labile protecting groups, compromising the integrity of the final high-purity pharmaceutical intermediate.
The Novel Approach
In stark contrast, the sulfonic acid-catalyzed method described in the patent data introduces a paradigm shift by utilizing inexpensive and readily available Brønsted acids such as p-toluenesulfonic acid (TsOH) or methanesulfonic acid. This novel approach capitalizes on the spatial proximity of hydroxyl groups to form transient 5-membered or 6-membered orthoester rings, which act as the vehicle for the acyl transfer. The reaction proceeds with exceptional regioselectivity, capable of directing the benzoyl group from a secondary or tertiary hydroxyl to a primary hydroxyl, or specifically from an axial position to an equatorial position in cyclic systems. This level of precision eliminates the need for complex protection-deprotection sequences that traditionally inflate the step count of a synthesis. Moreover, the reaction conditions are exceedingly gentle, typically operating between 0°C and 30°C, which preserves the stereochemical integrity of chiral centers and prevents thermal decomposition. This represents a substantial advancement in cost reduction in API manufacturing by streamlining synthetic routes and minimizing waste generation.
Mechanistic Insights into Sulfonic Acid-Catalyzed Orthoester Rearrangement
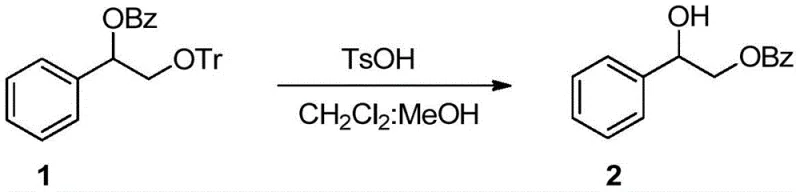
The core of this technological breakthrough lies in the mechanistic pathway involving the formation and hydrolysis of cyclic orthoester intermediates. When a substrate containing a benzoate ester and a spatially adjacent hydroxyl group is exposed to a sulfonic acid catalyst, the acid protonates the carbonyl oxygen, increasing its electrophilicity. This activation facilitates a nucleophilic attack by the neighboring hydroxyl group, resulting in the closure of a cyclic orthoester ring. The size of this ring, whether five-membered or six-membered, is dictated by the stereochemical arrangement of the substrate, which in turn governs the thermodynamic stability of the intermediate. Once formed, this orthoester species undergoes acid-catalyzed hydrolysis, but unlike random hydrolysis, the ring opening occurs in a highly specific manner that results in the net migration of the acyl group to the adjacent oxygen atom. This intramolecular process ensures that the acyl group does not simply detach into the solution but is transferred directly to the target position, thereby achieving the observed 1,2- or 1,3-migration with high fidelity.
From an impurity control perspective, this mechanism offers distinct advantages over intermolecular acylation strategies. Because the migration is intramolecular and mediated by a rigid cyclic transition state, the formation of scrambled byproducts or intermolecular transesterification products is significantly suppressed. The mild acidity of the sulfonic acid catalyst ensures that the reaction environment remains controlled, preventing the acid-catalyzed degradation of sensitive glycosidic bonds or other acid-labile functionalities often present in complex drug candidates. Additionally, the use of mixed solvent systems, such as dichloromethane with a minor fraction of methanol, helps to stabilize the transition state and solubilize the polar intermediates without competing aggressively in the transesterification equilibrium. This precise control over the reaction trajectory allows manufacturers to achieve superior purity profiles, which is a critical requirement for the commercial scale-up of complex pharmaceutical intermediates where regulatory compliance demands rigorous impurity profiling.
How to Synthesize Benzoyl Migration Intermediates Efficiently
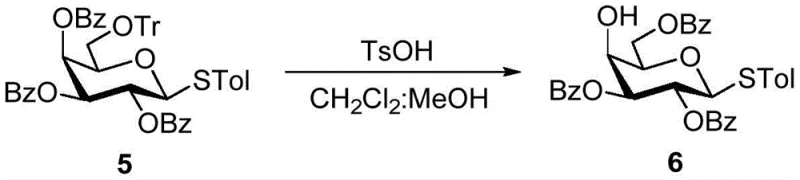
Implementing this sulfonic acid-catalyzed migration strategy in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and selectivity. The process begins with the dissolution of the benzoyl substrate in a dry organic solvent mixture, typically dichloromethane and methanol, ensuring that the concentration is maintained within the optimal range of 0.01 to 0.1 mol/L to prevent intermolecular side reactions. The addition of the sulfonic acid catalyst, such as TsOH, is performed at ambient or slightly cooled temperatures, followed by a stirring period that can range from 1 to 12 hours depending on the steric hindrance of the substrate. Monitoring the reaction progress is essential to determine the exact endpoint, after which the mixture is carefully neutralized to prevent over-acidification during workup. The detailed standardized synthesis steps, including specific stoichiometric ratios and purification protocols derived from the patent examples, are outlined below to guide technical teams in replicating this high-efficiency transformation.
- Dissolve the benzoyl compound substrate (such as protected sugars or non-sugar diols) in a mixed organic solvent system, typically dichloromethane and methanol, maintaining a substrate concentration between 0.01 and 0.1 mol/L.
- Add a sulfonic acid reagent, such as p-toluenesulfonic acid (TsOH), in a mass ratio of 1: 0.5 to 1:2.0 relative to the substrate, and stir the mixture at mild temperatures between 0°C and 30°C for 1 to 12 hours.
- Upon completion, neutralize the reaction mixture with triethylamine to weak alkalinity, wash with saturated sodium bicarbonate and brine, dry over anhydrous sodium sulfate, and purify the resulting migrated product via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this sulfonic acid-catalyzed technology translates into tangible strategic benefits that extend beyond mere chemical curiosity. The primary advantage stems from the drastic simplification of the supply chain for reagents; replacing expensive and supply-constrained transition metal catalysts like silver or zinc salts with commodity chemicals like p-toluenesulfonic acid significantly de-risks the sourcing process. This shift not only stabilizes the cost of goods sold (COGS) but also mitigates the volatility associated with the mining and refining of precious metals. Furthermore, the elimination of heavy metals from the process flow removes the need for specialized scavenging resins or complex aqueous extraction protocols designed to meet strict residual metal limits, thereby reducing the consumption of auxiliary materials and lowering waste disposal costs. The overall process intensification achieved through this method supports a leaner manufacturing model that is more resilient to market fluctuations.
- Cost Reduction in Manufacturing: The economic impact of switching to this catalytic system is profound, primarily driven by the replacement of high-cost noble metal catalysts with inexpensive sulfonic acids. By eliminating the need for silver or zinc reagents, the direct material cost per kilogram of product is significantly lowered. Additionally, the mild reaction conditions (0-30°C) reduce energy consumption for heating or cryogenic cooling, contributing to lower utility overheads. The high regioselectivity minimizes the formation of isomeric impurities, which reduces the burden on downstream purification units and improves the overall mass balance of the process. These factors combine to deliver substantial cost savings without compromising the quality of the final active pharmaceutical ingredient precursor.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals such as toluenesulfonic acid and common solvents like dichloromethane ensures a robust and uninterrupted supply chain. Unlike specialized catalysts that may have single-source suppliers or long lead times, the reagents for this process are globally available in bulk quantities. This availability enhances the agility of the manufacturing schedule, allowing for rapid scale-up from gram to tonne levels without waiting for critical reagent shipments. The simplicity of the workup procedure, involving standard aqueous washes and silica gel chromatography, further ensures that production timelines are not delayed by complex isolation steps, thereby reducing lead time for high-purity intermediates.
- Scalability and Environmental Compliance: The scalability of this method is supported by its operation under mild thermal conditions, which simplifies heat management in large-scale reactors and reduces the risk of thermal runaway incidents. The atom-economical nature of the intramolecular migration means that fewer byproducts are generated, aligning with green chemistry principles and reducing the volume of hazardous waste requiring treatment. The absence of toxic heavy metals in the waste stream simplifies effluent treatment and ensures compliance with increasingly stringent environmental regulations regarding metal discharge. This environmental compatibility makes the process highly attractive for long-term commercial production in regulated markets.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this technology into their existing pipelines, we have compiled a set of frequently asked questions based on the specific technical details and beneficial effects outlined in the patent documentation. These inquiries address the fundamental mechanistic questions, the scope of substrate compatibility, and the practical operational parameters required for successful implementation. Understanding these aspects is crucial for R&D teams planning to adapt this methodology for the synthesis of novel drug candidates or for process optimization of existing commercial routes. The answers provided below reflect the empirical data and theoretical framework established by the inventors.
Q: What is the mechanistic pathway for this acyl migration?
A: The reaction proceeds through the formation of a cyclic orthoester intermediate (either 5-membered or 6-membered ring) facilitated by the sulfonic acid catalyst. This intermediate subsequently undergoes hydrolysis under acidic conditions to complete the highly regioselective migration of the benzoyl group.
Q: Does this method support both 1,2 and 1,3 migrations?
A: Yes, the technology demonstrates versatility by promoting both 1,2-acyl migration and 1,3-acyl transfer reactions. It effectively migrates benzoyl groups from secondary or tertiary hydroxyls to primary hydroxyls, or from axial to equatorial positions in carbohydrate scaffolds.
Q: What are the typical reaction conditions required?
A: The process operates under exceptionally mild conditions, typically requiring temperatures between 0°C and 30°C. Common catalysts include p-toluenesulfonic acid or methanesulfonic acid, used in solvents like dichloromethane mixed with small amounts of methanol or ethanol.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzoyl Migration Intermediates Supplier
As the pharmaceutical industry continues to demand more efficient and sustainable synthetic routes, the ability to execute complex molecular rearrangements with precision becomes a key differentiator for CDMO partners. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at translating laboratory innovations, such as the sulfonic acid-catalyzed acyl migration described here, into robust industrial processes that meet stringent purity specifications. We utilize rigorous QC labs to ensure that every batch of intermediate delivered adheres to the highest standards of quality, providing our clients with the confidence needed to advance their drug development programs without supply chain interruptions.
We invite global partners to engage with our technical procurement team to discuss how this advanced catalytic technology can be tailored to your specific project needs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that evaluates the potential economic benefits of adopting this route for your target molecules. We encourage you to request specific COA data and route feasibility assessments to verify the compatibility of this method with your current portfolio. Let us help you optimize your synthesis strategy, reduce costs, and accelerate your time to market with our reliable supply of high-quality chemical intermediates.
