Scalable Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles: A Cost-Effective Route for High-Purity Pharmaceutical Intermediates
Scalable Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles: A Cost-Effective Route for High-Purity Pharmaceutical Intermediates
The landscape of modern pharmaceutical synthesis is increasingly defined by the demand for robust, scalable, and cost-efficient routes to complex heterocyclic scaffolds. Among these, the 1,2,4-triazole ring system stands out as a privileged structure, ubiquitous in bioactive molecules ranging from antifungal agents to HIV entry inhibitors. A recent technological breakthrough, detailed in patent CN111978265B, introduces a highly efficient preparation method for 5-trifluoromethyl substituted 1,2,4-triazole derivatives. This innovation addresses critical bottlenecks in traditional synthetic pathways by utilizing a ferric chloride-promoted cyclization strategy that operates under mild, aerobic conditions. The significance of this development cannot be overstated for R&D teams seeking to optimize impurity profiles and procurement managers aiming for reliable supply chains. By leveraging cheap and readily available starting materials such as acyl hydrazides and trifluoroethylimidoyl chlorides, this method eliminates the need for expensive transition metals or苛刻 (harsh) reaction environments.
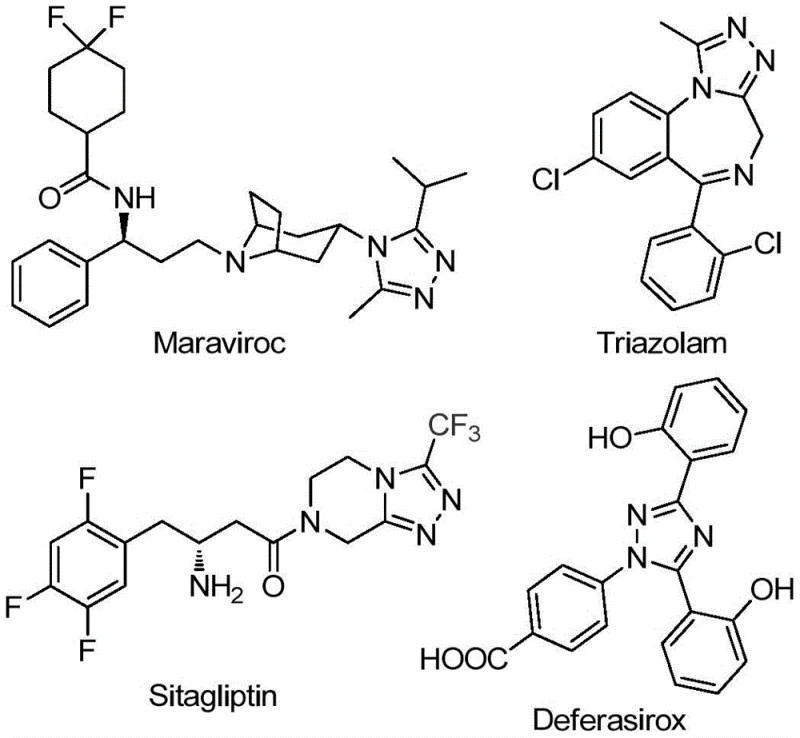
The commercial relevance of this chemistry is underscored by the presence of the 1,2,4-triazole motif in major blockbuster drugs. As illustrated in the structural overview, compounds like Maraviroc, Triazolam, Sitagliptin, and Deferasirox all rely on this nitrogen-containing five-membered heterocycle for their biological activity. Furthermore, the introduction of a trifluoromethyl group at the 5-position is known to significantly enhance metabolic stability, lipophilicity, and bioavailability, making these derivatives highly desirable candidates for drug discovery pipelines. This patent provides a direct pathway to access these high-value intermediates with superior atom economy and operational simplicity, positioning it as a cornerstone technology for the next generation of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has been plagued by significant technical hurdles that hinder large-scale production. Traditional literature methods often rely on the condensation of 3,5-bis(trifluoromethyl)-1,3,4-oxadiazoles with primary amines or the cyclization of trifluoromethyl hydrazides with amidines. These legacy processes are frequently characterized by harsh reaction conditions, requiring extreme temperatures or pressures that pose safety risks in a manufacturing setting. Moreover, many established routes suffer from narrow substrate scopes, failing to accommodate alkyl hydrazones effectively, which limits the chemical diversity accessible to medicinal chemists. Perhaps most critically for supply chain stability, these older methods often exhibit low reaction yields and generate complex impurity profiles that necessitate costly and time-consuming purification steps. The reliance on multi-step sequences further exacerbates the issue, leading to increased waste generation and higher overall production costs, making them less attractive for cost reduction in API manufacturing.
The Novel Approach
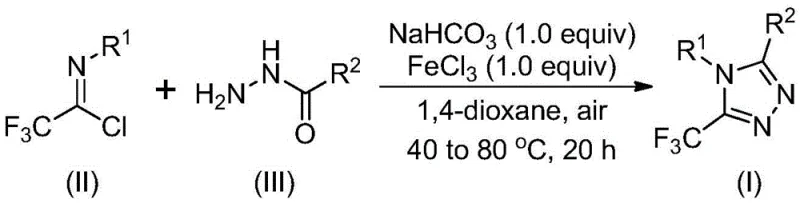
In stark contrast, the methodology disclosed in CN111978265B offers a streamlined, one-pot solution that circumvents these historical limitations. The novel approach utilizes a tandem sequence initiated by a base-promoted intermolecular carbon-nitrogen bond formation, followed by a Lewis acid-catalyzed intramolecular dehydration condensation. This strategy allows for the direct coupling of trifluoroethylimidoyl chlorides with various hydrazides to form the target triazole core efficiently. A defining feature of this new route is its operational robustness; it proceeds smoothly in common aprotic solvents like 1,4-dioxane without the stringent requirement for anhydrous or oxygen-free conditions. This tolerance to air and moisture dramatically simplifies the engineering controls needed for production, reducing capital expenditure on specialized equipment. Furthermore, the method demonstrates exceptional versatility, successfully synthesizing derivatives with diverse substituents at the 3 and 4 positions, including alkyl, alkenyl, and various substituted aryl groups, thereby expanding the chemical space available for drug design.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
Understanding the mechanistic underpinnings of this transformation is vital for R&D directors focused on process optimization and impurity control. The reaction mechanism is proposed to proceed through a distinct two-stage pathway that maximizes efficiency while minimizing side reactions. Initially, the presence of sodium bicarbonate facilitates a nucleophilic attack by the hydrazide nitrogen on the imidoyl chloride carbon, resulting in the formation of a trifluoroacetamidine intermediate. This step is crucial as it establishes the necessary carbon-nitrogen framework prior to ring closure. Subsequently, the addition of ferric chloride acts as a potent Lewis acid promoter, activating the intermediate for cyclization. The iron center coordinates with the nitrogen atoms, lowering the activation energy for the intramolecular dehydration step that最终 (finally) closes the triazole ring. This specific catalytic role of FeCl3 is pivotal, as it drives the equilibrium towards the product without generating excessive thermal stress that could degrade sensitive functional groups.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or high-temperature thermal cyclizations. The mild nature of the Lewis acid promotion ensures that sensitive substituents, such as halogens or electron-withdrawing groups on the aromatic rings, remain intact throughout the process. The patent data indicates that the reaction tolerates a wide range of electronic environments on both the R1 and R2 moieties, suggesting a stable catalytic cycle that is not easily poisoned by substrate variations. For quality assurance teams, this translates to a cleaner crude reaction profile, reducing the burden on downstream purification units. The ability to run the reaction in air also suggests that the active catalytic species is stable against oxidation, preventing the formation of iron-oxide precipitates that could complicate filtration. This mechanistic stability is a key factor in ensuring batch-to-batch consistency, a critical metric for any reliable pharmaceutical intermediate supplier.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Derivatives Efficiently
The practical implementation of this synthesis is designed for ease of execution in both laboratory and pilot plant settings. The protocol involves a straightforward addition of reagents followed by a controlled temperature ramp, eliminating the need for complex addition funnels or cryogenic cooling. The use of sodium bicarbonate as a mild base ensures that acid-sensitive byproducts are minimized, while the choice of 1,4-dioxane as the solvent provides excellent solubility for both organic substrates and inorganic salts. Detailed standard operating procedures regarding stoichiometry, specifically the molar ratios of imidoyl chloride to hydrazide and catalyst loading, are critical for maximizing yield. For a comprehensive guide on executing this transformation with optimal parameters, please refer to the standardized synthesis steps outlined below.
- Mix trifluoroethylimidoyl chloride, hydrazide, and sodium bicarbonate in 1,4-dioxane at 30-50°C for 8-16 hours to form the intermediate.
- Add ferric chloride (FeCl3) catalyst to the reaction mixture and heat to 70-90°C for 6-10 hours under air atmosphere.
- Filter the reaction mixture, purify via silica gel column chromatography to isolate the high-purity 5-trifluoromethyl-1,2,4-triazole derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift towards this FeCl3-catalyzed methodology represents a strategic opportunity to enhance margin and security of supply. The economic benefits are derived not just from the yield, but from the fundamental simplification of the process infrastructure. By removing the dependency on inert gas lines and glovebox operations, facilities can repurpose existing reactors for higher throughput without significant retrofitting costs. This flexibility is essential for maintaining agility in a volatile market where demand for specific API intermediates can fluctuate rapidly. Furthermore, the use of commodity chemicals like ferric chloride and sodium bicarbonate insulates the production cost from the volatility associated with precious metal catalysts like palladium or rhodium. This stability in raw material pricing allows for more accurate long-term forecasting and contract negotiation, directly contributing to cost reduction in fine chemical manufacturing.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts and the removal of strict anhydrous requirements lead to substantial operational savings. The process utilizes inexpensive iron salts which are globally available in bulk quantities, drastically reducing the bill of materials. Additionally, the simplified workup procedure, which involves basic filtration and standard chromatography, reduces solvent consumption and labor hours compared to multi-step legacy routes. These factors combine to lower the overall cost of goods sold (COGS), making the final intermediates more competitive in the global marketplace.
- Enhanced Supply Chain Reliability: The starting materials, specifically acyl chlorides and hydrazides, are widely produced commodities with established supply chains, reducing the risk of raw material shortages. The robustness of the reaction conditions means that production is less susceptible to delays caused by equipment maintenance related to inert atmosphere systems. This reliability ensures consistent delivery schedules for downstream customers, fostering stronger partnerships and reducing the need for safety stock inventory. The ability to scale this reaction from gram to kilogram levels without re-optimization further secures the supply continuity for clinical and commercial phases.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste compared to traditional methods that often require stoichiometric amounts of toxic reagents. The use of iron, a benign metal, simplifies wastewater treatment and aligns with increasingly stringent environmental regulations. The high atom economy of the cyclization step ensures that the majority of the input mass is converted into the desired product, minimizing the volume of organic waste requiring disposal. This environmental compatibility facilitates easier permitting for scale-up and supports corporate sustainability goals, which are becoming a key criterion for vendor selection in the pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of adopting this technology. Understanding these details is crucial for technical teams evaluating the fit of this chemistry within their existing manufacturing portfolios.
Q: Does this synthesis require strict anhydrous or anaerobic conditions?
A: No, a key advantage of this patented method (CN111978265B) is that it operates effectively under air atmosphere without the need for rigorous anhydrous or oxygen-free environments, significantly simplifying operational requirements.
Q: What is the substrate scope for the R1 and R2 groups in this triazole synthesis?
A: The method demonstrates broad tolerance, accommodating substituted or unsubstituted aryl groups for R1 and alkyl, alkenyl, or aryl groups for R2, allowing for the design of diverse molecular architectures.
Q: Why is Ferric Chloride preferred over other metal catalysts for this transformation?
A: Ferric chloride is selected due to its low cost, commercial availability, and high efficiency in promoting the intramolecular dehydration condensation step, making it ideal for large-scale industrial applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chemistry described in CN111978265B for the production of advanced pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facility is equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications required for GMP-grade intermediates. We understand that the integrity of your supply chain depends on consistent quality, and our team is committed to delivering 5-trifluoromethyl-1,2,4-triazole derivatives that meet the highest industry standards for impurity control and physical properties.
We invite you to collaborate with us to leverage this efficient synthetic route for your next drug development program. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. By partnering with us, you gain access to specific COA data and route feasibility assessments that will empower your decision-making process. Contact us today to discuss how we can support your commercial scale-up of complex pharmaceutical intermediates and drive value across your entire supply chain.
