Advanced One-Pot Synthesis of Indolyl-Substituted Indole-3-Ketones for Pharmaceutical Applications
Advanced One-Pot Synthesis of Indolyl-Substituted Indole-3-Ketones for Pharmaceutical Applications
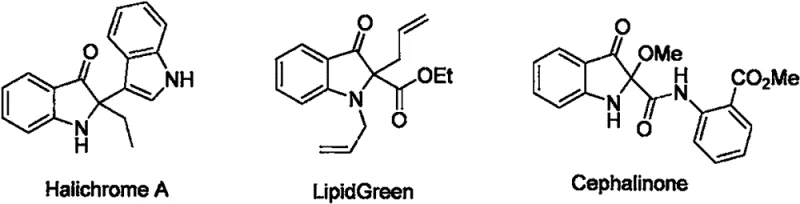
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally sustainable synthetic routes. A significant breakthrough in this domain is documented in patent CN111925312A, which details a novel method for synthesizing indolyl-substituted indole-3-ketones. These compounds represent a crucial class of nitrogen-containing aromatic heterocycles, serving as the core structural motif for numerous natural alkaloids and pharmacologically active agents. As illustrated in the structural diversity of bioactive molecules like Halichrome A and LipidGreen, the 2,2-disubstituted indol-3-one core is a privileged scaffold in medicinal chemistry. However, accessing these complex architectures has historically been challenging due to cumbersome multi-step syntheses. The technology disclosed in this patent offers a transformative solution, utilizing a PPh3-mediated reductive cyclization that streamlines production while maintaining high molecular stability and excellent chemical properties.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of the indolyl-substituted indol-3-one skeleton has been plagued by synthetic inefficiencies that pose significant hurdles for both R&D and commercial supply chains. Conventional pathways often require multiple discrete reaction steps, each necessitating isolation and purification, which drastically reduces overall yield and increases waste generation. Furthermore, many established methods rely on expensive transition metal catalysts or harsh reaction conditions that are difficult to control on a large scale. These factors contribute to extended lead times and inflated production costs, making it difficult for procurement teams to secure reliable supplies of high-purity intermediates. The complexity of these legacy processes also introduces higher risks of impurity formation, complicating the regulatory approval process for final drug substances. Consequently, there has been a persistent industry demand for a more direct, atom-economical approach that can bypass these bottlenecks without compromising on product quality or structural integrity.
The Novel Approach
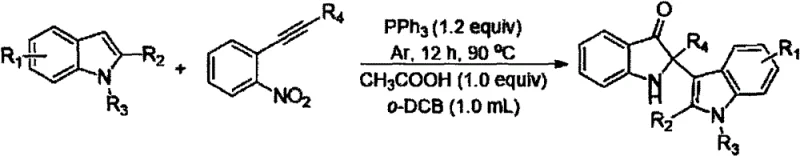
The methodology presented in patent CN111925312A fundamentally reimagines this synthesis through a clever one-pot strategy. By employing triphenylphosphine (PPh3) as a reducing agent under an argon atmosphere, the process directly converts readily available indole compounds and o-nitroalkyne compounds into the desired indolyl-substituted indol-3-ones. This approach eliminates the need for pre-functionalized starting materials or complex pretreatment steps, significantly simplifying the operational workflow. The reaction system is remarkably robust, tolerating a wide range of functional groups on both the indole and alkyne substrates, which allows for the rapid generation of diverse chemical libraries for drug discovery. As depicted in the general reaction scheme below, the transformation proceeds smoothly with just an acid additive and an organic solvent, showcasing a level of simplicity that is rare for such complex heterocyclic formations. This streamlined protocol not only accelerates the research timeline but also lays a solid foundation for cost-effective commercial manufacturing.
Mechanistic Insights into PPh3-Mediated Reductive Cyclization
From a mechanistic perspective, the success of this transformation lies in the dual role of the reagents and the specific reaction conditions employed. The use of PPh3 acts as a mild yet effective oxygen scavenger and reducing agent, facilitating the reduction of the nitro group on the alkyne substrate to a reactive intermediate, likely a nitroso or hydroxylamine species, which then undergoes cyclization with the indole nucleophile. The presence of an acid, such as acetic acid or trifluoroacetic acid, is critical for activating the electrophilic centers and promoting the subsequent ring-closing steps that form the spiro-oxindole or related fused structures. This acid-catalyzed pathway ensures high regioselectivity, minimizing the formation of unwanted isomers that often plague indole functionalization reactions. The choice of o-dichlorobenzene as a solvent provides the necessary thermal stability and solubility profile to sustain the reaction at elevated temperatures (typically 90°C) over a period of 8 to 16 hours, ensuring complete conversion of the starting materials.
Furthermore, the substrate scope analysis reveals exceptional versatility, which is paramount for R&D directors focusing on structure-activity relationship (SAR) studies. The indole component (Formula II) can accommodate various substituents at the R1, R2, and R3 positions, including electron-withdrawing groups like halogens and nitro groups, as well as electron-donating groups like methoxy and alkyl chains. Similarly, the o-nitroalkyne partner (Formula III) accepts diverse R4 groups ranging from cyclopropyl and phenyl to heteroaryl and alkyl halides. This broad compatibility suggests that the reaction mechanism is robust against steric and electronic variations, allowing chemists to access a vast array of analogues from a single standardized protocol. The ability to introduce functional handles like esters, aldehydes, and cyano groups directly onto the scaffold without protecting group manipulation further enhances the utility of this method for downstream derivatization in medicinal chemistry programs.
How to Synthesize Indolyl-Substituted Indole-3-Ketones Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and purity. The process begins with the precise mixing of the indole substrate, the o-nitroalkyne coupling partner, the acid promoter, and the phosphine reducing agent in the chosen organic solvent. Maintaining an inert atmosphere is crucial to prevent the oxidation of sensitive intermediates and the phosphine reagent. The reaction is then heated to the optimal temperature range, typically around 90°C, and stirred for a duration sufficient to drive the conversion to completion, usually between 8 to 16 hours depending on the specific substrate electronics. Following the reaction, standard workup and purification techniques, such as column chromatography or recrystallization, are employed to isolate the final product. For detailed operational specifics and stoichiometric ratios tailored to different substrates, please refer to the standardized synthesis guide below.
- Mix indole compounds, acid (e.g., acetic acid), triphenylphosphine (PPh3), and organic solvent (o-dichlorobenzene) in a reaction vessel.
- Evacuate the vessel with argon three times and heat the mixture to 50-120°C (optimally 90°C) for 8-16 hours to facilitate the reaction between indole and o-nitroalkyne compounds.
- Upon completion, purify the reaction mixture to isolate the target indolyl-substituted indol-3-one with high conversion rates.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials. Since the method utilizes commodity chemicals like indoles, nitro-alkynes, and triphenylphosphine, sourcing is straightforward and less susceptible to the volatility associated with specialized catalysts. This reliability translates directly into enhanced supply chain security, ensuring consistent availability of critical intermediates for downstream API production. Moreover, the elimination of transition metal catalysts removes the need for expensive and time-consuming metal scavenging steps, which are often required to meet stringent regulatory limits on residual metals in pharmaceutical products. This not only reduces the cost of goods sold (COGS) but also shortens the overall production cycle time.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound due to the removal of costly catalytic systems and the reduction of unit operations. By avoiding precious metals like palladium or rhodium, the direct material costs are significantly lowered. Additionally, the one-pot nature of the reaction minimizes solvent usage and waste disposal costs, contributing to a leaner manufacturing footprint. The high conversion rates observed across various examples mean that less starting material is wasted, further optimizing the cost efficiency of the process. These factors combine to deliver substantial cost savings in pharmaceutical intermediate manufacturing, allowing for more competitive pricing in the global market.
- Enhanced Supply Chain Reliability: The reliance on widely available, non-hazardous reagents mitigates the risk of supply disruptions. Unlike specialized catalysts that may have long lead times or single-source dependencies, the key components of this reaction are bulk chemicals with stable global supply networks. This robustness ensures that production schedules can be maintained without unexpected delays. Furthermore, the mild reaction conditions reduce the stress on reactor equipment, lowering maintenance requirements and extending the lifespan of manufacturing assets. This operational stability is critical for maintaining continuous supply lines to major pharmaceutical clients who demand just-in-time delivery of high-quality intermediates.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this process aligns perfectly with modern green chemistry principles. The absence of heavy metals simplifies wastewater treatment and reduces the environmental burden of the manufacturing facility. The high atom economy and reduced solvent intensity make it easier to scale the reaction from gram-scale laboratory batches to multi-ton commercial production without encountering the heat transfer or mixing issues common in complex multi-step syntheses. This ease of scale-up ensures that the technology can rapidly transition from R&D to commercial reality, supporting the fast-paced demands of the pharmaceutical industry while adhering to increasingly strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived from the detailed experimental data and beneficial effects described in the patent documentation. Understanding these aspects helps stakeholders evaluate the feasibility of integrating this technology into their existing workflows. The answers reflect the specific advantages of the PPh3-mediated route, focusing on its practical application in producing high-value heterocyclic compounds. For more specific technical data or custom synthesis requests, our team is available to provide detailed consultations.
Q: What are the key advantages of this PPh3-mediated synthesis over traditional methods?
A: This method eliminates the need for expensive transition metal catalysts and complex multi-step sequences. It utilizes cheap, readily available raw materials like indoles and o-nitroalkynes, operating under mild conditions (90°C) with a simple one-pot procedure, significantly reducing production costs and environmental impact.
Q: What is the substrate scope for this reaction?
A: The reaction demonstrates excellent tolerance for various functional groups. Indole substrates can bear substituents such as halogens, alkyls, methoxy, cyano, nitro, ester, and aldehyde groups. Similarly, the o-nitroalkyne component accepts cyclopropyl, phenyl, heteroaryl, and alkyl groups, allowing for the synthesis of a diverse library of bioactive scaffolds.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is highly scalable. It requires minimal equipment, uses stable reagents, and avoids hazardous pretreatment steps. The high conversion rates (up to 95% in optimized examples) and simple purification workflow make it ideal for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolyl-Substituted Indole-3-Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodologies described in patent CN111925312A for the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from the bench to the market. Our state-of-the-art facilities are equipped to handle the specific requirements of this PPh3-mediated chemistry, including inert atmosphere handling and high-temperature reactions, while our rigorous QC labs enforce stringent purity specifications to guarantee the quality of every batch. We are committed to delivering high-purity indole derivatives that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced technology for your drug development programs. Our technical procurement team is ready to assist you with a Customized Cost-Saving Analysis to quantify the economic benefits of switching to this efficient route for your specific targets. Please contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis projects. Together, we can accelerate the delivery of life-saving medicines to patients worldwide through superior chemical innovation and reliable supply chain partnerships.
