Revolutionizing N-Aryl Indole Production via Mild Copper Catalysis for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways for constructing nitrogen-containing heterocycles, which serve as critical scaffolds in drug discovery. Patent CN113072478B introduces a groundbreaking preparation method for N-aryl indoles under copper catalysis, addressing long-standing challenges in C-N bond formation. This technology leverages a mild oxidative cross-coupling strategy between indoles and aryl silanes, utilizing molecular oxygen as the terminal oxidant. By operating at a remarkably low temperature of 45°C in DMSO, this process eliminates the need for harsh bases, expensive ligands, or stoichiometric metal oxidants that typically plague traditional synthesis routes. For R&D directors and process chemists, this represents a significant leap forward in atom economy and operational simplicity, offering a robust platform for generating diverse libraries of bioactive molecules with high purity and minimal environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of N-aryl indole motifs has relied heavily on classical Ullmann-type coupling reactions or palladium-catalyzed Buchwald-Hartwig aminations. The traditional Ullmann reaction, while conceptually simple, suffers from severe practical limitations, often requiring excessive temperatures reaching up to 200°C and the use of strong, corrosive bases. These harsh conditions frequently lead to poor selectivity, decomposition of sensitive functional groups, and the generation of complex impurity profiles that are difficult to remove. Furthermore, the requirement for stoichiometric amounts of copper or specialized ligands increases both the material cost and the burden on downstream purification processes. For procurement managers, these inefficiencies translate into higher production costs and longer lead times, while supply chain heads face risks associated with the handling of hazardous reagents and the disposal of heavy metal waste streams.
The Novel Approach
In stark contrast, the methodology disclosed in CN113072478B offers a transformative solution by employing aryl silanes as coupling partners in the presence of a catalytic amount of copper acetate. This novel approach operates under exceptionally mild conditions, specifically at 45°C under an oxygen atmosphere, which dramatically enhances the tolerance for diverse functional groups. As illustrated in the substrate scope below, the reaction successfully accommodates electron-rich and electron-deficient substituents, including halogens, esters, and nitro groups, without compromising yield or selectivity. 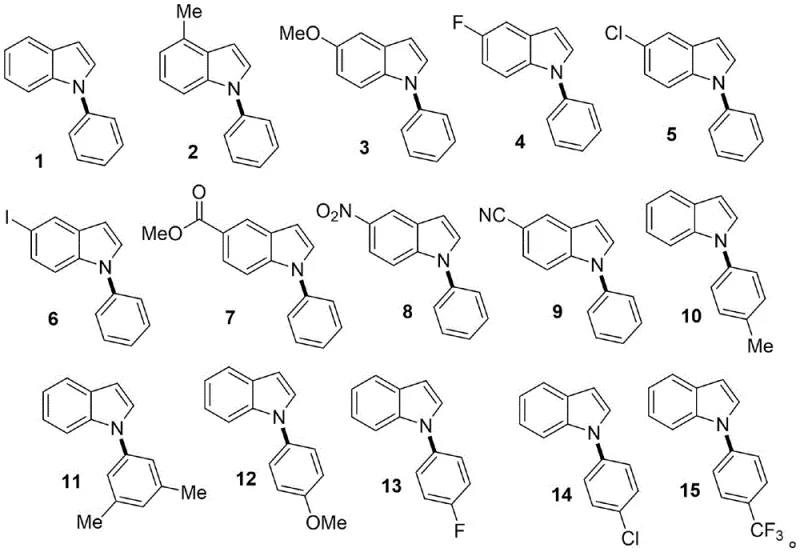 This broad compatibility allows for the direct synthesis of complex intermediates without the need for extensive protecting group strategies. The elimination of external ligands and metal oxidants further simplifies the reaction mixture, making the workup procedure straightforward and reducing the overall environmental footprint of the manufacturing process.
This broad compatibility allows for the direct synthesis of complex intermediates without the need for extensive protecting group strategies. The elimination of external ligands and metal oxidants further simplifies the reaction mixture, making the workup procedure straightforward and reducing the overall environmental footprint of the manufacturing process.
Mechanistic Insights into Copper-Catalyzed N-Arylation
The core of this innovation lies in the unique catalytic cycle facilitated by the copper-TBAF-oxygen system. The reaction initiates with the activation of the aryl silane by the fluoride source, TBAF, which generates a hypervalent silicon species capable of transmetallation with the copper center. Unlike traditional cross-couplings that require pre-functionalized aryl halides, this silane-based activation pathway avoids the formation of halide waste. The copper catalyst, likely cycling between Cu(I) and Cu(II) oxidation states, mediates the formation of the C-N bond through a concerted metallation-deprotonation or oxidative addition-reductive elimination sequence. Molecular oxygen serves as a green, abundant oxidant to regenerate the active copper species, ensuring the catalytic turnover continues efficiently without the accumulation of reduced metal byproducts. This mechanistic elegance ensures high conversion rates even at low catalyst loadings, providing a reliable route for high-purity pharmaceutical intermediate production.
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in maintaining product integrity. High-temperature processes often induce side reactions such as polymerization or rearrangement of the indole core, leading to difficult-to-separate impurities. By maintaining the reaction at 45°C, the thermal energy is insufficient to drive these degradation pathways, resulting in a cleaner crude reaction profile. Additionally, the use of DMSO as a polar aprotic solvent facilitates the solubility of both organic substrates and inorganic salts, promoting homogeneous reaction kinetics. The absence of strong bases prevents base-mediated hydrolysis of sensitive ester or nitrile groups often present in advanced intermediates. Consequently, the final product requires less aggressive purification, preserving yield and reducing solvent consumption during the isolation phase, which is a critical factor for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize N-Aryl Indole Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it highly suitable for both laboratory optimization and industrial manufacturing. The process begins with the precise selection of reagents, where indole and aryl triethoxysilane are combined with copper(II) acetate and tetrabutylammonium fluoride in dimethyl sulfoxide. The reaction mixture is then subjected to heating under an oxygen atmosphere, allowing the cross-coupling to proceed to completion over a 24-hour period. Following the reaction, a standard aqueous workup involving extraction with ethyl acetate and drying over sodium sulfate yields the crude product, which is subsequently purified via silica gel chromatography. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps outlined below.
- Combine indole, aryl triethoxysilane, Cu(OAc)2 catalyst, and TBAF in DMSO solvent within a reaction vessel.
- Heat the mixture to 45°C under an oxygen atmosphere and maintain stirring for 24 hours to ensure complete conversion.
- Cool the reaction, dilute with water, extract with ethyl acetate, dry over sodium sulfate, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this copper-catalyzed technology offers substantial strategic benefits beyond mere technical feasibility. The shift from precious metal catalysts to abundant copper salts directly impacts the bill of materials, driving down raw material costs significantly. Moreover, the mild reaction conditions reduce energy consumption associated with heating and cooling cycles, contributing to lower utility costs and a smaller carbon footprint. The robustness of the process against varying substrate electronics ensures consistent batch-to-batch quality, minimizing the risk of production delays caused by failed runs or off-spec material. This reliability is crucial for maintaining continuous supply chains in the fast-paced pharmaceutical sector, where downtime can have cascading effects on downstream drug development timelines.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts and specialized ligands with inexpensive copper acetate results in drastic cost savings on catalyst procurement. Furthermore, the elimination of stoichiometric metal oxidants and strong bases reduces the volume of chemical waste generated, lowering disposal costs and simplifying regulatory compliance. The high atom economy of using aryl silanes also means less raw material is wasted in the form of leaving groups, optimizing the overall material balance and enhancing the economic viability of large-scale production campaigns.
- Enhanced Supply Chain Reliability: Aryl silanes are commercially available and stable reagents, offering a secure supply chain compared to sensitive aryl halides or boronic acids that may suffer from stability issues during storage and transport. The simplified reaction setup, which does not require inert atmosphere gloveboxes or specialized high-pressure equipment, allows for flexible manufacturing in standard facilities. This accessibility reduces the barrier to entry for contract manufacturing organizations and ensures that production capacity can be scaled rapidly to meet market demand without significant capital investment in new infrastructure.
- Scalability and Environmental Compliance: The use of DMSO, a high-boiling polar solvent, facilitates heat management during exothermic events, making the process inherently safer for scale-up. The mild temperature profile minimizes the risk of thermal runaway, a critical safety consideration for kilogram-to-tonne scale operations. Additionally, the reliance on molecular oxygen from air as the oxidant aligns with green chemistry principles, reducing the need for hazardous oxidizing agents. This environmentally friendly profile supports corporate sustainability goals and eases the permitting process for new manufacturing lines in regions with strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the process capabilities and limitations. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production workflows or new drug substance development programs.
Q: What are the key advantages of this copper-catalyzed method over traditional Ullmann reactions?
A: Unlike traditional Ullmann reactions that require harsh conditions such as temperatures up to 200°C and strong bases, this method operates at a mild 45°C without additional ligands or metal oxidants, significantly improving functional group tolerance and safety.
Q: Does this process require expensive palladium catalysts?
A: No, this protocol utilizes inexpensive copper salts like Cu(OAc)2 as the catalyst, which drastically reduces raw material costs compared to precious metal-catalyzed cross-coupling reactions while maintaining high efficiency.
Q: What is the substrate scope for this N-arylation reaction?
A: The method demonstrates excellent substrate scope, tolerating various functional groups including electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as nitro, cyano, and halogens on both the indole and aryl silane components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Aryl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the development of next-generation therapeutics. Our team of expert process chemists has thoroughly analyzed the potential of this copper-catalyzed N-arylation technology and is fully equipped to implement it for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are designed to handle sensitive catalytic reactions with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this advanced chemistry for your pipeline. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating exactly how this method can optimize your budget. We encourage you to reach out today to request specific COA data for similar N-aryl indole derivatives and to discuss route feasibility assessments that could accelerate your project timelines while reducing overall production costs.
