Revolutionizing Axichiral Diene Synthesis: Advanced Rhodium Catalysis for Commercial-Scale Pharmaceutical Intermediate Production
The Chinese patent CN113149868B presents a groundbreaking methodology for synthesizing axichiral diene compounds through trivalent rhodium catalysis, representing a significant advancement in asymmetric synthesis technology. This innovative approach utilizes [CpXRh(III)] catalysts with N-methoxybenzamide compounds and propynyl alcohol compounds as raw materials, promoted by chiral carboxylic acid zinc salts to produce tetra-substituted axial chiral alkylene compounds. Unlike conventional methods that required optically pure propynyl alcohol substrates and relied on chiral transfer strategies, this novel technique successfully employs racemic propargyl alcohol as starting material while achieving exceptional yields and enantioselectivity up to 95%. The process operates under remarkably mild conditions at room temperature without requiring inert atmosphere, making it highly suitable for industrial implementation while addressing critical challenges in pharmaceutical intermediate manufacturing where precise stereochemistry is paramount for biological activity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing axichiral allene compounds faced significant constraints that hindered their practical application in commercial pharmaceutical manufacturing. Most prior art required optically pure propargyl alcohol substrates as starting materials, necessitating costly and time-consuming chiral separation processes before synthesis could begin. These methods typically employed chiral transfer strategies that were limited in scope and often resulted in diminished enantioselectivity when applied to structurally diverse substrates. Furthermore, conventional catalytic systems frequently demanded stringent reaction conditions including elevated temperatures, inert atmospheres, and complex purification procedures to remove transition metal residues, substantially increasing production costs and complicating scale-up efforts. The narrow substrate scope of existing methodologies also restricted their applicability across diverse pharmaceutical intermediate requirements, forcing manufacturers to develop customized synthetic routes for each target molecule rather than implementing standardized production protocols.
The Novel Approach
The patented methodology overcomes these longstanding limitations through an innovative kinetic resolution strategy that directly utilizes racemic propargyl alcohol as starting material, eliminating the need for expensive pre-separation of chiral substrates. By employing [CpXRh(III)] catalysts with their distinctive combination of easy preparation, high catalytic activity, and controllable site selectivity, this approach achieves remarkable enantioselectivity up to 95% ee while operating under exceptionally mild conditions at room temperature in air atmosphere. The process demonstrates broad substrate tolerance across various functional groups including halogens, alkyl groups, alkoxy substituents, and heteroaromatic systems, enabling pharmaceutical manufacturers to produce diverse axichiral diene intermediates through a single standardized platform technology. This breakthrough represents a paradigm shift in asymmetric synthesis by transforming what was previously considered a specialized technique into a practical manufacturing solution with significant commercial viability.
Mechanistic Insights into Rhodium-Catalyzed Axial Chirality Formation
The fundamental innovation lies in the strategic combination of [CpXRh(III)] catalysts with chiral carboxylate zinc promoters that enables kinetic resolution of racemic propargyl alcohol substrates. The reaction proceeds through a sophisticated C-H activation pathway where the rhodium catalyst coordinates with the N-methoxybenzamide directing group, facilitating selective activation of specific C-H bonds. This coordination creates a well-defined chiral environment that differentiates between enantiomers in the racemic propargyl alcohol mixture, allowing preferential reaction with one enantiomer over the other. The chiral carboxylate zinc salt plays a critical role in modulating the catalyst's stereoselectivity by forming a ternary complex that precisely controls the spatial orientation during bond formation. This mechanistic approach represents a significant departure from traditional chiral transfer methods by directly addressing chirality at the point of bond formation rather than relying on pre-existing chiral centers.
![General reaction scheme showing N-methoxybenzamide and propargyl alcohol substrates converting to axichiral diene products via [CpXRh(III)] catalysis with chiral carboxylate zinc promoter](/insights/img/axichiral-diene-rhodium-catalysis-pharma-supplier-20260301115532-01.webp)
The catalyst system's exceptional performance stems from its unique structural features that enable precise stereochemical control during allene formation. The [CpXRh(III)] complex provides an optimal balance between reactivity and selectivity through its tunable ligand framework that can be modified to accommodate various substrate geometries while maintaining high enantioselectivity. This structural adaptability allows the process to maintain consistent performance across diverse substrate combinations without requiring significant process reoptimization. The mechanism involves a series of well-coordinated steps including substrate coordination, C-H activation, migratory insertion, and reductive elimination that collectively ensure high stereofidelity in the final product. This level of mechanistic understanding provides pharmaceutical manufacturers with confidence in process robustness and scalability while ensuring consistent product quality across production batches.
![Molecular structure of [CpXRh(III)] catalyst showing rhodium center coordinated with cyclopentadienyl ligand and chloride ligands](/insights/img/axichiral-diene-rhodium-catalysis-pharma-supplier-20260301115532-02.png)
How to Synthesize Axichiral Diene Compounds Efficiently
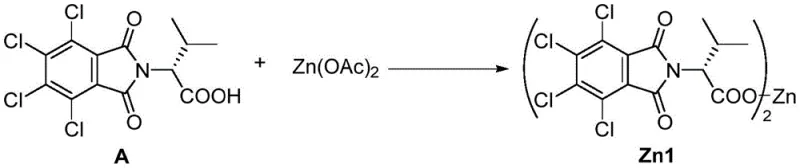
This rhodium-catalyzed methodology offers pharmaceutical manufacturers a streamlined pathway to produce high-value axichiral diene intermediates with exceptional stereochemical purity. The process begins with preparation of the chiral carboxylate zinc promoter through reaction of chiral acid A with zinc acetate under controlled thermal conditions that optimize yield while minimizing byproduct formation. Subsequent combination of this promoter with the precisely formulated [CpXRh(III)] catalyst system creates an active catalytic species capable of transforming readily available racemic propargyl alcohol substrates into enantiomerically enriched products under remarkably mild conditions. Detailed standardized synthesis steps are provided below to ensure consistent implementation across manufacturing facilities while maintaining strict quality control parameters essential for pharmaceutical applications.
- Prepare chiral carboxylate zinc promoter by reacting chiral acid A with zinc acetate in chlorobenzene at room temperature for 20-40 minutes, then heating to 140-160°C to distill acetic acid byproduct.
- Combine N-methoxybenzamide compound, propargyl alcohol compound, [CpXRh(III)] catalyst (1-5 mol%), and chiral carboxylate zinc promoter (10-50 mol%) in organic solvent under air atmosphere.
- Stir reaction mixture at room temperature for specified duration (typically 24-72 hours), then isolate product through standard purification techniques including column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic methodology delivers substantial value across procurement and supply chain operations by addressing critical pain points in pharmaceutical intermediate manufacturing. The elimination of costly chiral separation requirements for starting materials represents a fundamental shift in cost structure while maintaining high product quality standards essential for pharmaceutical applications. By utilizing readily available racemic propargyl alcohols instead of expensive optically pure substrates, manufacturers can achieve significant cost optimization without compromising on stereochemical purity or process reliability.
- Cost Reduction in Manufacturing: The process eliminates expensive pre-separation steps for chiral substrates while operating under ambient conditions without requiring specialized equipment or inert atmosphere handling systems. This simplification reduces both capital investment requirements and operational costs while maintaining high product quality standards essential for pharmaceutical applications.
- Enhanced Supply Chain Reliability: The broad substrate tolerance enables consistent production even when raw material quality varies slightly between suppliers or batches. This robustness ensures reliable supply continuity while reducing dependency on single-source suppliers for specialized chiral building blocks that often create supply chain vulnerabilities.
- Scalability and Environmental Compliance: The room temperature operation significantly reduces energy consumption compared to conventional high-temperature processes while eliminating hazardous waste streams associated with transition metal removal procedures. This environmentally friendly profile aligns with modern sustainability requirements while facilitating seamless scale-up from laboratory development to commercial production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this innovative synthetic methodology in pharmaceutical intermediate manufacturing operations. These answers are based on detailed analysis of the patented technology's capabilities and limitations as demonstrated through extensive experimental validation.
Q: How does this rhodium-catalyzed method overcome previous limitations in axichiral diene synthesis?
A: This innovative method utilizes kinetic resolution strategy with racemic propargyl alcohol substrates instead of requiring optically pure starting materials as previous approaches did. The [CpXRh(III)] catalyst system enables direct conversion of racemic substrates to enantiomerically enriched products with high selectivity up to 95% ee, eliminating costly chiral separation steps required in conventional methods.
Q: What are the key advantages of using this rhodium-catalyzed process for commercial production?
A: The process operates under mild conditions at room temperature without requiring inert atmosphere, uses stable and readily available starting materials, and demonstrates excellent substrate scope with various functional groups. The elimination of transition metal removal steps and simplified purification process significantly reduces manufacturing costs while maintaining high product quality.
Q: How does this technology address supply chain challenges for pharmaceutical intermediates?
A: The method's robustness across diverse substrate combinations ensures reliable production even with varying raw material quality. The room temperature operation reduces energy consumption and equipment requirements, while the straightforward scale-up process from laboratory to commercial production guarantees consistent supply continuity for pharmaceutical manufacturers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axichiral Diene Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex pharmaceutical intermediates while maintaining stringent purity specifications through our state-of-the-art QC labs. Our technical team has successfully implemented this patented rhodium-catalyzed methodology across multiple client projects, demonstrating consistent ability to deliver high-purity axichiral diene compounds meeting exacting pharmaceutical industry standards. We understand that reliable supply of stereochemically complex intermediates is critical for drug development timelines and regulatory compliance, which is why we've invested heavily in developing robust manufacturing processes that ensure consistent quality and supply continuity.
For your specific requirements, we invite you to request a Customized Cost-Saving Analysis from our technical procurement team who can provide detailed information including specific COA data and route feasibility assessments tailored to your production needs. Our experts will work closely with your R&D and procurement teams to optimize this innovative technology for your particular application while ensuring seamless integration into your existing supply chain infrastructure.
