Advancing NSAID Intermediate Production: A Novel Copper-Catalyzed Asymmetric Reduction Strategy
Advancing NSAID Intermediate Production: A Novel Copper-Catalyzed Asymmetric Reduction Strategy
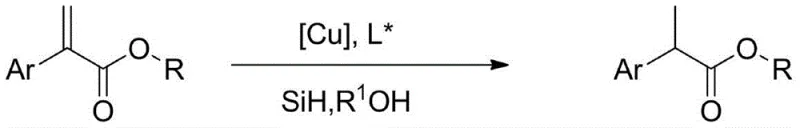
The pharmaceutical industry continuously seeks robust, cost-effective methodologies for synthesizing chiral building blocks, particularly for non-steroidal anti-inflammatory drugs (NSAIDs). Patent CN110903189B introduces a transformative approach to the enantioselective synthesis of chiral 2-aryl propionates, utilizing an earth-abundant copper catalytic system. This technology represents a significant departure from conventional noble metal catalysis, offering a pathway that combines mild reaction conditions with high atom economy. By leveraging copper salts in conjunction with specialized chiral phosphine ligands and silane reducing agents, this method effectively reduces 2-aryl acrylates to their corresponding chiral esters. For global procurement teams and R&D directors, this innovation signals a potential paradigm shift in how key intermediates for drugs like Ibuprofen and Naproxen are manufactured, promising enhanced supply chain resilience and reduced dependency on volatile precious metal markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2-arylpropionic acids has relied on processes that present substantial economic and operational challenges. Traditional routes, such as the Boots method, involve multi-step sequences including Friedel-Crafts acylation and Darzens condensation, which suffer from poor atom economy and generate significant chemical waste. Alternatively, the BHC method, while more efficient, relies on palladium-catalyzed carbonylation requiring high-pressure carbon monoxide, posing severe safety risks and necessitating expensive corrosion-resistant reactor infrastructure. Furthermore, asymmetric hydrogenation strategies employing rhodium or ruthenium complexes, while effective, are constrained by the exorbitant cost of these precious metals and the logistical complexities of handling high-pressure hydrogen gas. These factors collectively inflate the cost of goods sold (COGS) and introduce fragility into the supply chain for critical pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN110903189B utilizes a copper-catalyzed hydrosilylation protocol that operates under remarkably mild conditions, typically ranging from -50°C to 40°C. This process replaces hazardous high-pressure hydrogen with stable silane reagents and substitutes expensive rhodium or palladium catalysts with inexpensive copper salts. The reaction proceeds efficiently in common organic solvents such as toluene or tetrahydrofuran, simplifying the engineering requirements for commercial scale-up of complex NSAID intermediates. By avoiding extreme pressures and temperatures, this novel approach not only enhances operator safety but also drastically reduces energy consumption and capital expenditure on specialized high-pressure equipment, positioning it as a superior alternative for modern green chemistry manufacturing.
Mechanistic Insights into Cu-Catalyzed Asymmetric Hydrosilylation
The core of this technological breakthrough lies in the generation of a reactive copper-hydride species in situ, which serves as the active reducing agent. Upon mixing the copper salt precursor with the silane reductant and a chiral phosphine ligand, a coordinatively unsaturated copper complex is formed. This complex facilitates the migratory insertion of the olefinic double bond of the 2-aryl acrylate into the copper-hydride bond. The chirality of the final product is strictly governed by the steric and electronic properties of the ligand framework, which creates a chiral pocket around the metal center, directing the facial selectivity of the hydride attack. This precise control is essential for achieving the high enantiomeric excess required for high-purity active pharmaceutical ingredients, ensuring that the biologically active (S)-enantiomer is produced preferentially over the inactive or harmful (R)-isomer.
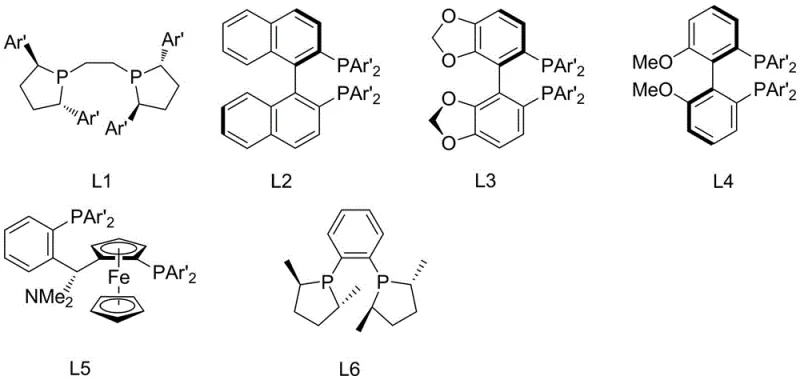
Impurity control is another critical aspect addressed by this catalytic system. The patent specifies careful optimization of the molar ratios between the copper source, the base (such as sodium tert-butoxide), and the ligand to prevent racemization. Excess base can lead to the epimerization of the chiral center, degrading the optical purity of the product. By maintaining a strict stoichiometric balance, typically keeping the ratio of copper salt to base between 2:1 and 1:1, the process minimizes side reactions and ensures a clean impurity profile. The versatility of the ligand library, encompassing structures L1 through L6, allows chemists to fine-tune the electronic environment of the catalyst to accommodate various substrates, from simple phenyl derivatives to complex naphthyl systems found in Naproxen precursors.
How to Synthesize Chiral 2-Aryl Propionate Efficiently
Implementing this synthesis route requires strict adherence to inert atmosphere techniques and precise reagent dosing to maximize catalytic turnover and enantioselectivity. The process begins with the activation of the copper catalyst in a dry, oxygen-free environment, followed by the sequential addition of the silane reducing agent and the alcohol additive, which plays a crucial role in protonating the intermediate silyl enol ether. Detailed standard operating procedures regarding specific molar equivalents, solvent choices, and workup protocols are essential for reproducibility at scale. For a comprehensive breakdown of the experimental parameters and purification steps validated in the patent literature, please refer to the technical guide below.
- Prepare the catalyst system by mixing copper salt (e.g., Cu(OAc)2·H2O) and chiral phosphine ligand (L1-L6) in an inert solvent like toluene or THF under nitrogen.
- Add the silane reducing agent (e.g., PMHS or Ph2SiH2) and alcohol additive (R1OH) to the mixture, followed by the 2-aryl acrylate substrate.
- Maintain the reaction temperature between -50°C and 40°C for 0.25 to 6 hours, then quench with aqueous solution and purify via extraction and column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed technology offers compelling strategic benefits beyond mere technical feasibility. The transition from precious metal catalysts to base metal copper fundamentally alters the cost structure of the synthesis, removing exposure to the volatile pricing of rhodium and palladium markets. Additionally, the use of silanes instead of high-pressure hydrogen gas eliminates the need for specialized high-pressure reactors and the associated safety certifications, thereby lowering the barrier to entry for contract manufacturing organizations (CMOs). This simplification of the process hardware translates directly into cost reduction in API manufacturing, allowing for more competitive pricing models without sacrificing quality or yield.
- Cost Reduction in Manufacturing: The substitution of expensive noble metals with abundant copper salts results in a drastic decrease in raw material costs. Since copper is orders of magnitude cheaper than rhodium or palladium, the catalyst loading cost becomes negligible. Furthermore, the mild reaction conditions reduce energy consumption for heating and cooling, while the simplified workup procedure minimizes solvent usage and waste disposal fees. These cumulative savings significantly improve the gross margin profile for manufacturers producing bulk quantities of chiral intermediates.
- Enhanced Supply Chain Reliability: Relying on earth-abundant metals mitigates the risk of supply disruptions often associated with geopolitically sensitive precious metals. Copper and silane reagents are commodity chemicals with robust, diversified global supply chains, ensuring consistent availability. This reliability is crucial for maintaining continuous production schedules for essential medicines, reducing lead time for high-purity active pharmaceutical ingredients and preventing stockouts that could impact downstream drug formulation and patient access.
- Scalability and Environmental Compliance: The process operates at near-ambient pressures and moderate temperatures, making it inherently safer and easier to scale from kilogram to multi-ton production volumes. The absence of toxic carbon monoxide (used in carbonylation routes) and high-pressure hydrogen aligns with increasingly stringent environmental, health, and safety (EHS) regulations. This green chemistry profile facilitates smoother regulatory approvals and reduces the environmental footprint of the manufacturing facility, supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed asymmetric reduction technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the scope and limitations of the method for potential licensees and manufacturing partners.
Q: What are the primary advantages of this copper-catalyzed method over traditional rhodium or palladium systems?
A: The primary advantage is cost efficiency and safety. Copper is significantly cheaper than precious metals like Rhodium or Palladium. Furthermore, this method utilizes silanes as reducing agents under mild conditions, eliminating the need for high-pressure hydrogen gas equipment required in traditional asymmetric hydrogenation.
Q: Can this synthesis method be applied to the production of active pharmaceutical ingredients like Naproxen?
A: Yes, the patent explicitly demonstrates the synthesis of precursors for major NSAIDs. Specifically, the method produces chiral 2-aryl propionates which, upon simple hydrolysis, yield active ingredients such as (S)-(+)-Ibuprofen and (S)-(+)-Naproxen with high enantioselectivity.
Q: How does the choice of ligand affect the enantiomeric excess (ee) in this reaction?
A: The chiral environment is dictated by the phosphine ligand structure (L1-L6). The patent data indicates that bulky substituents on the ligand, such as tert-butyl groups in ligand L4, significantly enhance stereocontrol, leading to higher ee values compared to simpler ligand structures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 2-Aryl Propionate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the copper-catalyzed synthesis route described in CN110903189B for the production of high-value chiral intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped to handle air-sensitive catalytic reactions under inert atmospheres, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required for pharmaceutical applications.
We invite global pharmaceutical companies and chemical distributors to collaborate with us to leverage this cost-effective technology for your supply chain. By partnering with our technical team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of technical confidence and commercial viability.
