Advanced Chiral Monophosphine Ligand Yu-Phos: Scalable Synthesis and Catalytic Applications
The landscape of asymmetric catalysis has been fundamentally transformed by the development of specialized chiral ligands, as detailed in patent CN109851634B. This intellectual property introduces a novel class of chiral monophosphine ligands, collectively designated as Yu-Phos, which represent a significant leap forward in stereochemical control. Unlike traditional ligands that rely solely on carbon-centered chirality, Yu-Phos incorporates a sophisticated dual-chiral architecture featuring both carbon and sulfur stereocenters. This structural innovation allows for precise tuning of the steric and electronic environment around the metal center, thereby enhancing catalytic performance in complex organic transformations. The patent discloses the preparation of all four possible stereoisomers (1(S,Rs), 1(R,Rs), 1(S,Ss), and 1(R,Ss)), providing chemists with a comprehensive toolkit for optimizing enantioselectivity in diverse synthetic pathways. As a reliable chiral ligands supplier, understanding the depth of this technology is crucial for advancing high-value synthesis.
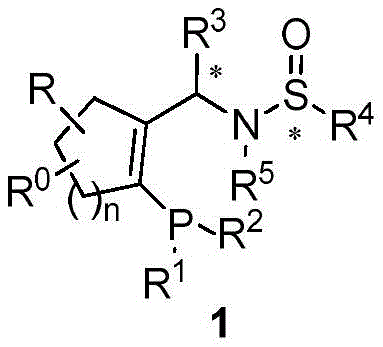
The structural versatility of Yu-Phos is defined by its variable substituents, where R groups can range from simple alkyl chains to complex aryl systems, allowing for fine-tuning to specific substrate requirements. The presence of the sulfinamide moiety introduces a robust sulfur-based chiral element that works synergistically with the carbon backbone. This dual-chirality approach addresses a long-standing challenge in the field: achieving 100% selectivity and yield without by-products. By enabling the efficient conversion of racemic compounds to optically pure products using only catalytic amounts of the ligand, this technology aligns perfectly with the modern demands for economical, safe, and environmentally friendly chemistry. The ability to access the full configuration space means that process chemists can empirically determine the optimal match between ligand geometry and substrate topology, significantly de-risking the development of new asymmetric processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of high-performance chiral phosphine ligands has been plagued by significant bottlenecks that hinder widespread industrial adoption. Conventional methods often rely on the resolution of racemic mixtures, a process that is inherently inefficient as it theoretically caps the maximum yield at 50% unless dynamic kinetic resolution is employed. Furthermore, traditional synthetic routes frequently involve lengthy multi-step sequences requiring expensive chiral pool starting materials or hazardous reagents that pose safety risks on a large scale. The toxicity of certain phosphorus halides and the sensitivity of intermediates to air and moisture often necessitate rigorous exclusion protocols, driving up operational costs and complicating waste management. Additionally, achieving high enantiomeric purity often requires repeated recrystallization or chromatographic purification, which generates substantial solvent waste and reduces the overall process mass intensity. These factors collectively contribute to high production costs and supply chain vulnerabilities, making many advanced ligands inaccessible for routine commercial manufacturing.
The Novel Approach
The methodology disclosed in CN109851634B offers a paradigm shift by streamlining the synthesis of Yu-Phos into a clear, operationally simple, and high-yielding process. The novel approach circumvents the need for difficult chiral resolutions by utilizing commercially available chiral sulfinamides as the source of sulfur chirality, which are then coupled with a readily accessible phosphine-containing aldehyde intermediate. This convergent strategy allows for the modular assembly of the ligand, where the carbon chirality is introduced via stereoselective organometallic addition. The patent reports impressive overall yields ranging from 71% to 88%, which is a substantial improvement over many literature precedents. By avoiding toxic reagents and minimizing the number of purification steps, this route significantly reduces the environmental footprint and enhances the safety profile of the manufacturing process. The robustness of the synthesis ensures consistent quality and batch-to-batch reproducibility, which are critical parameters for establishing a stable supply chain for critical catalytic materials.
Mechanistic Insights into Dual-Chiral Center Induction
The exceptional performance of Yu-Phos stems from its unique ability to create a highly defined chiral pocket around the transition metal center. The ligand design integrates a rigid cyclic backbone with a flexible sulfinamide arm, creating a three-dimensional environment that effectively differentiates between the enantiotopic faces of the substrate. During catalysis, the phosphorus atom coordinates strongly with the metal (such as Gold), while the sulfinamide oxygen or nitrogen may participate in secondary interactions that further lock the conformation. This cooperative binding mode ensures that the substrate approaches the reactive center from a specific trajectory, leading to high stereoselectivity. The presence of both carbon and sulfur chiral centers allows for a "match-mismatch" effect, where the relative configuration of the two centers can be tuned to maximize the energy difference between the diastereomeric transition states. This mechanistic feature is particularly valuable in reactions where subtle changes in ligand structure can lead to dramatic reversals in enantioselectivity.
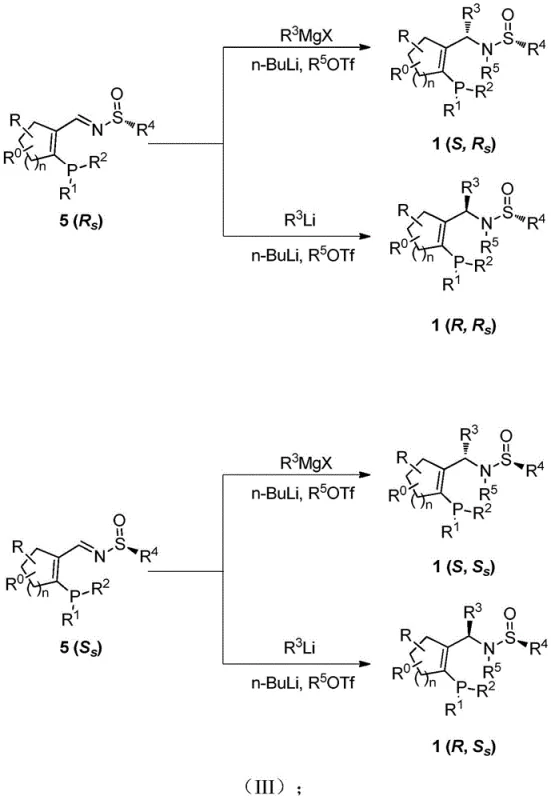
Impurity control is another critical aspect of the Yu-Phos mechanism. The synthetic route is designed to minimize the formation of diastereomeric by-products through the use of highly stereoselective addition reactions. For instance, the addition of Grignard or Organolithium reagents to the chiral imine intermediate proceeds with high diastereofacial selectivity, driven by the existing chirality of the sulfinamide group. This inherent stereocontrol reduces the burden on downstream purification, ensuring that the final ligand product possesses high optical purity. Furthermore, the stability of the P-C and S-N bonds in the final structure prevents racemization under typical reaction conditions, guaranteeing that the chiral information is preserved throughout the catalytic cycle. This robustness is essential for maintaining high turnover numbers and preventing the erosion of enantiomeric excess in the final pharmaceutical or agrochemical product.
How to Synthesize Yu-Phos Efficiently
The synthesis of Yu-Phos is a testament to modern process chemistry, balancing complexity with efficiency to deliver a high-value catalyst. The process begins with the functionalization of a cyclic ketone to introduce the phosphine moiety, followed by condensation with a chiral sulfinamide to establish the imine linkage. The final stereochemical outcome is determined in the subsequent nucleophilic addition step, where the choice of metal reagent (Grignard vs. Organolithium) and reaction temperature dictates the configuration of the new carbon center. This modularity allows for the targeted synthesis of any of the four possible stereoisomers simply by selecting the appropriate enantiomer of the sulfinamide and the specific organometallic reagent. The detailed standardized synthesis steps see guide below, which outlines the precise conditions for maximizing yield and purity.
- Prepare the phosphine-containing aldehyde intermediate via bromination of cyclic ketone, acetal protection, and lithiation-substitution with chlorophosphine.
- Condense the aldehyde intermediate with chiral sulfinamide (Rs or Ss configuration) using a titanium ester condensing agent to form the imine intermediate.
- Perform nucleophilic addition using Grignard or Organolithium reagents followed by N-alkylation to yield the final Yu-Phos ligand with controlled stereocenters.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of Yu-Phos technology offers tangible strategic benefits beyond mere technical performance. The streamlined synthesis route directly translates to cost reduction in pharmaceutical intermediate manufacturing by eliminating the need for expensive chiral resolution agents and reducing the number of unit operations. The high yields reported in the patent mean that less raw material is required to produce a given amount of ligand, lowering the cost of goods sold (COGS) and improving margin potential. Furthermore, the use of common, commercially available reagents such as n-BuLi and standard phosphine chlorides mitigates supply risk, as these materials are produced by multiple global vendors, ensuring continuity of supply even during market fluctuations. This resilience is crucial for maintaining uninterrupted production schedules for downstream API manufacturing.
- Cost Reduction in Manufacturing: The elimination of complex resolution steps and the high overall yield of the synthesis pathway significantly lower the production costs associated with chiral ligand acquisition. By avoiding the 50% yield loss inherent in traditional resolution methods, the process maximizes atom economy and reduces waste disposal costs. The simplified workup procedures, which often require only standard extraction and crystallization rather than preparative HPLC, further decrease operational expenses and solvent consumption. These efficiencies compound over large-scale production runs, resulting in substantial cost savings that can be passed down the supply chain.
- Enhanced Supply Chain Reliability: The reliance on robust, scalable chemistry ensures that Yu-Phos can be manufactured in multi-kilogram to ton quantities without compromising quality. The synthetic intermediates are stable and can be stored or transported if necessary, adding flexibility to the logistics network. Since the process does not depend on rare natural products or proprietary biological enzymes, it is less susceptible to agricultural or biological supply shocks. This stability allows for long-term contracting and inventory planning, giving procurement teams greater confidence in securing the materials needed for critical drug development programs.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, utilizing solvents and reagents that are manageable within standard industrial waste treatment frameworks. The high selectivity of the reaction minimizes the generation of hazardous by-products, simplifying regulatory compliance and reducing the environmental liability associated with chemical manufacturing. The ability to scale from gram to kilogram scale without re-optimizing the core chemistry accelerates the timeline from R&D to commercial production, allowing companies to bring new chiral drugs to market faster while meeting increasingly stringent environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of Yu-Phos ligands in industrial processes. These answers are derived directly from the experimental data and claims within patent CN109851634B, providing a factual basis for decision-making. Understanding these details helps R&D and procurement teams evaluate the feasibility of integrating this technology into their existing workflows.
Q: What distinguishes Yu-Phos from conventional chiral phosphine ligands?
A: Yu-Phos uniquely combines both carbon-centered and sulfur-centered chirality within a single monophosphine structure, offering enhanced stereoselectivity and tunable electronic properties compared to traditional carbon-only chiral ligands.
Q: Is the synthesis of Yu-Phos scalable for industrial production?
A: Yes, the patented method utilizes common reagents like n-BuLi and Grignard reagents with high overall yields (71%-88%), avoiding expensive resolution steps and making it highly suitable for commercial scale-up.
Q: What types of reactions can Yu-Phos catalyze effectively?
A: Yu-Phos forms active complexes with Gold (Au) salts and, after anion exchange with Silver (Ag) salts, effectively catalyzes intermolecular asymmetric cycloaddition reactions of enynones with high endo:exo ratios and enantiomeric excess.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Yu-Phos Supplier
The development of advanced chiral ligands like Yu-Phos represents a critical enabler for the next generation of asymmetric syntheses. NINGBO INNO PHARMCHEM stands at the forefront of this technological wave, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the sensitive organometallic chemistry required for Yu-Phos synthesis, ensuring that every batch meets stringent purity specifications. With rigorous QC labs and a commitment to process safety, we provide a secure source for high-performance catalysts that drive efficiency in your manufacturing operations. We understand the critical nature of chiral purity in pharmaceutical applications and maintain the highest standards of quality control.
We invite you to engage with our technical procurement team to discuss how Yu-Phos can optimize your specific synthetic challenges. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this superior ligand system. We encourage you to contact us for specific COA data and route feasibility assessments tailored to your target molecules. Let us partner with you to accelerate your development timelines and reduce your overall production costs through superior catalytic technology.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
