Advanced Palladium-Catalyzed Synthesis of Benzofuran-3-Carboxamide Intermediates for Commercial Scale-Up
Introduction to Next-Generation Benzofuran Synthesis
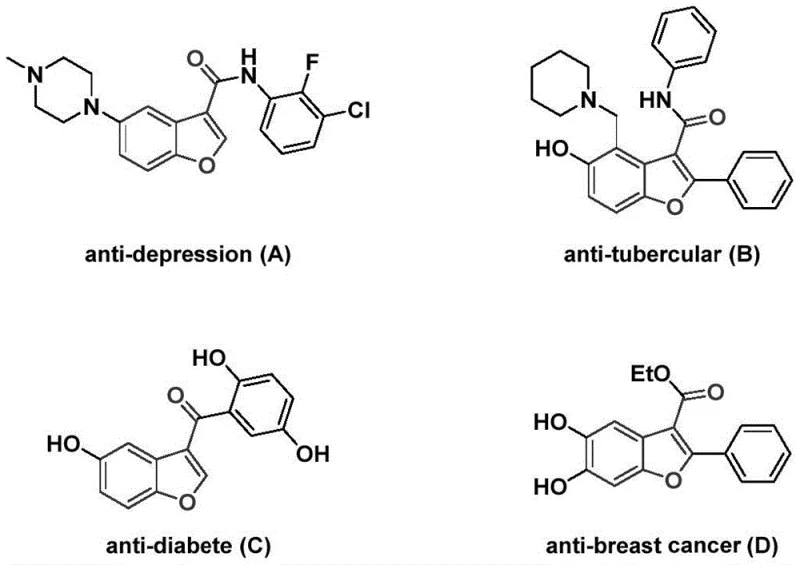
The pharmaceutical industry continuously seeks robust methodologies for constructing privileged scaffolds, and the benzofuran-3-carboxamide motif stands out as a critical structure in modern drug discovery. As detailed in the recent patent CN114751883A, a groundbreaking preparation method has been developed that addresses long-standing challenges in synthesizing these bioactive cores. This innovation is particularly relevant given that benzofuran derivatives exhibit a broad spectrum of biological activities, ranging from antidepressant and antitubercular properties to antidiabetic and antitumor effects, as illustrated by the diverse pharmacological profiles shown in the reference data. The ability to access these complex molecules efficiently is paramount for accelerating the development of new therapeutic agents.
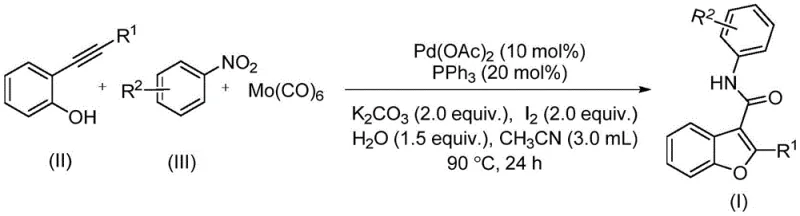
The disclosed technology leverages a sophisticated palladium-catalyzed carbonylation strategy that transforms simple, commercially available starting materials into high-value intermediates in a single operational step. By utilizing 2-alkynylphenols and nitroarenes as the foundational building blocks, this method bypasses the need for pre-functionalized acid chlorides or hazardous gaseous carbon monoxide. Instead, it employs a solid carbon monoxide substitute, molybdenum hexacarbonyl, which releases CO in situ under controlled thermal conditions. This approach not only mitigates safety risks associated with high-pressure gas handling but also streamlines the reaction setup, making it an attractive candidate for both laboratory optimization and industrial manufacturing.
For procurement specialists and supply chain managers, the implications of this patent are profound. The reliance on readily accessible raw materials, such as substituted nitrobenzenes and alkynyl phenols, ensures a stable supply chain with reduced vulnerability to raw material shortages. Furthermore, the reaction conditions—operating at a moderate temperature of 90°C in acetonitrile—are compatible with standard glass-lined or stainless-steel reactors found in most fine chemical production facilities. This compatibility reduces the barrier to entry for adopting this technology, allowing manufacturers to integrate it into existing production lines with minimal capital expenditure on specialized equipment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of benzofuran-3-carboxamides has often relied on multi-step sequences that are both time-consuming and resource-intensive. Classical approaches frequently involve the initial construction of the benzofuran ring followed by separate functionalization steps to introduce the carboxamide moiety, often requiring the use of reactive acid chlorides or activated esters. These intermediates can be unstable, moisture-sensitive, and difficult to handle on a large scale, posing significant safety and quality control challenges. Additionally, traditional carbonylation reactions typically necessitate the use of gaseous carbon monoxide, which requires specialized high-pressure autoclaves and rigorous safety protocols to prevent leakage and exposure, thereby increasing the overall cost of manufacturing and limiting the accessibility of the technology to facilities with advanced infrastructure.
Moreover, conventional methods often suffer from limited substrate scope, where the presence of certain functional groups can inhibit the reaction or lead to undesirable side products. This lack of versatility forces chemists to employ protecting group strategies, adding further steps to the synthesis and reducing the overall atom economy. The cumulative effect of these inefficiencies is a higher cost of goods sold (COGS) and longer lead times for delivering critical pharmaceutical intermediates to clients. In an industry where speed to market is a competitive advantage, these bottlenecks can delay the progression of drug candidates from preclinical studies to clinical trials, ultimately impacting the availability of life-saving medications.
The Novel Approach
In stark contrast, the novel methodology described in patent CN114751883A offers a paradigm shift by consolidating multiple transformation steps into a single, efficient pot. This one-pot synthesis utilizes a palladium catalyst system to simultaneously facilitate the cyclization of the alkyne and the carbonylation of the resulting intermediate, directly coupling it with the amine generated from the reduction of the nitroarene. The use of molybdenum hexacarbonyl as a solid CO source is a game-changer, eliminating the need for dangerous gas cylinders and allowing the reaction to proceed under atmospheric or slightly elevated pressure conditions. This simplification of the reaction setup significantly lowers the operational complexity and enhances the safety profile of the manufacturing process.
Furthermore, this new approach demonstrates exceptional functional group tolerance, accommodating a wide variety of substituents on both the alkyne and the nitroarene components without the need for extensive protection and deprotection sequences. Whether the substrate contains electron-donating groups like methoxy or methyl, or electron-withdrawing groups like halogens, the catalytic system maintains high efficiency and selectivity. This robustness translates directly into cost reduction in pharmaceutical intermediate manufacturing, as it reduces waste generation and simplifies the purification workflow. The ability to produce diverse analogs rapidly also supports medicinal chemistry campaigns, enabling the quick exploration of structure-activity relationships (SAR) to optimize drug candidates.
Mechanistic Insights into Pd-Catalyzed Carbonylative Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors aiming to optimize the process for specific substrates. The reaction is believed to initiate with the coordination of elemental iodine to the carbon-carbon triple bond of the 2-alkynylphenol. This activation step is critical as it primes the alkyne for the subsequent intramolecular nucleophilic attack by the adjacent hydroxyl group. This attack results in the formation of a vinyl iodide intermediate, effectively closing the furan ring and setting the stage for the palladium catalytic cycle. The presence of iodine acts as a promoter, facilitating the oxidative addition of the palladium species into the carbon-iodine bond, which is a key turnover-limiting step in many cross-coupling reactions.
Once the vinyl-palladium intermediate is formed, the carbon monoxide released from the thermal decomposition of molybdenum hexacarbonyl inserts into the palladium-carbon bond. This carbonylation step generates an acyl-palladium species, which is the precursor to the final amide bond. Concurrently, the nitroarene undergoes reduction, likely facilitated by the reducing environment created by the CO/Mo system or the palladium catalyst itself, to generate the corresponding aniline in situ. This aniline then performs a nucleophilic attack on the electrophilic acyl-palladium intermediate. The final step involves reductive elimination, which releases the desired benzofuran-3-carboxamide product and regenerates the active palladium catalyst, completing the cycle. This intricate interplay of cyclization, carbonylation, and reduction in a single vessel highlights the elegance and efficiency of the designed catalytic system.
From an impurity control perspective, this mechanism offers distinct advantages. Because the amine is generated in situ and immediately consumed by the acyl-palladium species, the concentration of free aniline remains low, minimizing the risk of bis-acylation or other side reactions involving the amine. Additionally, the use of a specific ligand system, such as triphenylphosphine, helps stabilize the palladium center and prevents the formation of palladium black, which can lead to catalyst deactivation and metal contamination in the final product. This control over the catalytic species ensures consistent reaction performance and simplifies the downstream removal of residual metals, a critical requirement for meeting stringent pharmaceutical purity specifications.
How to Synthesize Benzofuran-3-carboxamide Efficiently
The practical implementation of this synthesis is straightforward, designed to be accessible for both research-scale optimization and pilot-plant production. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, base, additives, and the solid CO source in an organic solvent, followed by the addition of the substrates. The mixture is then heated to promote the reaction, after which standard workup procedures yield the pure product. This simplicity belies the chemical sophistication of the transformation, offering a reliable route to high-purity pharmaceutical intermediates. For detailed standardized operating procedures and specific stoichiometric ratios tailored to your specific substrate, please refer to the technical guide below.
- Combine palladium acetate, triphenylphosphine, potassium carbonate, iodine, water, and molybdenum hexacarbonyl in acetonitrile solvent.
- Add the 2-alkynylphenol and nitroarene substrates to the reaction mixture under stirring.
- Heat the reaction mixture to 90°C for 24 hours, then filter and purify via column chromatography to isolate the target benzofuran-3-carboxamide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology represents a strategic opportunity to optimize costs and enhance supply reliability. The shift from multi-step, hazardous processes to a streamlined one-pot reaction fundamentally alters the cost structure of producing benzofuran derivatives. By eliminating the need for specialized high-pressure equipment for gaseous CO and reducing the number of isolation steps, the process significantly lowers capital and operational expenditures. This efficiency gain allows suppliers to offer more competitive pricing without compromising on quality, providing a tangible financial benefit to downstream pharmaceutical companies looking to manage their R&D budgets effectively.
- Cost Reduction in Manufacturing: The economic benefits of this method are driven by several factors, primarily the use of cheap and easily obtained starting materials like nitroarenes and alkynyl phenols, which are commodity chemicals with stable global supply chains. The elimination of gaseous carbon monoxide removes the need for expensive safety infrastructure and monitoring systems, while the high atom economy of the one-pot reaction minimizes solvent usage and waste disposal costs. Furthermore, the high reaction efficiency and yields reported in the patent reduce the amount of raw material required per kilogram of product, directly lowering the variable cost of production and improving overall margin potential for manufacturers.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on robust, commercially available reagents such as palladium acetate, triphenylphosphine, and molybdenum hexacarbonyl, all of which are produced by multiple vendors worldwide. This diversification of the supply base mitigates the risk of single-source dependency and ensures continuity of supply even during market fluctuations. Additionally, the mild reaction conditions and tolerance to various functional groups mean that the process is less sensitive to minor variations in raw material quality, reducing the likelihood of batch failures and ensuring consistent delivery schedules to customers who depend on just-in-time inventory models.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, the process aligns well with green chemistry principles by reducing waste and avoiding toxic gases. The simplified workup, involving filtration and chromatography, is easily adaptable to large-scale continuous flow or batch processing, facilitating the commercial scale-up of complex pharmaceutical intermediates. The reduced generation of hazardous byproducts simplifies wastewater treatment and regulatory compliance, lowering the environmental footprint of the manufacturing site. This sustainability aspect is increasingly important for multinational corporations aiming to meet their corporate social responsibility goals and adhere to strict environmental regulations in various jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived from the specific capabilities and advantages outlined in the patent documentation. Understanding these details helps stakeholders assess the feasibility of integrating this technology into their existing portfolios. The answers reflect the robust nature of the catalytic system and its suitability for producing high-quality intermediates required for drug development.
Q: What are the key advantages of using Mo(CO)6 over gaseous CO in this synthesis?
A: Using molybdenum hexacarbonyl (Mo(CO)6) as a solid carbon monoxide substitute significantly enhances operational safety and simplifies reactor requirements compared to handling high-pressure toxic CO gas, making the process more suitable for standard pharmaceutical manufacturing facilities.
Q: Does this method tolerate diverse functional groups on the aromatic rings?
A: Yes, the patented methodology demonstrates excellent substrate compatibility, successfully accommodating various substituents such as halogens (Cl, Br, F), alkyl groups (Me, nBu), and alkoxy groups (OMe) on both the alkyne and nitroarene components without compromising yield.
Q: Is the purification process complex for large-scale production?
A: The post-treatment is streamlined, involving simple filtration followed by silica gel mixing and standard column chromatography, which avoids complex extraction sequences and facilitates easier scale-up for industrial applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzofuran-3-carboxamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed carbonylation technology for the pharmaceutical sector. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped to handle sensitive organometallic chemistry safely, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of benzofuran-3-carboxamide meets the highest international standards. We are committed to being a reliable pharmaceutical intermediate supplier that drives your success through technical excellence and operational reliability.
We invite you to collaborate with us to leverage this advanced synthesis route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your drug development timeline while optimizing your supply chain costs.
