Revolutionizing Aryl Azide Production: A Green Copper-Catalyzed Route for Commercial Scale-Up
Introduction to Advanced Azide Synthesis Technology
The landscape of fine chemical manufacturing is undergoing a paradigm shift towards greener, more atom-economical processes, a transition vividly exemplified by the technology disclosed in patent CN102180755A. This intellectual property introduces a robust methodology for synthesizing aryl azide compounds, specifically phenylazide derivatives, which serve as critical building blocks in modern drug discovery and materials science. Unlike conventional routes that rely on hazardous reagents or harsh conditions, this innovation leverages a cuprous catalyst system in an aqueous medium to facilitate the substitution of aryl boronic acids. For R&D directors and process chemists, this represents a significant leap forward in operational safety and environmental compliance. The ability to conduct these transformations at ambient temperatures using water as the primary solvent not only mitigates the risks associated with volatile organic compounds but also aligns with the stringent sustainability goals of multinational pharmaceutical corporations. By anchoring our analysis in the specific experimental data provided within this patent, we can appreciate the profound implications for scalable manufacturing of high-purity intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of organic azides has been fraught with significant safety and efficiency challenges that hinder large-scale adoption. Traditional methods often involve the diazotization of aromatic amines followed by displacement with azide ions, a process that necessitates the handling of thermally unstable diazonium salts which pose severe explosion hazards if not meticulously controlled. Alternatively, nucleophilic substitution of aryl halides typically requires high temperatures, polar aprotic solvents like DMF or DMSO, and stoichiometric amounts of expensive palladium catalysts or harsh conditions that are incompatible with sensitive functional groups. These legacy processes generate substantial volumes of toxic waste, require complex downstream purification to remove heavy metal residues, and often suffer from moderate yields due to competing side reactions. Furthermore, the reliance on organic solvents increases the fire load of the manufacturing facility and escalates the cost of solvent recovery and disposal, creating a bottleneck for cost-effective production of pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in CN102180755A offers a transformative solution by utilizing readily available aryl boronic acids as the starting material. This approach capitalizes on the stability and commercial availability of boronic acids, which are increasingly produced on a multi-ton scale for Suzuki couplings. The core innovation lies in the use of inexpensive cuprous oxide (Cu2O) as the catalyst and water as the reaction medium, creating a benign chemical environment. 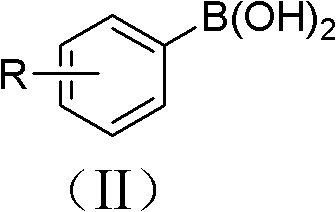 As illustrated by the general structure of the reactants, this system tolerates a wide array of substituents on the aromatic ring, allowing for the direct functionalization of complex molecules without the need for protective groups. The reaction proceeds smoothly under an open atmosphere at temperatures ranging from 10°C to 30°C, eliminating the need for energy-intensive heating or inert gas blanketing. This drastic simplification of the reaction setup not only reduces capital expenditure on specialized equipment but also streamlines the operator workflow, making it an ideal candidate for continuous flow processing or batch manufacturing in existing facilities.
As illustrated by the general structure of the reactants, this system tolerates a wide array of substituents on the aromatic ring, allowing for the direct functionalization of complex molecules without the need for protective groups. The reaction proceeds smoothly under an open atmosphere at temperatures ranging from 10°C to 30°C, eliminating the need for energy-intensive heating or inert gas blanketing. This drastic simplification of the reaction setup not only reduces capital expenditure on specialized equipment but also streamlines the operator workflow, making it an ideal candidate for continuous flow processing or batch manufacturing in existing facilities.
Mechanistic Insights into Cu-Catalyzed Azidation
From a mechanistic perspective, this transformation represents a sophisticated interplay between the copper catalyst, the ammonia additive, and the azide source. The cuprous species acts as a Lewis acid to activate the boron-carbon bond, facilitating the transmetallation step that is crucial for the subsequent substitution. The presence of aqueous ammonia plays a dual role: it serves as a ligand to stabilize the copper center in the aqueous phase, preventing the formation of inactive copper aggregates, and it helps to solubilize the inorganic azide salt, thereby increasing the effective concentration of the nucleophile near the catalytic center. This synergistic effect ensures high turnover numbers and minimizes the induction period often seen in heterogeneous catalysis. For the R&D team, understanding this mechanism is vital for troubleshooting and optimizing the process for specific substrates, as the electronic nature of the substituents on the aryl ring can influence the rate of transmetallation.
Furthermore, the tolerance of this catalytic system towards diverse functional groups is a key differentiator for synthesizing high-purity pharmaceutical intermediates. The mild conditions prevent the decomposition of sensitive moieties such as aldehydes, esters, and nitro groups, which are frequently present in advanced drug candidates. 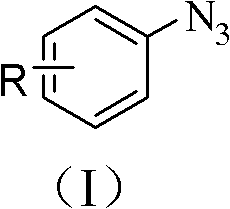 As shown in the product structure, the azide group is installed with high regioselectivity, preserving the integrity of the aromatic scaffold. This high fidelity in chemical transformation translates directly to a cleaner impurity profile, reducing the burden on downstream purification units like chromatography or recrystallization. The ability to maintain such high selectivity in an aqueous environment is rare and underscores the robustness of this copper-ammonia catalytic cycle, offering a reliable pathway for generating complex azide libraries for click chemistry applications.
As shown in the product structure, the azide group is installed with high regioselectivity, preserving the integrity of the aromatic scaffold. This high fidelity in chemical transformation translates directly to a cleaner impurity profile, reducing the burden on downstream purification units like chromatography or recrystallization. The ability to maintain such high selectivity in an aqueous environment is rare and underscores the robustness of this copper-ammonia catalytic cycle, offering a reliable pathway for generating complex azide libraries for click chemistry applications.
How to Synthesize Phenylazide Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and safety. The process begins with the precise charging of cuprous oxide, aqueous ammonia, the specific aryl boronic acid, and sodium azide into a reaction vessel containing water. The simplicity of the reagent list belies the sophistication of the outcome, as this mixture spontaneously generates the active catalytic species upon stirring. Detailed standardized operating procedures regarding mixing rates, addition sequences, and quenching protocols are essential to ensure reproducibility across different batches. The following guide outlines the critical steps derived from the patent embodiments to assist technical teams in replicating this high-yielding transformation.
- Charge a reaction vessel with cuprous oxide catalyst, aqueous ammonia, aryl boronic acid substrate, and sodium azide in water.
- Stir the reaction mixture at room temperature (10-30°C) under an open air atmosphere for 18 to 24 hours to ensure complete conversion.
- Upon completion, adjust pH with sodium hydroxide solution, extract the product with ethyl acetate, and purify via standard washing and concentration techniques.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of adopting this technology are profound, driven primarily by the elimination of costly inputs and the simplification of waste management. The shift from organic solvents to water as the reaction medium results in a drastic reduction in raw material costs, as water is universally available and virtually free compared to dried, HPLC-grade organic solvents. Moreover, the use of cuprous oxide, a bulk commodity chemical, replaces expensive palladium or rhodium catalysts often required in cross-coupling alternatives, leading to substantial savings in catalyst procurement budgets. The open-air operation eliminates the need for nitrogen or argon purging systems, further reducing utility costs and infrastructure requirements. These factors combine to create a manufacturing process with a significantly lower cost of goods sold (COGS), providing a competitive edge in pricing negotiations for bulk intermediates.
- Cost Reduction in Manufacturing: The economic benefits extend beyond simple reagent costs to the entire lifecycle of the production batch. By avoiding the use of volatile organic compounds (VOCs), the facility saves heavily on solvent recovery distillation and hazardous waste incineration fees, which are often the hidden cost drivers in fine chemical synthesis. The high yields reported in the patent embodiments, often exceeding 80% for various substrates, mean that less raw material is wasted, improving the overall mass balance and atom economy. Additionally, the potential for catalyst recycling, as demonstrated in the patent's cyclic experiments, suggests that the effective catalyst cost per kilogram of product can be minimized even further. This holistic reduction in operational expenditure allows for more aggressive pricing strategies while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commoditized starting materials that are less susceptible to geopolitical supply shocks compared to specialized organometallic reagents. Aryl boronic acids and sodium azide are produced by multiple global suppliers, ensuring a diversified sourcing strategy that mitigates the risk of single-source dependency. The robustness of the reaction conditions—tolerating ambient temperature and pressure—means that production is less vulnerable to utility failures such as cooling water loss or power fluctuations that might disrupt cryogenic or high-pressure processes. This operational stability ensures consistent on-time delivery performance, a critical metric for maintaining trust with downstream pharmaceutical clients who operate on tight Just-In-Time inventory schedules.
- Scalability and Environmental Compliance: Scaling this process from grams to tons is inherently safer and more straightforward due to the absence of exothermic runaway risks associated with diazonium chemistry. The aqueous nature of the reaction provides excellent heat dissipation, allowing for larger batch sizes without the need for complex reactor cooling jackets. From an environmental standpoint, the process aligns perfectly with green chemistry principles, generating minimal hazardous waste and avoiding the release of toxic solvents into the environment. This compliance simplifies the permitting process for new manufacturing lines and enhances the corporate sustainability profile, which is increasingly a prerequisite for qualifying as a vendor for top-tier global pharmaceutical companies committed to carbon neutrality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed azidation technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing supply chains.
Q: How does this copper-catalyzed method improve safety compared to traditional diazotization?
A: Traditional diazotization requires handling unstable diazonium salts and strong mineral acids at low temperatures, posing explosion risks. This patented method utilizes stable aryl boronic acids and sodium azide in water at room temperature, drastically reducing thermal hazards and operational complexity.
Q: Can the cuprous oxide catalyst system be recycled for industrial production?
A: Yes, the patent explicitly demonstrates catalyst recyclability. After product extraction, the aqueous mother liquor retains catalytic activity. By replenishing alkali salts and substrates, the system can undergo multiple cycles, significantly lowering catalyst consumption costs.
Q: What is the functional group tolerance of this synthesis route?
A: The method exhibits exceptional tolerance for diverse functional groups including halogens, nitro, aldehyde, ester, and hydroxyl groups. This broad compatibility allows for the direct synthesis of complex pharmaceutical intermediates without requiring extensive protecting group strategies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylazide Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of this patented synthesis route in securing a stable supply of high-quality aryl azides for the global market. As a dedicated CDMO partner, we possess the technical expertise to translate this laboratory-scale innovation into a robust commercial manufacturing process. Our facilities are equipped to handle the specific requirements of aqueous copper catalysis, ensuring that every batch meets stringent purity specifications through our rigorous QC labs. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, allowing us to offer flexible volume options that grow with your clinical and commercial needs. Our commitment to quality assurance means that we can deliver phenylazide intermediates with the consistency and reliability required for GMP-regulated drug substance manufacturing.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your budget. Please contact us to request specific COA data for our reference standards and to discuss route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain that prioritizes both economic efficiency and scientific excellence.
