Advanced Stereoselective Synthesis of Chiral Spiro Pyrazolones for Commercial API Manufacturing
Advanced Stereoselective Synthesis of Chiral Spiro Pyrazolones for Commercial API Manufacturing
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing complex heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. Patent CN112645958A introduces a groundbreaking approach to synthesizing chiral spiro pyrazolone compounds, a privileged structural motif known for its potent biological activities ranging from antitumor to antiviral properties. This innovation leverages a highly efficient [3+2] cycloaddition strategy between N,N'-cyclic imine 1,3-dipoles and pyrazolone exocyclic olefins, facilitated by simple organic additives. By addressing the longstanding challenges of stereoselectivity and operational complexity, this technology offers a compelling value proposition for R&D teams aiming to accelerate lead optimization and for supply chain managers focused on cost-effective manufacturing of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral spiro pyrazolone skeletons has relied heavily on organocatalytic strategies or transition-metal-catalyzed cycloadditions that often suffer from significant practical drawbacks in a commercial setting. Traditional protocols frequently necessitate the use of expensive chiral catalysts, stringent anhydrous conditions, or cryogenic temperatures to achieve acceptable levels of stereocontrol, which drastically inflates the cost of goods sold (COGS). Furthermore, many existing methods exhibit narrow substrate scopes, failing to tolerate diverse functional groups required for modern drug discovery, and often result in poor diastereoselectivity that complicates purification. The reliance on complex catalytic systems also introduces the risk of heavy metal contamination, necessitating additional downstream processing steps that extend lead times and reduce overall process efficiency for large-scale API manufacturing.
The Novel Approach
In stark contrast to these legacy techniques, the methodology disclosed in CN112645958A utilizes a thermally driven [3+2] cycloaddition that operates under remarkably mild and operationally simple conditions. By employing readily available N,N'-cyclic imine 1,3-dipoles and pyrazolone exocyclic olefins in the presence of inexpensive additives like benzoic acid, the reaction proceeds efficiently in common organic solvents such as toluene at elevated temperatures. This approach eliminates the need for sophisticated catalytic systems while delivering exceptional yields and diastereoselectivity, as evidenced by the successful synthesis of various derivatives with dr values exceeding 20:1. The simplicity of the workup, which typically involves direct column chromatography without complex quenching or extraction procedures, represents a paradigm shift towards greener and more economically viable chemical manufacturing.
![General reaction scheme showing the [3+2] cycloaddition between N,N'-cyclic imine 1,3-dipole and pyrazolone exo-olefin](/insights/img/chiral-spiro-pyrazolone-synthesis-pharma-supplier-20260303150424-04.webp)
Mechanistic Insights into 1,3-Dipolar Cycloaddition
The core of this technological breakthrough lies in the unique reactivity of the N,N'-cyclic imine 1,3-dipole, which acts as a versatile three-atom synthon capable of engaging in highly regio- and stereoselective cycloadditions. The reaction mechanism likely proceeds through a concerted or stepwise pathway where the dipole attacks the electron-deficient exocyclic double bond of the pyrazolone derivative, forming two new sigma bonds and generating the spirocyclic center with high fidelity. The presence of the additive, such as benzoic acid, is believed to activate the dipole or stabilize the transition state through hydrogen bonding interactions, thereby lowering the activation energy barrier and accelerating the reaction kinetics without compromising stereochemical integrity. This mechanistic elegance allows for the precise construction of the quaternary stereocenter at the spiro junction, a feat that is notoriously difficult to achieve with high selectivity using conventional alkylation or condensation reactions.
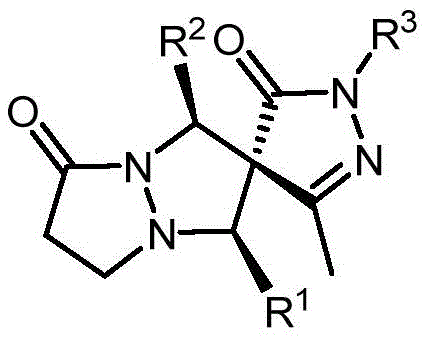
From an impurity control perspective, the high diastereoselectivity observed (dr > 20:1) is a critical advantage that minimizes the formation of unwanted stereoisomers which are often difficult to separate. The rigid transition state imposed by the cyclic nature of the 1,3-dipole and the steric environment of the pyrazolone ring ensures that the reaction favors the formation of a single dominant diastereomer. This inherent selectivity reduces the burden on purification resources and ensures that the final pharmaceutical intermediate meets stringent purity specifications required by regulatory bodies. Additionally, the stability of the starting materials and the robustness of the reaction conditions mean that side reactions such as polymerization or decomposition are minimized, further enhancing the quality profile of the crude product prior to final isolation.
How to Synthesize Chiral Spiro Pyrazolone Efficiently
The practical implementation of this synthesis route is designed for seamless integration into existing laboratory and pilot plant workflows, requiring only standard heating and stirring equipment. The protocol begins with the precise weighing of the N,N'-cyclic imine 1,3-dipole and the pyrazolone exocyclic olefin in a molar ratio of approximately 1:1.25, ensuring complete consumption of the more valuable dipole component. These reactants are dissolved in a suitable organic solvent like toluene, and a catalytic amount of benzoic acid is added to initiate the transformation upon heating to 110°C. Detailed standardized synthetic steps for scaling this reaction from milligram to kilogram quantities are provided in the guide below, ensuring reproducibility and safety compliance.
- Combine N,N'-cyclic imine 1,3-dipole (1.0 equiv) and pyrazolone exo-olefin (1.25 equiv) with a catalytic amount of benzoic acid additive in toluene solvent.
- Heat the reaction mixture to 110°C and stir for approximately 2 hours, monitoring progress via TLC until the starting materials are fully consumed.
- Upon completion, purify the crude reaction mixture directly by column chromatography using an ethyl acetate and petroleum ether gradient to isolate the high-purity target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical novelty, directly impacting the bottom line through operational efficiencies. The elimination of precious metal catalysts and the use of commodity chemicals as additives significantly reduce the raw material costs associated with production, while the simplified workup procedure lowers labor and utility expenses. Moreover, the short reaction time of approximately 2 hours enhances equipment turnover rates, allowing for higher throughput in multipurpose reactors and reducing the overall manufacturing cycle time. These factors collectively contribute to a more resilient and cost-competitive supply chain for high-value pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The substitution of expensive chiral catalysts with inexpensive organic acids like benzoic acid results in substantial cost savings on a per-kilogram basis, while the high yields minimize waste disposal costs. By avoiding the need for specialized ligands or cryogenic cooling systems, the capital expenditure required for process implementation is drastically reduced, making this technology accessible for both small-scale R&D and large-scale commercial production. The high atom economy of the cycloaddition reaction further ensures that raw materials are converted efficiently into the desired product, maximizing resource utilization and minimizing the environmental footprint of the manufacturing process.
- Enhanced Supply Chain Reliability: The starting materials, specifically the N,N'-cyclic imine 1,3-dipoles and pyrazolone derivatives, are synthesized from readily available precursors, mitigating the risk of supply disruptions often associated with exotic reagents. The robustness of the reaction conditions, which tolerate a wide range of functional groups and do not require strictly anhydrous environments, ensures consistent batch-to-batch quality even when scaling up. This reliability allows procurement teams to secure long-term contracts with confidence, knowing that the production timeline is not vulnerable to the complexities of handling sensitive reagents or maintaining苛刻 reaction parameters.
- Scalability and Environmental Compliance: The use of common solvents like toluene and the absence of toxic heavy metals simplify the waste treatment process, facilitating compliance with increasingly stringent environmental regulations. The straightforward purification via column chromatography or crystallization can be easily adapted for industrial scale-up using continuous chromatography or recrystallization techniques, ensuring that the process remains viable at the 100 MT annual production level. This scalability ensures that the supply of chiral spiro pyrazolones can grow in tandem with the clinical development of drug candidates, preventing bottlenecks during the critical transition from preclinical to commercial stages.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology, providing clarity for stakeholders evaluating its potential for their specific projects. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these nuances is essential for assessing the feasibility of integrating this route into your existing portfolio of pharmaceutical intermediates.
Q: What is the diastereoselectivity achieved in this novel synthesis method?
A: The patented methodology demonstrates exceptional stereocontrol, consistently achieving diastereomeric ratios (dr) greater than 20:1 across a wide range of substrates, which significantly simplifies downstream purification processes.
Q: Does this process require expensive transition metal catalysts?
A: No, the process utilizes simple organic additives such as benzoic acid rather than costly transition metal catalysts, thereby eliminating the need for rigorous heavy metal removal steps and reducing overall production costs.
Q: Is the substrate scope limited to specific aromatic groups?
A: The method exhibits broad substrate tolerance, successfully accommodating various aryl, heteroaryl, and alkyl substituents including electron-withdrawing groups like fluoro, chloro, and bromo, as well as electron-donating groups like methoxy and methyl.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Spiro Pyrazolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality intermediates that meet the rigorous demands of modern drug development. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the market. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of chiral spiro pyrazolone delivered adheres to the highest international standards for pharmaceutical applications.
We invite you to collaborate with our technical team to explore how this innovative synthesis route can optimize your specific supply chain requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis, along with specific COA data and route feasibility assessments tailored to your target molecules. By leveraging our expertise in process chemistry and scale-up engineering, we can help you realize the full commercial potential of these valuable chiral scaffolds while minimizing development risks and timelines.
