Scalable Synthesis of Axial Chiral Indole-Naphthalene Compounds for Advanced Catalytic Applications
Scalable Synthesis of Axial Chiral Indole-Naphthalene Compounds for Advanced Catalytic Applications
The landscape of asymmetric catalysis is constantly evolving, driven by the demand for more efficient and selective chiral scaffolds. Patent CN110452150B introduces a groundbreaking methodology for the preparation of axial chiral indole-naphthalene compounds, a class of molecules critical for next-generation organocatalysts. This technology leverages a dynamic kinetic resolution strategy mediated by chiral phosphoric acids to construct complex biaryl architectures in a single operational step. Unlike traditional methods that often struggle with low atom economy or harsh conditions, this process operates under remarkably mild parameters, typically around 25°C, utilizing a binary solvent system of 1,1,2,2-tetrachloroethane and p-xylene. For R&D directors seeking robust pathways to high-value chiral intermediates, this patent represents a significant leap forward in synthetic efficiency and stereocontrol.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of axial chiral indole-naphthalene skeletons has relied heavily on cross-coupling reactions or resolution of racemates, processes that are inherently inefficient and costly. Traditional coupling strategies often require expensive transition metal catalysts, rigorous exclusion of air and moisture, and elevated temperatures that can degrade sensitive functional groups. Furthermore, resolving racemic mixtures typically caps the theoretical yield at 50%, necessitating recycling loops that complicate process flow and increase waste generation. These limitations create substantial bottlenecks for procurement managers aiming to secure reliable supplies of high-purity chiral building blocks, as the supply chain becomes vulnerable to the volatility of precious metal prices and the complexities of multi-step purification protocols.
The Novel Approach
The methodology disclosed in CN110452150B circumvents these hurdles by employing an organocatalytic asymmetric addition reaction. By utilizing a specialized chiral phosphoric acid, the process achieves dynamic kinetic resolution, theoretically allowing for 100% conversion of racemic starting materials into a single enantiomer. The reaction proceeds smoothly at ambient temperatures (20-30°C), significantly reducing energy consumption compared to thermal coupling methods. This approach not only simplifies the operational workflow but also enhances the structural diversity accessible to chemists, as evidenced by the broad substrate scope tolerated by the catalyst system. For a reliable pharmaceutical intermediate supplier, adopting such a streamlined protocol translates directly into improved throughput and reduced environmental footprint.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Dynamic Kinetic Resolution
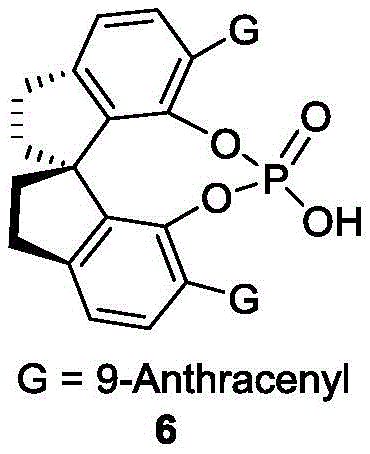
The core of this innovation lies in the precise stereochemical control exerted by the chiral phosphoric acid catalyst, specifically derivatives featuring bulky substituents like the 9-anthracenyl group found in Formula 6. The catalyst functions by simultaneously activating the electrophilic naphthol derivative and the nucleophilic indole species through a dual hydrogen-bonding network. This activation lowers the energy barrier for the C-C bond formation while the chiral pocket of the catalyst sterically directs the approach of the substrates, ensuring high enantioselectivity. The dynamic nature of the resolution allows for the rapid interconversion of the racemic starting material, funneling both enantiomers into the desired product pathway, which is a crucial mechanism for maximizing yield in industrial settings.
Impurity control is intrinsically managed by the high specificity of the organocatalyst. Since the reaction avoids transition metals, the final product is free from heavy metal residues, a common pain point in API manufacturing that requires costly scavenging steps. The mild acidic conditions prevent side reactions such as polymerization or decomposition of the sensitive indole moiety, which are prevalent in stronger acidic or basic environments. Consequently, the crude reaction mixture exhibits a cleaner profile, facilitating easier downstream purification via standard silica gel chromatography. This mechanistic elegance ensures that the resulting axial chiral indole-naphthalene compounds meet the stringent purity specifications required for use as ligands or catalysts in subsequent high-value transformations.
How to Synthesize Axial Chiral Indole-Naphthalene Efficiently
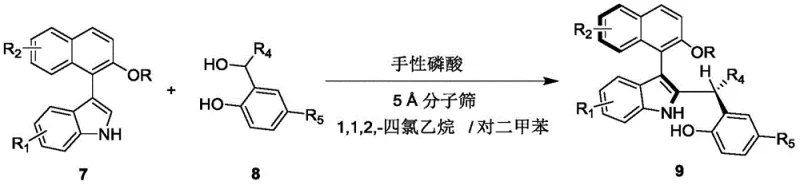
The synthesis protocol outlined in the patent is designed for scalability and reproducibility, making it ideal for translation from benchtop to pilot plant. The process begins with the preparation of a reaction mixture containing the indole-naphthol precursor (Formula 7) and the benzylic alcohol derivative (Formula 8) in a specific solvent ratio. The addition of 5 Å molecular sieves is critical to sequester water generated during the reaction, driving the equilibrium towards product formation. Following the reaction period, standard workup procedures involving filtration and concentration yield the crude product, which can be purified to >95:5 dr and high er values.
- Mix compound of formula 7 and compound of formula 8 in a solvent system of 1,1,2,2-tetrachloroethane and p-xylene (1: 4 v/v).
- Add 5 Å molecular sieves and 10 mol% of chiral phosphoric acid catalyst (Formula 6) to the reaction mixture.
- Stir at 25°C until completion, then filter, concentrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain heads and procurement managers, the shift to this organocatalytic platform offers profound logistical and economic benefits. The elimination of precious metal catalysts removes a major cost driver and supply risk associated with metals like palladium or rhodium. Furthermore, the use of readily available organic starting materials ensures a stable supply chain, insulating production schedules from the geopolitical volatility often seen in the mining sector. The mild reaction conditions also imply lower energy costs and reduced safety hazards, contributing to a more sustainable and cost-effective manufacturing operation overall.
- Cost Reduction in Manufacturing: The transition to metal-free organocatalysis drastically simplifies the purification process by eliminating the need for expensive metal scavengers and extensive washing protocols. This reduction in downstream processing steps leads to substantial cost savings in terms of both materials and labor. Additionally, the high atom economy of the dynamic kinetic resolution means that raw material utilization is maximized, further driving down the cost per kilogram of the final active pharmaceutical ingredient or catalyst precursor.
- Enhanced Supply Chain Reliability: By relying on robust organic small molecule catalysts rather than sensitive transition metal complexes, the manufacturing process becomes more resilient to storage and handling variations. The starting materials, primarily substituted indoles and naphthols, are commodity chemicals with established global supply chains, ensuring consistent availability. This reliability reduces lead times for high-purity chiral intermediates, allowing pharmaceutical partners to maintain tighter production schedules without the risk of catalyst-related delays.
- Scalability and Environmental Compliance: The reaction operates at near-ambient temperatures and uses a solvent system that, while requiring careful management, avoids the extreme conditions that stress reactor infrastructure. The absence of heavy metals simplifies waste treatment and disposal, aligning with increasingly strict environmental regulations. This ease of scale-up from grams to tons ensures that commercial demands can be met without the need for specialized high-pressure or high-temperature equipment, facilitating rapid capacity expansion.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and application of this synthesis technology. Understanding these details is crucial for integrating this method into existing production workflows and assessing its fit for specific project requirements.
Q: What is the primary advantage of this synthesis method over traditional coupling?
A: This method utilizes dynamic kinetic resolution (DKR) to construct the axial chiral skeleton in a single step from racemic原料,avoiding complex multi-step resolutions and achieving high enantiomeric ratios (up to 98:2 er).
Q: What are the optimal reaction conditions for high yield?
A: The optimal conditions involve using a mixed solvent of 1,1,2,2-tetrachloroethane and p-xylene (1:4), a reaction temperature of 25°C, and a specific spiro-binaphthyl chiral phosphoric acid catalyst (Formula 6).
Q: Can these compounds be used directly as catalysts?
A: Yes, the resulting axial chiral indole-naphthalene compounds can be derivatized into chiral phosphine catalysts (like compound 12) which effectively catalyze asymmetric [4+1] cyclization reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Indole-Naphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technologies described in CN110452150B for the development of advanced chiral catalysts. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market supply is seamless. Our facilities are equipped with rigorous QC labs capable of verifying the stringent purity specifications and enantiomeric excess required for these sophisticated molecules, guaranteeing batch-to-batch consistency for your critical applications.
We invite you to collaborate with us to leverage this efficient synthesis route for your specific needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this metal-free approach can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the chiral chemistry market.
