Revolutionizing C-5 Functionalization: Scalable Photoredox Borylation for Advanced Pharmaceutical Intermediates
Revolutionizing C-5 Functionalization: Scalable Photoredox Borylation for Advanced Pharmaceutical Intermediates
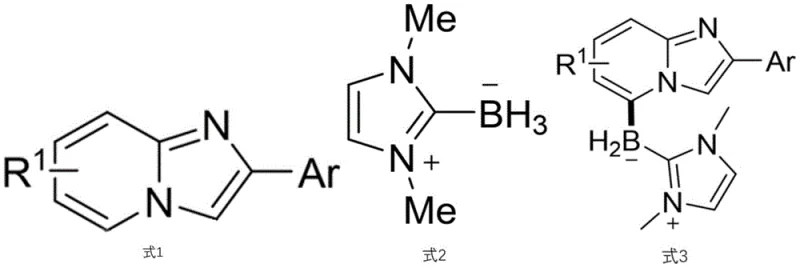
The landscape of heterocyclic functionalization is undergoing a significant transformation driven by the urgent need for sustainable and selective synthetic methodologies in the pharmaceutical industry. Patent CN115232159A introduces a groundbreaking approach for the preparation of C-5-position boronated imidazopyridine derivatives, addressing a long-standing challenge in organic synthesis. Traditionally, the direct functionalization of the imidazo[1,2-a]pyridine scaffold has been heavily biased towards the electron-rich C-3 position, leaving the C-5 position difficult to access without pre-functionalization or harsh conditions. This invention leverages visible-light-induced photoredox catalysis to achieve highly regioselective C-H borylation at the C-5 position. By utilizing stable and inexpensive N-heterocyclic carbene boranes (NHC-BH3) as the boron source and organic photosensitizers like 4CzIPN, this method offers a robust pathway to valuable building blocks. These derivatives serve not only as critical intermediates for drug synthesis due to the versatility of the carbon-boron bond but also exhibit potential as fluorescent active molecules, opening new avenues in material science and bioscience applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbon-boron bonds on nitrogen-containing heterocycles has relied heavily on transition metal catalysis, particularly using iridium or rhodium complexes. While effective for simple arenes, these traditional methods face severe limitations when applied to poly-azacyclic aromatic hydrocarbons like imidazopyridines. The primary issue lies in regioselectivity; the inherent electronic properties of the pyridine ring often direct metal catalysts to the C-3 position or result in unselective mixtures of C-2 and C-3 functionalized products. Furthermore, transition metal catalysts are often expensive, sensitive to air and moisture, and require rigorous removal processes to meet the stringent purity standards of pharmaceutical manufacturing. The presence of trace heavy metals in the final Active Pharmaceutical Ingredient (API) is a major regulatory concern, necessitating additional purification steps that increase cost and reduce overall yield. Additionally, many conventional borylation strategies require high temperatures or stoichiometric amounts of directing groups, which adds complexity to the synthetic route and generates significant chemical waste, contradicting the principles of green chemistry.
The Novel Approach
In stark contrast, the methodology disclosed in CN115232159A represents a paradigm shift by employing a metal-free photoredox strategy. This novel approach utilizes visible light to activate the reaction, operating under mild room temperature conditions which significantly enhances safety and energy efficiency. The use of 4CzIPN or 3CzClIPN as organic photosensitizers eliminates the dependency on scarce precious metals, thereby simplifying the supply chain and reducing raw material costs. Crucially, this method achieves exceptional regioselectivity for the C-5 position, a site that is notoriously difficult to target using standard electrophilic or nucleophilic substitution patterns. The reaction tolerates a wide range of functional groups on the aryl ring, including electron-donating and electron-withdrawing substituents, demonstrating broad substrate scope. This flexibility allows medicinal chemists to access diverse chemical space rapidly without the need for protecting group strategies. The simplicity of the operation, combined with the use of commercially available reagents like tert-butyl peroxybenzoate (TBPB) as an oxidant, makes this process highly attractive for both laboratory discovery and potential industrial scale-up.
Mechanistic Insights into Photoredox-Catalyzed C-H Borylation
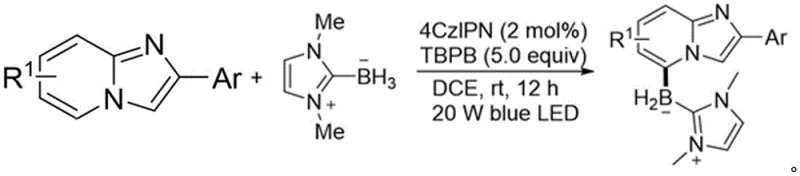
The success of this transformation relies on a sophisticated interplay between the excited state of the photosensitizer and the radical reactivity of the boron source. Upon irradiation with blue LED light, the 4CzIPN photosensitizer enters an excited state capable of engaging in single-electron transfer (SET) processes. This initiates a Minisci-type radical reaction mechanism, which is distinct from the two-electron pathways typical of transition metal catalysis. The N-heterocyclic carbene borane (NHC-BH3) serves as a radical precursor, generating a boryl radical species under the oxidative conditions provided by TBPB. This boryl radical is electrophilic in nature and selectively attacks the electron-deficient positions of the protonated or coordinated imidazopyridine ring. The unique electronic environment of the imidazo[1,2-a]pyridine scaffold, combined with the specific redox potentials involved, directs the radical addition preferentially to the C-5 position. This mechanistic pathway bypasses the thermodynamic preference for C-3 functionalization observed in ionic reactions. Following the radical addition, the intermediate undergoes rearomatization and oxidation to yield the stable C-5 boronated product. Understanding this radical manifold is crucial for R&D teams aiming to further optimize reaction parameters or adapt this chemistry to other heterocyclic systems.
From an impurity control perspective, the radical nature of this reaction offers distinct advantages. Traditional metal-catalyzed reactions often suffer from homocoupling side reactions or over-borylation, leading to complex impurity profiles that are difficult to separate. In this photoredox system, the stoichiometry of the oxidant and the boron source can be finely tuned to minimize such side reactions. The use of NHC-BH3, which is more stable than amine-boranes, reduces the formation of decomposition byproducts during the reaction course. Furthermore, the absence of metal ligands prevents the formation of metal-complexed impurities that often co-elute with the product during chromatography. This results in a cleaner crude reaction mixture, facilitating easier downstream processing. For quality control laboratories, this translates to more straightforward analytical method development and higher confidence in the purity of the final intermediate. The ability to predict and control the radical lifecycle ensures consistent batch-to-batch reproducibility, a key metric for any process intended for commercial manufacturing.
How to Synthesize C-5 Boronated Imidazopyridine Efficiently
To implement this synthesis effectively, precise control over reaction conditions is essential to maximize yield and selectivity. The protocol involves mixing the imidazopyridine substrate with the NHC-BH3 reagent in a suitable solvent such as dichloroethane (DCE). The addition of the photosensitizer 4CzIPN at a loading of approximately 2 mol% is critical to initiate the catalytic cycle efficiently without causing excessive background absorption of light. The oxidant, preferably TBPB, is added in excess to drive the reaction to completion, typically requiring a molar ratio of 1:5 to 1:8 relative to the substrate. The reaction vessel must be transparent to blue light to ensure uniform irradiation throughout the mixture. Stirring is maintained for 12 to 18 hours at room temperature to allow full conversion. Monitoring via TLC is recommended to determine the optimal endpoint. Once completed, the solvent is removed under reduced pressure, and the residue is purified using standard silica gel chromatography. This streamlined workflow minimizes unit operations and reduces the overall processing time compared to multi-step traditional syntheses.
- Mix the imidazo[1,2-a]pyridine substrate with N-heterocyclic carbene borane (NHC-BH3), 4CzIPN photosensitizer, and TBPB oxidant in DCE solvent.
- Irradiate the reaction mixture with a 20 W blue LED light source at room temperature for 12 to 18 hours under stirring.
- Purify the resulting C-5 boronated product via preparative TLC or column chromatography to obtain the high-purity derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this photoredox borylation technology presents substantial strategic benefits regarding cost structure and supply reliability. The shift away from precious metal catalysts fundamentally alters the cost dynamics of producing these specialized intermediates. By eliminating the need for iridium or rhodium complexes, which are subject to volatile market pricing and geopolitical supply risks, manufacturers can achieve significant cost reduction in pharmaceutical intermediate manufacturing. The stability of the reagents involved, particularly the NHC-BH3 boron source, ensures longer shelf life and reduced waste due to reagent degradation. This stability simplifies inventory management and reduces the frequency of urgent replenishment orders. Furthermore, the mild reaction conditions reduce the energy load on production facilities, as there is no need for high-temperature heating or cryogenic cooling systems. These operational efficiencies contribute to a lower carbon footprint and align with corporate sustainability goals, which are increasingly becoming a criterion for supplier selection in the global pharmaceutical market.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes a major cost driver from the bill of materials. Additionally, the simplified purification process, which does not require specialized scavengers for heavy metal removal, reduces the consumption of auxiliary materials and labor hours. The high atom economy of the direct C-H functionalization approach means less starting material is wasted in pre-functionalization steps. These factors combine to create a leaner manufacturing process with substantially lower variable costs per kilogram of product. The use of commodity chemicals like TBPB and DCE further insulates the production cost from the fluctuations seen in the specialty chemical market.
- Enhanced Supply Chain Reliability: The reagents required for this synthesis, including the organic photosensitizer and the boron source, are becoming increasingly available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction to ambient conditions means that transportation and storage requirements are less stringent compared to air-sensitive metal catalysts. This resilience ensures continuous production capability even during logistical disruptions. For supply chain heads, this translates to shorter lead times for high-purity pharmaceutical intermediates and a more predictable delivery schedule. The scalability of the photochemical process, potentially adaptable to continuous flow reactors, further enhances the ability to ramp up production volume quickly in response to market demand spikes.
- Scalability and Environmental Compliance: The environmental profile of this method is superior to traditional routes, generating less hazardous waste and avoiding the discharge of heavy metals. This simplifies compliance with increasingly strict environmental regulations regarding effluent treatment. The scalability is supported by the use of standard LED lighting arrays which can be easily integrated into existing reactor setups or dedicated photochemical flow systems. The reduced thermal load lowers the risk of thermal runaway incidents, enhancing overall plant safety. These attributes make the technology highly suitable for commercial scale-up of complex pharmaceutical intermediates, ensuring that the supply can grow in tandem with the clinical progression of drug candidates utilizing this scaffold.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this C-5 borylation technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing R&D pipelines or manufacturing portfolios. The answers reflect the practical realities of working with photoredox systems and the specific advantages of the imidazopyridine scaffold modification.
Q: What is the primary advantage of this photoredox borylation method over traditional transition metal catalysis?
A: The primary advantage is the elimination of expensive and toxic transition metals like iridium. This method uses an organic photosensitizer (4CzIPN), which simplifies purification, reduces heavy metal contamination risks, and lowers overall production costs while maintaining high regioselectivity at the C-5 position.
Q: Why is C-5 selective borylation of imidazopyridines considered challenging?
A: Imidazopyridines are electron-rich at the C-3 position, making C-3 functionalization thermodynamically favored. Traditional methods often struggle to overcome this inherent electronic bias to target the C-5 position. This patent utilizes a specific radical pathway that successfully targets the proximal C-5 site with high selectivity.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the method operates at room temperature using visible light and common solvents like DCE. The absence of stringent temperature controls and the use of stable reagents like NHC-BH3 make it highly suitable for scale-up in flow chemistry or large batch reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C-5 Boronated Imidazopyridine Supplier
As the pharmaceutical industry continues to explore novel heterocyclic scaffolds for next-generation therapeutics, the ability to access functionalized building blocks like C-5 boronated imidazopyridines is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this technological evolution, leveraging advanced photoredox methodologies to deliver high-quality intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your needs from early-stage discovery through to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for drug substance synthesis. Our commitment to innovation allows us to offer customized solutions that optimize both performance and cost-efficiency for our global partners.
We invite you to collaborate with us to unlock the potential of this cutting-edge chemistry for your projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply chain partner dedicated to advancing your scientific goals through superior chemical manufacturing excellence.
