Advanced Palladium-Catalyzed Synthesis of Benzofuranone Spirocycle Intermediates for Commercial Immunosuppressant Production
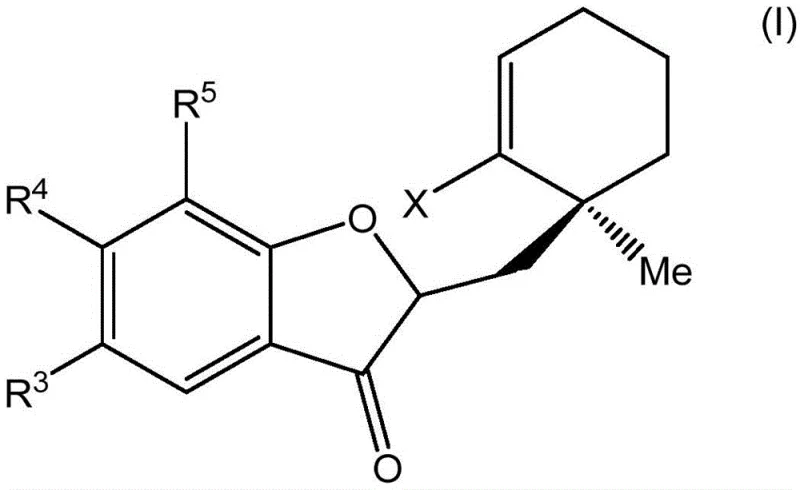
The pharmaceutical industry continuously seeks robust synthetic routes for complex natural product analogs, particularly those exhibiting potent immunosuppressive activity. Patent CN110650735A introduces a groundbreaking diastereoselective process for preparing benzofuranone-yl 4,5-spirocycles via metal-catalyzed intramolecular alkenylation. This technology addresses the critical supply bottleneck associated with Phyllanthus hainanensis compounds, which are naturally available in negligible quantities of approximately 5-20 milligrams per 5 kilograms of dry plant material. By establishing a reliable synthetic pathway, this innovation enables the consistent production of high-purity pharmaceutical intermediates that mimic the western portion of the natural product structure. The method leverages advanced transition metal catalysis to construct highly strained ring systems that were previously difficult to access with adequate stereocontrol. For R&D directors and procurement specialists, this represents a significant shift from dependency on unstable natural sources to a controllable, scalable chemical manufacturing process. The ability to synthesize these complex motifs opens new avenues for developing novel immunosuppressive therapies with improved safety profiles compared to current standards like cyclosporine A.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to obtaining Phyllanthus hainanensis compounds rely heavily on extraction from native shrubs, a method plagued by severe inefficiencies and environmental unsustainability. The natural abundance of these bioactive triterpenoids is exceptionally low, making large-scale clinical development economically unfeasible without synthetic alternatives. Furthermore, previous synthetic attempts often struggled with the formation of the unique benzofuranone-based 4,5-spiro ring, which contains a strained exocyclic olefin motif essential for biological activity. Conventional cyclization strategies frequently resulted in poor diastereoselectivity, generating complex mixtures of isomers that required costly and material-intensive chromatographic purification. The lack of precise stereocontrol not only reduced overall yields but also complicated the regulatory approval process due to impurity profile concerns. Additionally, older methods often utilized harsh reaction conditions or expensive stoichiometric reagents that generated significant chemical waste, conflicting with modern green chemistry principles demanded by supply chain heads. These limitations collectively hindered the commercial viability of this promising class of immunosuppressive agents.
The Novel Approach
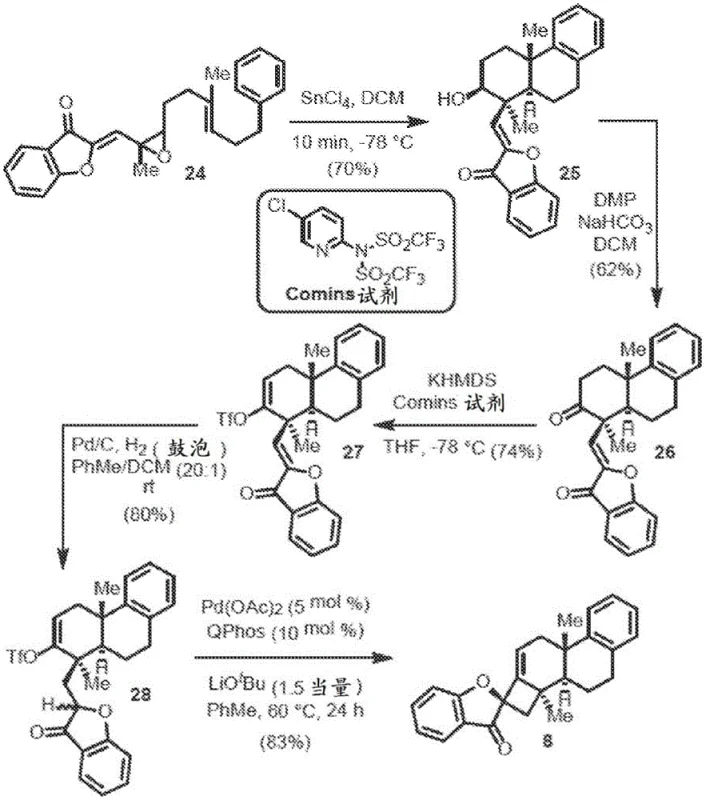
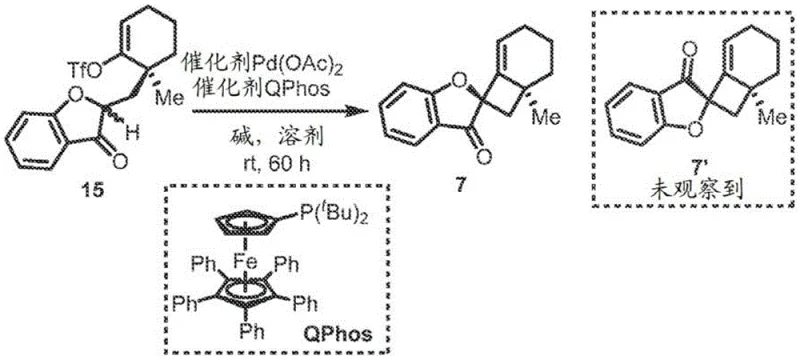
The novel approach disclosed in the patent utilizes a sophisticated palladium-catalyzed intramolecular alkenylation strategy to overcome the inherent difficulties of forming four-membered spirocyclic rings. By employing a specific combination of palladium acetate and the bulky QPhos ligand, the method successfully facilitates the challenging reductive elimination step required to close the strained cyclobutane ring. This catalytic system operates under relatively mild conditions, often at room temperature or moderately elevated temperatures around 60°C, depending on the substrate complexity. The process demonstrates exceptional diastereoselectivity, achieving ratios greater than 20:1 in optimized examples, which drastically simplifies downstream purification requirements. Unlike extraction, this synthetic route allows for the introduction of diverse substituents on the aromatic rings, enabling the creation of a broad library of analogs for structure-activity relationship studies. The use of commercially available starting materials and standard organic solvents like tetrahydrofuran and toluene further enhances the practicality of this method for industrial application. This strategic shift from extraction to catalytic synthesis ensures a stable supply chain capable of meeting the rigorous demands of global pharmaceutical manufacturing.
Mechanistic Insights into Pd-Catalyzed Intramolecular Alkenylation
The core mechanistic advantage of this technology lies in the precise orchestration of the palladium catalytic cycle to favor the formation of the desired spirocyclic geometry. The reaction initiates with the oxidative addition of the palladium catalyst to the vinyl triflate moiety present on the ketone intermediate, generating a reactive organopalladium species. The presence of the QPhos ligand is paramount, as its substantial steric bulk and electron-rich character promote the subsequent migratory insertion and, crucially, the reductive elimination step. Density functional theory calculations support the observation that the transition state leading to the desired diastereomer is energetically favored by approximately 7.0 kcal/mol over the undesired pathway. This energy difference is attributed to favorable interactions between the carbonyl lone pair electrons and the palladium center during the ring-closing event. Such mechanistic understanding allows chemists to fine-tune reaction parameters to maximize yield and selectivity. For technical teams, this level of mechanistic clarity reduces the risk associated with process scale-up, as the key drivers of selectivity are well-defined and reproducible. The robustness of the catalytic cycle ensures that even with complex polycyclic substrates, the reaction proceeds with high fidelity, minimizing the formation of byproducts that could compromise product quality.
Impurity control is inherently built into the high diastereoselectivity of the reaction, which serves as a critical quality attribute for pharmaceutical intermediates. By achieving diastereomeric ratios of 20:1 or higher, the process significantly reduces the burden on purification units, thereby lowering solvent consumption and processing time. The use of non-nucleophilic bases such as lithium tert-butoxide prevents unwanted side reactions at the carbonyl center, ensuring that the alkenylation proceeds cleanly. Furthermore, the reaction conditions are compatible with various functional groups, allowing for late-stage diversification of the molecular scaffold without the need for extensive protecting group manipulations. This chemoselectivity is vital for maintaining high overall throughput in a manufacturing setting. The patent data indicates that the reaction can be performed at concentrations suitable for production, avoiding the dilution issues that often plague intramolecular cyclizations. Consequently, the impurity profile of the final spirocycle is markedly cleaner compared to traditional methods, facilitating easier compliance with stringent regulatory standards for drug substances. This technical superiority translates directly into operational efficiency and cost effectiveness for the manufacturing partner.
How to Synthesize Benzofuranone Spirocycle Efficiently
The synthesis of these high-value intermediates follows a streamlined protocol designed for reproducibility and safety in a controlled laboratory or pilot plant environment. The process begins with the preparation of the ketone precursor, which involves an aldol condensation followed by a selective reduction to install the necessary vinyl triflate leaving group. Once the precursor is secured, the critical cyclization step is executed by combining the substrate with the palladium catalyst system in an anhydrous solvent under an inert atmosphere. Detailed standardized operating procedures regarding reagent grades, addition rates, and temperature profiles are essential to maintain the high diastereoselectivity observed in the patent examples. Operators must ensure strict exclusion of moisture and oxygen to prevent catalyst deactivation, which could lead to incomplete conversion and increased impurity levels. The reaction progress is typically monitored using chromatographic techniques to determine the optimal quench point, ensuring maximum yield before workup. Following the reaction, standard aqueous workup and crystallization or chromatography techniques are employed to isolate the pure spirocycle. Adhering to these validated parameters ensures that the resulting material meets the rigorous specifications required for downstream drug development.
- Prepare the ketone intermediate containing a vinyl triflate leaving group and a benzofuranone moiety through aldol condensation and selective reduction steps.
- Combine the intermediate with a palladium catalyst such as Pd(OAc)2 and a bulky phosphine ligand like QPhos in an anhydrous ether or aromatic solvent.
- Add a non-nucleophilic base such as lithium tert-butoxide and heat the mixture to facilitate intramolecular alkenylation, yielding the spirocycle with high diastereoselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic methodology offers substantial advantages by decoupling the supply of critical immunosuppressive intermediates from the volatility of agricultural sourcing. The transition to a fully synthetic route eliminates the risks associated with crop failures, seasonal variations, and geopolitical instability that often affect natural product supply chains. By utilizing commodity chemicals and widely available transition metal catalysts, the manufacturing process becomes more resilient to market fluctuations in raw material pricing. The high efficiency of the catalytic system means that less starting material is wasted, directly contributing to cost reduction in pharmaceutical intermediates manufacturing. Furthermore, the scalability of the reaction, demonstrated from milligram to gram scales in the patent, suggests a clear path to commercial tonnage production without fundamental changes to the chemistry. This predictability allows supply chain managers to plan inventory levels more accurately and negotiate long-term contracts with greater confidence. The ability to produce diverse analogs from a common intermediate also provides flexibility in responding to changing clinical demands without retooling the entire production line.
- Cost Reduction in Manufacturing: The implementation of this catalytic process drives cost efficiency primarily through the elimination of expensive and time-consuming purification steps necessitated by low-selectivity methods. By achieving high diastereoselectivity inherently within the reaction, the need for repetitive chromatography is drastically simplified, leading to substantial savings in solvent usage and labor hours. The use of catalytic amounts of palladium, rather than stoichiometric reagents, further reduces the material cost per kilogram of product. Additionally, the mild reaction conditions reduce energy consumption associated with heating or cooling large reactors, contributing to a lower overall carbon footprint and utility costs. The robustness of the reaction minimizes batch failures, ensuring that capital equipment is utilized effectively without downtime for cleaning or reprocessing. These cumulative efficiencies result in a more competitive cost structure for the final active pharmaceutical ingredient, benefiting the entire value chain.
- Enhanced Supply Chain Reliability: Adopting this synthetic route significantly enhances supply chain reliability by establishing a consistent and controllable source of key intermediates. Unlike natural extraction, which is subject to biological variability, chemical synthesis offers batch-to-batch consistency that is critical for regulatory compliance and patient safety. The reliance on commercially available reagents ensures that production is not bottlenecked by scarce specialty chemicals, reducing the risk of supply disruptions. The modular nature of the synthesis allows for parallel processing or campaign manufacturing, enabling rapid scaling to meet sudden increases in demand. This reliability fosters stronger partnerships between chemical suppliers and pharmaceutical companies, as trust is built on the ability to deliver on time and to specification consistently. Ultimately, a stable supply of high-quality intermediates accelerates the drug development timeline, bringing life-saving therapies to patients faster.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents and conditions that are manageable in large-scale reactor systems. The high atom economy of the intramolecular alkenylation reduces the generation of chemical waste, aligning with increasingly strict environmental regulations governing pharmaceutical manufacturing. The ability to recycle solvents and recover catalyst residues further minimizes the environmental impact of the production process. This compliance not only avoids potential fines but also enhances the corporate social responsibility profile of the manufacturing entity. The straightforward workup procedures reduce the complexity of waste treatment facilities, lowering capital expenditure for environmental control systems. As the industry moves towards greener manufacturing practices, this technology positions the supply chain as a leader in sustainable chemical production, appealing to environmentally conscious stakeholders and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. They are derived from the specific experimental data and beneficial effects outlined in the patent documentation to provide clarity for potential partners. Understanding these aspects is crucial for evaluating the feasibility of integrating this route into existing manufacturing portfolios. The answers reflect the proven capabilities of the method as demonstrated in the provided examples, ensuring that expectations are grounded in verified scientific results. This transparency aids in risk assessment and strategic planning for R&D and operations teams considering adoption.
Q: What is the primary advantage of this Pd-catalyzed method over natural extraction?
A: Natural extraction yields are extremely low (5-20mg per 5kg plant material), whereas this synthetic route offers scalable production with yields exceeding 65% and high stereoselectivity.
Q: Why is the QPhos ligand critical for this reaction?
A: The bulky QPhos ligand facilitates the difficult reductive elimination step required to form the highly strained four-membered cyclobutane ring within the spirocycle structure.
Q: Can this process be scaled for commercial manufacturing?
A: Yes, the patent demonstrates gram-scale synthesis with robust conditions using common solvents like THF and toluene, indicating strong potential for kilogram to ton-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzofuranone Spirocycle Supplier
NINGBO INNO PHARMCHEM stands ready to leverage this advanced palladium-catalyzed technology to support your development of next-generation immunosuppressive therapies. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop to marketplace. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of analyzing complex spirocyclic structures to guarantee the highest quality standards. We understand the critical nature of supply continuity for clinical trials and commercial launch, and our robust supply chain management systems are designed to mitigate risks proactively. By partnering with us, you gain access to deep technical expertise in transition metal catalysis and process optimization, allowing you to focus on your core competencies while we handle the complexities of chemical manufacturing. Our commitment to excellence ensures that every batch delivered meets the exacting requirements of global regulatory agencies.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can be tailored to your specific project needs. Request a Customized Cost-Saving Analysis to understand the economic benefits of switching to this catalytic route for your supply chain. Our experts are available to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver high-purity benzofuranone spirocycle intermediates reliably. Let us collaborate to accelerate your drug development timeline and secure a competitive advantage in the immunosuppressant market through superior chemical manufacturing solutions.
