Advanced Catalytic Strategy for Scalable Avacopan Manufacturing and Cost Efficiency
Advanced Catalytic Strategy for Scalable Avacopan Manufacturing and Cost Efficiency
The pharmaceutical industry is constantly seeking more efficient pathways for the production of complex active pharmaceutical ingredients (APIs) and their intermediates. Patent CN114262291B introduces a groundbreaking synthetic methodology for Avacopan, a critical therapeutic agent for ANCA-associated vasculitis. This innovation addresses long-standing challenges in chiral synthesis by replacing traditional resolution techniques with a highly selective asymmetric hydrogenation process. By leveraging a specialized rhodium-based catalytic system, the disclosed method achieves a remarkable overall yield of 42.55% across just four reaction steps. For R&D directors and procurement specialists, this represents a significant leap forward in process chemistry, offering a streamlined alternative to the cumbersome multi-step sequences previously described in literature such as WO2016/053890. The elimination of chiral resolution not only simplifies the workflow but also drastically reduces material loss, positioning this technology as a cornerstone for next-generation manufacturing of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Avacopan has been plagued by inefficiencies inherent to classical chiral resolution strategies. As illustrated in prior art documents like WO2016/053890, traditional routes often necessitate the use of resolving agents such as ditoluoyl-L-tartaric acid to separate enantiomers. This approach is fundamentally flawed from a process economics perspective because it theoretically caps the maximum yield at 50% for the desired isomer, unless dynamic kinetic resolution is employed, which adds further complexity. Furthermore, these legacy methods typically involve lengthy preparation steps that increase the cumulative impurity profile and extend production lead times. The reliance on stoichiometric amounts of chiral acids generates substantial waste streams, complicating downstream purification and environmental compliance. For supply chain managers, these factors translate into higher raw material costs and unpredictable batch-to-batch consistency, creating bottlenecks in the reliable supply of avacopan intermediates required for global clinical and commercial demands.
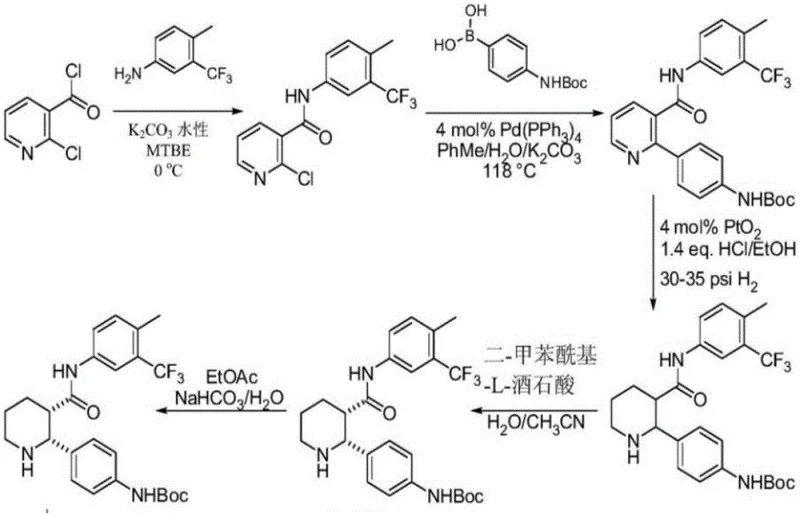
The Novel Approach
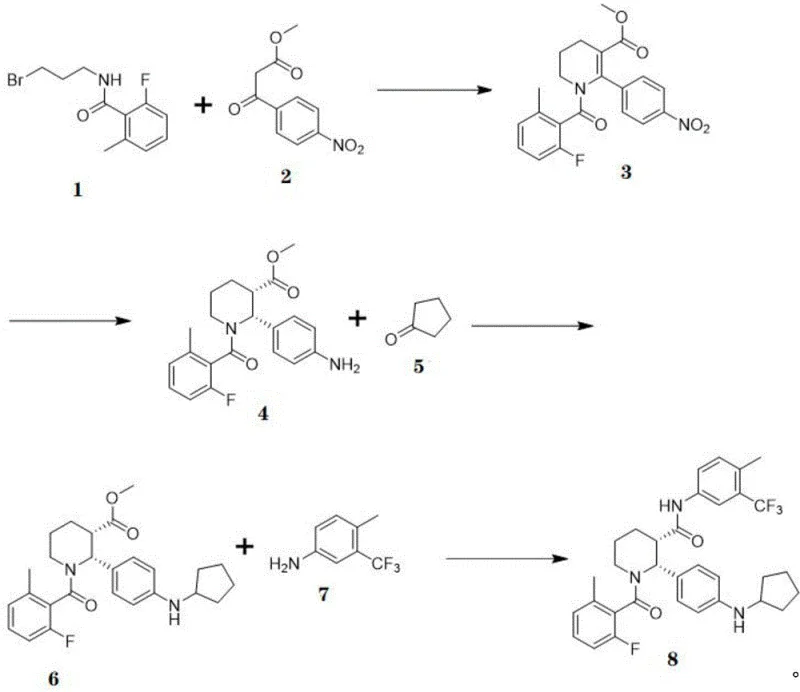
In stark contrast, the methodology disclosed in CN114262291B revolutionizes the production landscape by implementing a direct asymmetric hydrogenation strategy. This novel approach bypasses the need for chiral resolution entirely, utilizing a catalytic system that instills chirality directly during the bond-forming event. The synthetic route is condensed into four distinct operations: ring closure, chiral reduction, coupling with cyclopentanone, and final amide formation. This reduction in step count is pivotal for cost reduction in API manufacturing, as fewer unit operations mean lower solvent consumption, reduced labor hours, and minimized equipment occupancy. The process demonstrates exceptional efficiency, with the key hydrogenation step yielding Compound 4 in 96% yield with an enantiomeric excess (ee) of 98.8%. Such high selectivity ensures that the final product meets stringent purity specifications without the need for extensive recrystallization or chromatographic purification, thereby enhancing the overall throughput and economic viability of the synthesis.
Mechanistic Insights into Rh-Catalyzed Asymmetric Hydrogenation
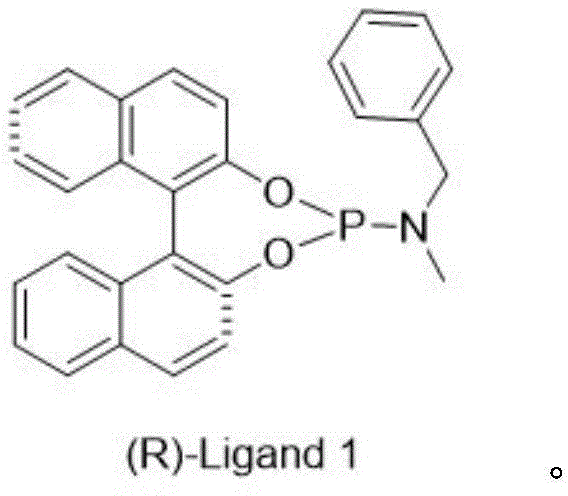
The heart of this technological breakthrough lies in the sophisticated catalytic cycle employed in Step 2 of the synthesis. The reaction utilizes di(1,5-cyclooctadiene)rhodium(I) tetrafluoroborate (Rh(COD)2BF4) as the metal precursor, coordinated with a bespoke chiral ligand known as (R)-Ligand 1. This specific ligand architecture is critical for inducing the desired stereochemistry during the reduction of the olefinic bond in Compound 3. The mechanism likely involves the formation of a cationic rhodium-hydride species which coordinates to the substrate in a highly organized transition state dictated by the steric bulk of the ligand. Operating under mild conditions—specifically at 1 atmosphere of hydrogen pressure and temperatures between 20-28°C—the catalyst facilitates the transfer of hydrogen to the prochiral face of the molecule with extraordinary precision. This mildness is advantageous for scale-up, as it eliminates the need for high-pressure autoclaves or cryogenic cooling, reducing both capital expenditure and operational risks associated with high-energy processes.
Furthermore, the choice of solvent system plays a crucial role in maintaining catalyst stability and activity. The patent specifies a mixture of dichloromethane and isopropanol, which provides an optimal balance of solubility for both the organic substrate and the polar catalytic species. Impurity control is inherently built into this mechanism; because the chirality is established catalytically with 98.8% ee, the formation of the unwanted enantiomer is suppressed to negligible levels. This contrasts sharply with resolution methods where the unwanted isomer must be physically separated and often discarded. The robustness of this catalytic system allows for consistent performance over extended reaction times (10-25 hours), ensuring complete conversion of the starting material. For quality assurance teams, this mechanistic reliability translates to a cleaner impurity profile, simplifying analytical validation and regulatory filing processes for the final drug substance.
How to Synthesize Avacopan Efficiently
Implementing this advanced synthetic route requires precise adherence to the optimized parameters outlined in the patent to ensure reproducibility and safety. The process begins with the cyclization of precursors to form the core scaffold, followed by the critical asymmetric hydrogenation that sets the stereocenter. Subsequent functionalization involves reductive amination and final amide coupling to complete the molecular architecture. While the chemistry is robust, attention to detail regarding reagent quality, particularly the anhydrous conditions for the cyclization and coupling steps, is paramount for achieving the reported yields. The following guide summarizes the standardized operational procedure derived from the patent examples, serving as a foundational reference for process engineers looking to adopt this technology for commercial production.
- Cyclization of Compound 1 and Compound 2 in anhydrous acetone and DMF with K2CO3 to form Compound 3.
- Asymmetric hydrogenation of Compound 3 using Rh(COD)2BF4 and (R)-Ligand 1 under 1 atm H2 to obtain chiral Compound 4.
- Reductive amination of Compound 4 with cyclopentanone using sodium triacetoxyborohydride to generate Compound 6.
- Hydrolysis of Compound 6 followed by amide coupling with 4-methyl-3-(trifluoromethyl)aniline (Compound 7) to yield Avacopan.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology offers transformative benefits that extend beyond simple yield improvements. The primary advantage lies in the drastic simplification of the manufacturing workflow. By eliminating the chiral resolution step, the process removes a major bottleneck that traditionally consumes significant time and resources. This streamlining leads to substantially reduced production cycles, allowing manufacturers to respond more agilely to market fluctuations and urgent supply demands. Additionally, the reduction in step count from lengthy multi-step sequences to a concise four-step pathway minimizes the accumulation of intermediates inventory, thereby lowering working capital requirements and storage costs. The use of commercially available starting materials and standard reagents further enhances supply chain reliability, mitigating the risk of shortages associated with exotic or custom-synthesized chiral auxiliaries.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, driven primarily by the elimination of stoichiometric chiral resolving agents and the associated loss of 50% of the material during resolution. By achieving high yields through catalytic asymmetric hydrogenation, the mass efficiency of the process is significantly improved, leading to lower raw material costs per kilogram of final product. Furthermore, the mild reaction conditions reduce energy consumption for heating and cooling, while the simplified workup procedures decrease solvent usage and waste disposal fees. These cumulative efficiencies result in a leaner cost structure, enabling competitive pricing strategies for high-purity pharmaceutical intermediates in a price-sensitive global market.
- Enhanced Supply Chain Reliability: Supply continuity is critical for API production, and this route enhances reliability by relying on robust, scalable chemistry. The avoidance of complex resolution steps reduces the variability often seen in crystallization processes, ensuring consistent batch quality and throughput. The use of a heterogeneous-like catalytic approach (in terms of ease of handling) with standard hydrogenation equipment means that the process can be easily transferred between manufacturing sites without extensive requalification. This flexibility allows for diversified sourcing strategies, reducing dependency on single-source suppliers for specialized chiral building blocks and strengthening the overall resilience of the pharmaceutical supply network against disruptions.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process aligns well with green chemistry principles. The high atom economy of the hydrogenation step and the reduction in solvent-intensive purification stages contribute to a lower E-factor (mass of waste per mass of product). The ability to run the key stereoselective step at ambient pressure and temperature simplifies engineering controls, making the transition from pilot scale to multi-ton commercial production smoother and safer. This scalability ensures that the method can meet growing global demand for Avacopan without requiring prohibitive capital investments in high-pressure infrastructure, while simultaneously meeting increasingly stringent environmental regulations regarding chemical waste and emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in CN114262291B, providing clarity on how this technology resolves specific pain points in Avacopan manufacturing. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their own production pipelines or for procurement officers negotiating supply contracts based on this superior methodology.
Q: How does this new method improve upon conventional Avacopan synthesis?
A: Unlike conventional methods requiring chiral resolution with ditoluoyl-L-tartaric acid which lowers yield, this patent utilizes direct asymmetric hydrogenation, achieving a final yield of 42.55% and eliminating the need for resolution steps.
Q: What represents the key catalytic system for ensuring high enantiomeric purity?
A: The process employs di(1,5-cyclooctadiene)rhodium(I) tetrafluoroborate combined with a specific chiral phosphoramidite ligand, (R)-Ligand 1, which delivers an ee value of up to 98.8% under mild conditions.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the route consists of only four steps with robust reaction conditions (e.g., 1 atm hydrogen pressure, ambient temperature for key steps), making it highly scalable and industrially practical compared to lengthier prior art methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Avacopan Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes like the one described in CN114262291B for securing the future supply of vital therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped to handle complex catalytic reactions, including asymmetric hydrogenations, with stringent purity specifications and rigorous QC labs to guarantee product integrity. We are committed to leveraging such advanced technologies to deliver high-quality intermediates that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to explore the full potential of this optimized synthesis for your supply chain needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can enhance your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a reliable, cost-effective supply of Avacopan intermediates for your upcoming projects.
