Revolutionizing Pharmaceutical Intermediate Production Through Metal-Free Isothiourea Building Block Synthesis at Commercial Scale
The groundbreaking patent CN113735752B introduces a transformative metal-free methodology for synthesizing S-aryl isothiourea compounds using substituted iodobenzenes as key substrates. This innovation addresses critical limitations in traditional C-S coupling approaches by eliminating transition metal catalysts entirely while operating under exceptionally mild conditions at room temperature. The process represents a significant advancement in building block chemistry for pharmaceutical applications where structural precision and purity are paramount. By leveraging sodium hydride as a base mediator rather than expensive or polluting metal catalysts, this method provides a sustainable pathway to access complex isothiourea structures that serve as essential intermediates in drug discovery pipelines. The patent demonstrates how strategic substrate selection enables direct nucleophilic addition without requiring air-sensitive environments or high-energy inputs that have historically constrained industrial adoption of similar transformations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing S-isothiourea compounds have relied heavily on transition metal-catalyzed C-S coupling reactions that present multiple operational challenges for industrial-scale manufacturing. These methods typically require elevated temperatures exceeding -20°C along with strong alkaline conditions that necessitate specialized equipment and stringent safety protocols due to air sensitivity concerns. The substantial catalyst loading requirements—often exceeding stoichiometric amounts—create significant downstream purification hurdles as residual metals must be rigorously removed to meet pharmaceutical quality standards. Furthermore, sulfur-containing species frequently cause irreversible deactivation of metal catalysts during reactions, leading to inconsistent yields and unpredictable batch-to-batch performance that compromise supply chain reliability. The environmental burden from metal waste streams also imposes additional processing costs and regulatory complexities that make these conventional routes increasingly unsustainable for modern pharmaceutical manufacturing operations seeking greener chemistry solutions.
The Novel Approach
The patented methodology overcomes these limitations through an elegant metal-free nucleophilic addition strategy that utilizes substituted iodobenzenes as direct coupling partners with thiourea compounds under mild conditions mediated by sodium hydride. This innovative process operates efficiently at room temperature without requiring inert atmospheres or specialized reactor configurations typically needed for transition metal systems. By eliminating metal catalysts entirely, the method avoids both catalyst deactivation issues and costly metal removal steps while delivering consistent high yields across diverse substrate combinations. The strategic use of substituted iodobenzenes provides superior raw material availability compared to alternative precursors like phenylboronic acids while maintaining excellent functional group tolerance across halogenated and alkyl-substituted variants. This approach represents a paradigm shift toward more sustainable pharmaceutical intermediate production by significantly reducing environmental impact while enhancing operational simplicity and scalability potential.
Mechanistic Insights into NaH-Mediated C-S Coupling
The core innovation lies in sodium hydride's dual role as both base and mediator in facilitating direct nucleophilic addition between substituted iodobenzenes and thiourea derivatives without transition metal involvement. Under ambient conditions in mixed THF/DMA solvent systems, NaH deprotonates thiourea to generate a highly nucleophilic thiolate species that attacks the electrophilic carbon adjacent to iodine on the aromatic ring through an SNAr mechanism. This process proceeds through a well-defined Meisenheimer complex intermediate where electron-withdrawing substituents on the iodobenzene ring significantly accelerate reaction kinetics while maintaining excellent regioselectivity even with ortho-substituted diiodobenzenes. The absence of competing side reactions typically observed in metal-catalyzed systems ensures clean conversion to single products without requiring additional purification steps that would otherwise introduce yield losses or impurity profiles problematic for pharmaceutical applications.
Impurity control is inherently achieved through the reaction's regioselective nature and simplified mechanistic pathway that avoids multiple catalytic cycles responsible for byproduct formation in conventional methods. The absence of transition metals eliminates potential heavy metal contaminants that would require extensive chromatographic purification to remove below pharmacopeial limits. Furthermore, the mild reaction conditions prevent thermal degradation pathways that could generate undesired side products during prolonged high-temperature operations common in traditional approaches. This intrinsic selectivity combined with straightforward aqueous workup procedures ensures consistent production of high-purity intermediates meeting stringent regulatory requirements without additional processing steps that would otherwise increase manufacturing complexity and cost.
How to Synthesize Isothiourea Building Blocks Efficiently
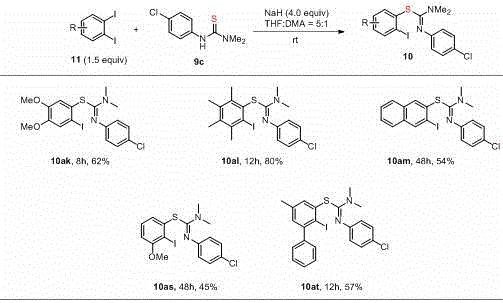
This patented methodology provides a robust framework for synthesizing complex S-isothiourea building blocks through a streamlined three-step procedure that eliminates transition metal requirements while maintaining excellent functional group tolerance across diverse substrates. The process begins with preparation of a reactive nucleophilic intermediate by deprotonating thiourea derivatives using sodium hydride under inert atmosphere before introducing substituted iodobenzene substrates at ambient temperature. Reaction monitoring via standard TLC techniques ensures precise endpoint determination without requiring specialized analytical equipment during scale-up operations. Detailed standardized synthesis protocols have been developed based on this patent's technical disclosures to enable seamless technology transfer from laboratory discovery to commercial manufacturing environments while maintaining consistent product quality attributes essential for pharmaceutical applications.
- Prepare a suspension of sodium hydride (4.0 equivalents) in anhydrous THF under nitrogen atmosphere in a reaction flask while maintaining room temperature conditions.
- Add thiourea derivative dissolved in DMA dropwise with continuous stirring for two minutes to form the reactive nucleophilic intermediate before introducing substrates.
- Introduce substituted iodobenzene (1.5 equivalents) dissolved in THF and monitor reaction progression via TLC until completion at ambient temperature.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing approach delivers substantial value across procurement and supply chain functions by addressing critical pain points associated with traditional isothiourea production methods that have historically constrained reliable sourcing of these essential pharmaceutical intermediates. The elimination of transition metal catalysts removes significant cost drivers related to catalyst procurement and subsequent heavy metal removal processes that typically require specialized equipment and generate hazardous waste streams requiring costly disposal protocols. Furthermore, the simplified reaction pathway operating under ambient conditions reduces energy consumption while enhancing operational flexibility across diverse manufacturing facilities without requiring capital-intensive reactor modifications typically needed for high-pressure or cryogenic processes common in conventional methodologies.
- Cost Reduction in Manufacturing: The complete removal of expensive transition metal catalysts eliminates both procurement costs and downstream purification expenses associated with metal residue removal from final products. This streamlined approach significantly reduces overall manufacturing complexity by avoiding multi-step workup procedures required in traditional methods while utilizing commercially available raw materials at optimal stoichiometric ratios that minimize waste generation throughout production cycles.
- Enhanced Supply Chain Reliability: The use of widely available substituted iodobenzenes as starting materials provides greater sourcing flexibility compared to specialized precursors required by alternative synthetic routes. The ambient temperature operation enables consistent production across diverse geographical locations without climate-controlled facilities while reducing sensitivity to seasonal variations that could otherwise disrupt manufacturing schedules through simplified logistics requirements.
- Scalability and Environmental Compliance: The inherently safe room temperature process facilitates seamless scale-up from laboratory validation to commercial production volumes without requiring specialized engineering controls typically needed for high-pressure or cryogenic operations. The elimination of toxic metal waste streams substantially reduces environmental compliance burdens while aligning with global sustainability initiatives through minimized energy consumption and reduced hazardous material handling requirements throughout manufacturing operations.
Frequently Asked Questions (FAQ)
The following questions address critical technical considerations raised by procurement teams evaluating this patented technology for integration into pharmaceutical supply chains. Each response draws directly from experimental data and technical disclosures within patent CN113735752B to provide evidence-based insights supporting informed sourcing decisions regarding this innovative manufacturing approach.
Q: How does this metal-free method improve upon conventional copper-catalyzed C-S coupling for isothiourea synthesis?
A: The patented process eliminates transition metal catalysts entirely by utilizing sodium hydride as a base mediator under mild room temperature conditions. This avoids catalyst deactivation issues common with sulfur-containing species in metal-catalyzed systems while preventing heavy metal contamination that complicates purification and increases waste treatment costs in traditional copper-based methods.
Q: What are the key advantages of using substituted iodobenzenes over phenylboronic acids in this synthesis?
A: Substituted iodobenzenes offer superior raw material availability compared to phenylboronic acids while enabling direct C-S coupling without ligand requirements. The process demonstrates excellent functional group tolerance across diverse substituents including halogens and alkyl groups, providing broader synthetic flexibility for complex pharmaceutical intermediate production without air-sensitive reagents.
Q: How does the process ensure high purity and minimal impurities in final isothiourea products?
A: The metal-free mechanism prevents transition metal residues that typically create challenging purification hurdles. The regioselective nature of the nucleophilic addition reaction with ortho-substituted diiodobenzenes ensures single-product formation without requiring additional separation steps. This inherent selectivity combined with straightforward workup procedures delivers consistently high-purity intermediates suitable for stringent pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isothiourea Compound Supplier
Our company leverages this patented technology to deliver exceptional value through extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required by global regulatory authorities. NINGBO INNO PHARMCHEM's state-of-the-art manufacturing facilities incorporate rigorous QC labs that ensure consistent product quality through comprehensive analytical testing protocols specifically designed for complex heterocyclic intermediates like isothioureas. Our technical team possesses deep expertise in optimizing this metal-free synthesis route to meet exacting customer requirements while providing robust supply chain solutions that guarantee uninterrupted delivery of mission-critical pharmaceutical building blocks.
We invite procurement teams to request a Customized Cost-Saving Analysis demonstrating how this innovative process can reduce your total cost of ownership through simplified manufacturing workflows and reduced waste streams. Contact our technical procurement team today to obtain specific COA data and comprehensive route feasibility assessments tailored to your unique production requirements.
