Advanced Synthesis of 3-Cyano-N-Oxidoisoxazoline Compounds for Pharmaceutical Applications
The pharmaceutical and fine chemical industries continuously seek robust methodologies for constructing complex heterocyclic scaffolds, particularly those serving as versatile building blocks for bioactive molecules. Patent CN113149923B introduces a significant advancement in this domain by disclosing a novel synthetic route for 3-cyano-N-oxidized isoxazoline compounds. These specific heterocycles are invaluable intermediates in organic synthesis, frequently utilized in the construction of amino alcohols, amino acids, and various natural product analogues. The innovation lies in the utilization of copper nitrate trihydrate as a nitrogen-oxygen source within a dimethyl sulfoxide solvent system. This approach represents a paradigm shift from traditional methods that often rely on hazardous reagents or multi-step sequences. By streamlining the synthesis into a direct rearrangement protocol, the technology offers a pathway to high-purity intermediates with improved atom economy. 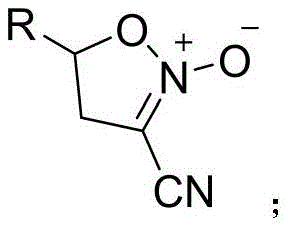
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-oxidized isoxazolines has been fraught with significant chemical and operational challenges that hinder efficient large-scale production. Early methodologies, such as those reported by Holy et al. in 1976, relied on the reaction of nitroalkenes with methylating reagents under basic conditions, which often resulted in limited substrate scope and moderate yields. Subsequent developments, like the 1,3-dipolar cycloaddition described by Loffe, required the use of alpha-nitroalkyl bromides and silylating agents, leading to prolonged reaction times and the generation of stoichiometric silicon waste. Furthermore, the rearrangement of nitrocyclopropanes reported by Dailey necessitated the use of highly toxic diazo compounds as precursors, posing severe safety risks and environmental burdens. These legacy processes typically involve harsh conditions, expensive catalysts, or difficult-to-remove impurities, making them less attractive for modern green chemistry standards and cost-sensitive manufacturing environments.
The Novel Approach
In stark contrast to these cumbersome historical precedents, the method disclosed in CN113149923B offers a remarkably streamlined and environmentally benign alternative. The core of this innovation is the direct rearrangement of 1,1-dicyanocyclopropane derivatives using copper nitrate trihydrate in DMSO. This catalytic system operates under mild thermal conditions, typically between 60°C and 70°C, which significantly reduces energy consumption compared to high-temperature pyrolysis methods. The reaction proceeds efficiently under a nitrogen atmosphere, ensuring stability and reproducibility. Crucially, this protocol avoids the use of toxic diazo precursors entirely, substituting them with readily available dicyanocyclopropanes. The workup procedure is equally simplified, involving standard extraction and chromatography techniques that facilitate easy isolation of the target molecule. 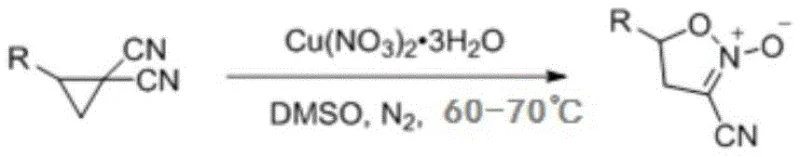
Mechanistic Insights into Copper-Catalyzed Rearrangement
The mechanistic pathway of this transformation involves a sophisticated ring-expansion rearrangement driven by the copper species and the polar aprotic solvent. The copper nitrate trihydrate serves a dual function, acting both as a Lewis acid to activate the cyclopropane ring and as the source of the nitrogen-oxygen moiety required for the isoxazoline N-oxide formation. In the presence of DMSO, the coordination environment around the copper center likely facilitates the cleavage of the strained cyclopropane bond, allowing for the insertion of the nitro group and subsequent cyclization. This intramolecular process is highly regioselective, favoring the formation of the five-membered isoxazoline ring over potential competing pathways. The mild conditions prevent the decomposition of sensitive functional groups on the aromatic rings, such as halogens or esters, which might degrade under more aggressive acidic or basic conditions found in older literature methods. This selectivity is paramount for maintaining the integrity of complex molecular architectures required in late-stage pharmaceutical synthesis.
From an impurity control perspective, the mechanism inherently limits the formation of byproducts commonly associated with radical-based rearrangements. The use of a defined copper salt minimizes the risk of heavy metal contamination that can occur with heterogeneous catalysts, while the homogeneous nature of the reaction in DMSO ensures uniform heat and mass transfer. This uniformity is critical for preventing localized hot spots that could lead to polymerization or degradation of the nitrile groups. Consequently, the crude reaction mixture typically exhibits a cleaner profile, reducing the burden on downstream purification units. For R&D teams, understanding this mechanism allows for precise tuning of reaction parameters, such as the molar ratio of copper to substrate, to maximize conversion while minimizing residual metal content. This level of control is essential for meeting the stringent regulatory requirements of API intermediate manufacturing.
How to Synthesize 3-Cyano-N-Oxidoisoxazoline Efficiently
The practical execution of this synthesis is designed for operational simplicity, making it accessible for both laboratory research and pilot plant operations. The process begins with the precise weighing of the 1,1-dicyanocyclopropane substrate and the copper nitrate trihydrate oxidant, which are then introduced into a dry reaction vessel containing dimethyl sulfoxide. Maintaining an inert nitrogen atmosphere is critical during the heating phase to prevent oxidative side reactions that could compromise yield. Once the reaction reaches completion, indicated by the disappearance of the starting material via TLC monitoring, the mixture is cooled to ambient temperature to halt the catalytic cycle. The subsequent workup involves liquid-liquid extraction using dichloromethane, followed by washing with saturated brine to remove residual DMSO and inorganic salts. Finally, the crude solid is subjected to column chromatography using a petroleum ether and ethyl acetate gradient to afford the pure product. Detailed standardized synthesis steps are provided in the guide below.
- Combine 1,1-dicyanocyclopropane and copper nitrate trihydrate in dry DMSO under nitrogen at 60-70°C.
- Cool the reaction mixture, extract with dichloromethane, wash with brine, and dry to obtain the crude product.
- Purify the crude material via column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical efficiency. The primary advantage lies in the substantial simplification of the raw material supply chain. By utilizing 1,1-dicyanocyclopropanes and commodity copper salts, manufacturers can avoid the volatility and high costs associated with specialized diazo reagents or complex organometallic catalysts. This shift to widely available feedstocks enhances supply security and mitigates the risk of production stoppages due to raw material shortages. Furthermore, the elimination of toxic precursors significantly reduces the regulatory burden and costs related to hazardous waste disposal and worker safety compliance. The mild reaction conditions also translate to lower energy expenditures, contributing to a reduced carbon footprint and aligning with corporate sustainability goals.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive, bulk-available reagents like copper nitrate trihydrate and DMSO. Unlike previous methods that required expensive silylating agents or precious metal catalysts, this protocol relies on base metals that are cost-effective even at large scales. The high yields observed across various substrates mean that less raw material is wasted, directly improving the overall material cost per kilogram of the final intermediate. Additionally, the simplified workup reduces solvent consumption and processing time, further driving down operational expenses without compromising quality.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical factor for long-term production planning. The starting materials for this reaction are chemically stable and can be sourced from multiple global suppliers, reducing dependency on single-source vendors. The robustness of the reaction conditions ensures consistent output quality, minimizing the need for re-processing or batch rejection. This reliability allows supply chain planners to maintain leaner inventory levels while ensuring continuous availability of key intermediates for downstream drug synthesis, thereby optimizing working capital.
- Scalability and Environmental Compliance: The transition from bench scale to commercial production is facilitated by the homogeneous nature of the reaction and the absence of exothermic hazards associated with diazo compounds. The process generates minimal hazardous waste, simplifying effluent treatment and ensuring compliance with increasingly strict environmental regulations. The ability to run the reaction at moderate temperatures (60-70°C) reduces the demand on cooling and heating infrastructure, making it easier to scale up in existing multipurpose reactors without major capital investment in specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation. Understanding these aspects is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the practical realities of scaling this chemistry while maintaining high standards of quality and safety.
Q: What are the primary advantages of this copper-catalyzed method over traditional synthesis routes?
A: This method eliminates the need for toxic diazo precursors and harsh reaction conditions found in older protocols. It utilizes inexpensive copper nitrate and operates at mild temperatures (60-70°C), significantly improving operational safety and reducing waste treatment costs.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process uses common solvents like DMSO and dichloromethane and avoids sensitive reagents. The mild thermal requirements and straightforward workup procedure make it highly suitable for commercial scale-up from kilogram to multi-ton quantities.
Q: What is the typical purity profile of the resulting isoxazoline intermediates?
A: The reaction demonstrates high selectivity, yielding products that can be purified to stringent specifications via standard column chromatography. The absence of complex side reactions ensures a clean impurity profile suitable for downstream pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Cyano-N-Oxidoisoxazoline Supplier
As the demand for high-quality heterocyclic intermediates continues to grow, partnering with an experienced CDMO is essential for navigating the complexities of process development and scale-up. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our technical team is well-versed in the nuances of copper-catalyzed rearrangements and can optimize this specific patent technology to meet your stringent purity specifications. With our rigorous QC labs and state-of-the-art manufacturing facilities, we guarantee that every batch of 3-cyano-N-oxidoisoxazoline delivered meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you secure a reliable supply chain for your critical pharmaceutical intermediates while maximizing efficiency and minimizing costs.
