Revolutionizing Chiral Intermediate Production with Aqueous Polyionic Liquid Copper Catalysts
Introduction to Green Asymmetric Catalysis
The landscape of fine chemical manufacturing is undergoing a paradigm shift towards sustainability, driven by the urgent need to reduce volatile organic compound (VOC) emissions and enhance process safety. Patent CN111285769B introduces a groundbreaking methodology for the asymmetric Henry addition reaction, utilizing a novel polyionic liquid-type chiral amino acid copper catalyst, designated as P(IL-A)n-Cu. This innovation addresses the longstanding challenge of performing high-efficiency asymmetric catalysis in pure aqueous media, a feat previously hindered by poor solubility and mass transfer limitations of traditional metal complexes. By leveraging the unique amphiphilic properties of polyionic liquids, this technology enables the synthesis of chiral beta-nitroalcohol compounds—a critical class of pharmaceutical intermediates—with remarkable enantioselectivity and yield. For R&D directors and procurement strategists, this represents a significant opportunity to transition from hazardous organic solvent-based processes to greener, water-based alternatives without compromising on product quality or stereochemical purity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the Henry reaction, which couples nitroalkanes with aldehydes to form beta-nitroalcohols, has been predominantly conducted in organic solvents such as dichloromethane, THF, or ethanol. While effective, these conventional methods suffer from inherent drawbacks that impact both operational costs and environmental compliance. The primary issue lies in the solubility mismatch; many chiral metal-salen or amino acid-based catalysts exhibit poor solubility in water, leading to heterogeneous reaction conditions that severely limit mass transfer and reaction kinetics. Furthermore, the reliance on volatile organic solvents necessitates complex downstream processing, including energy-intensive distillation and solvent recovery systems, which inflate production costs and increase the carbon footprint. Additionally, separating the catalyst from the product often requires chromatographic purification, which is impractical for large-scale commercial manufacturing. These factors collectively create a bottleneck for the cost reduction in pharmaceutical intermediate manufacturing, particularly when scaling up processes that require stringent control over impurity profiles.
The Novel Approach
The methodology described in CN111285769B circumvents these obstacles by employing a polyionic liquid architecture that acts as a "nanoreactor" within the aqueous phase. The P(IL-A)n-Cu catalyst is designed with a hydrophilic polyionic liquid backbone and hydrophobic chiral ligand sites, granting it dual solubility in both water and organic substrates. This unique property creates a homogeneous microenvironment where the catalyst, water, and organic reactants interact freely, drastically increasing collision frequency and accelerating reaction rates. Unlike traditional systems that precipitate or aggregate in water, this polymeric catalyst remains stable and active, facilitating efficient asymmetric induction. Moreover, the system allows for a simple phase-transfer workup; upon completion, the addition of a non-polar solvent like n-hexane causes the catalyst to separate cleanly from the aqueous product phase. This eliminates the need for complex filtration or chromatography, streamlining the workflow and enabling the effective recovery and reuse of the expensive chiral catalyst, thereby enhancing the overall economic viability of the process.
Mechanistic Insights into Polyionic Liquid Copper Catalysis
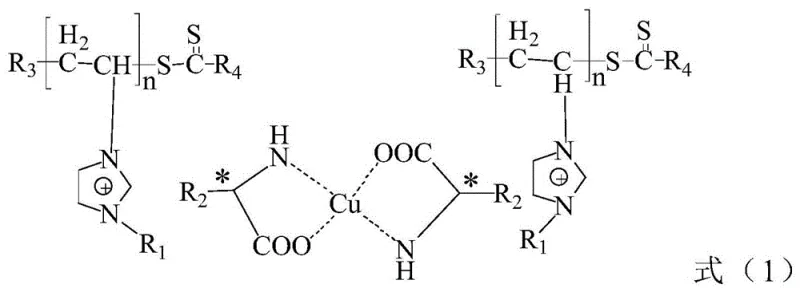
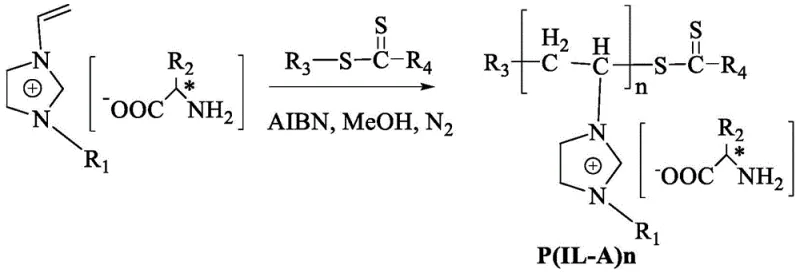
The efficacy of the P(IL-A)n-Cu system stems from its sophisticated molecular design, which integrates controlled radical polymerization with coordination chemistry. The synthesis begins with the preparation of a vinyl-functionalized chiral ionic liquid monomer, derived from natural chiral amino acids. This monomer undergoes Reversible Addition-Fragmentation Chain Transfer (RAFT) polymerization, a technique that ensures precise control over molecular weight and polydispersity. The resulting polymer chain serves as a robust scaffold that prevents the leaching of metal ions while maintaining the accessibility of the active sites. Subsequent coordination with copper salts, such as copper acetate, generates the active catalytic species where the copper ion is chelated by the amino acid moieties pendant from the polymer backbone. This structural integrity is crucial for maintaining high enantioselectivity, as the rigid chiral environment around the copper center dictates the stereochemical outcome of the nucleophilic attack.
Furthermore, the mechanism involves a dynamic interplay between the hydrophobic effect and hydrogen bonding networks within the aqueous medium. The polyionic liquid chains tend to aggregate into nano-spherical particles in water, as evidenced by characterization data, creating localized hydrophobic pockets that concentrate the organic reactants near the catalytic centers. This "concentration effect" mimics enzymatic catalysis, significantly boosting reaction efficiency even at low catalyst loadings. The presence of the ionic liquid groups also enhances the stability of the transition state through electrostatic interactions, further lowering the activation energy barrier. For technical teams, understanding this mechanism is vital for optimizing reaction parameters such as pH, temperature, and ionic strength to maximize turnover numbers. The ability to tune the polymer side chains (R1, R2, R3, R4) offers additional flexibility to tailor the catalyst for specific substrate classes, ensuring broad applicability across diverse synthetic routes for high-purity pharmaceutical intermediates.
How to Synthesize Chiral Beta-Nitroalcohols Efficiently
The practical implementation of this technology involves a straightforward protocol that aligns well with existing reactor infrastructure. The process initiates with the dissolution of the pre-synthesized P(IL-A)n-Cu catalyst in deionized water, followed by the sequential addition of the aldehyde substrate and the nitroalkane reagent. The reaction proceeds under mild thermal conditions, typically between 0°C and 20°C, which minimizes energy consumption and reduces the risk of thermal runaway. Detailed standardized synthesis steps, including precise molar ratios, stirring speeds, and workup procedures, are outlined in the technical guide below to ensure reproducibility and optimal yield.
- Preparation of the polyionic liquid precursor via RAFT polymerization of vinyl-functionalized chiral amino acid ionic liquids.
- Coordination of the polymer with copper salts (e.g., copper acetate) to form the active P(IL-A)n-Cu catalyst complex.
- Execution of the Henry reaction in pure water at mild temperatures (0-20°C), followed by phase-transfer separation for catalyst recovery.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this aqueous catalytic system offers profound advantages that extend beyond mere technical performance. The shift to water as the primary reaction medium fundamentally alters the cost structure of manufacturing chiral intermediates. By eliminating the need for large volumes of expensive, flammable, and toxic organic solvents, facilities can significantly reduce raw material expenditures and lower the costs associated with solvent storage, handling, and disposal. This transition also mitigates regulatory risks related to VOC emissions and workplace safety, ensuring long-term operational continuity in increasingly regulated markets. For supply chain heads, the simplicity of the catalyst recovery process translates to reduced lead times and higher throughput, as the bottlenecks associated with solvent removal and catalyst purification are effectively removed from the production timeline.
- Cost Reduction in Manufacturing: The economic benefits are driven primarily by the drastic simplification of the downstream processing workflow. Traditional methods often require multiple extraction steps, drying agents, and column chromatography to remove metal residues and achieve the necessary purity levels. In contrast, the phase-separation capability of the P(IL-A)n-Cu catalyst allows for a clean split between the product and the catalyst phase. This reduction in unit operations directly lowers utility costs (steam, electricity) and labor hours. Additionally, the high reusability of the polymer-supported catalyst means that the effective cost per kilogram of product decreases with each cycle, providing a sustainable path for margin improvement in competitive generic drug markets.
- Enhanced Supply Chain Reliability: Relying on water as a solvent enhances supply security by removing dependence on petrochemical-derived solvents, whose prices can be volatile and subject to geopolitical disruptions. The robustness of the polyionic liquid catalyst also ensures consistent batch-to-batch quality, reducing the incidence of failed batches that can disrupt supply schedules. The mild reaction conditions further contribute to equipment longevity and reduced maintenance downtime. For procurement managers, this reliability is paramount when securing long-term contracts for critical API intermediates, as it guarantees a steady flow of material without the unpredictability associated with complex, solvent-heavy chemistries.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often reveals hidden challenges related to heat transfer and mixing, especially in heterogeneous systems. The homogeneous nature of this aqueous system at the microscopic level facilitates excellent heat dissipation and mixing, making the commercial scale-up of complex pharmaceutical intermediates significantly safer and more predictable. Furthermore, the generation of aqueous waste streams is far easier and cheaper to treat than halogenated organic waste. This alignment with green chemistry principles not only improves the corporate sustainability profile but also future-proofs the manufacturing site against tightening environmental regulations, ensuring uninterrupted operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of polyionic liquid catalysts in industrial settings. These insights are derived directly from the experimental data and mechanistic studies presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this technology into their existing portfolios.
Q: How does the polyionic liquid catalyst improve reaction efficiency compared to traditional methods?
A: Unlike traditional catalysts that struggle with mass transfer in water, the P(IL-A)n-Cu catalyst is amphiphilic. It dissolves in both the aqueous phase and the organic substrate, significantly increasing collision frequency and reaction rates without needing toxic organic solvents.
Q: Can the catalyst be reused effectively in industrial settings?
A: Yes. The patent demonstrates that the catalyst can be recovered simply by adding an organic solvent like n-hexane to induce phase separation. This allows for effective recycling and reuse, maintaining high catalytic activity over multiple cycles.
Q: What level of enantioselectivity can be achieved with this aqueous system?
A: The technology achieves exceptional stereocontrol, with experimental data showing enantiomeric excess (ee) values up to 99% for various substituted benzaldehydes, rivaling or exceeding traditional organic-phase protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Beta-Nitroalcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient catalytic technologies is essential for the future of the pharmaceutical industry. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries like the P(IL-A)n-Cu system can be successfully translated into robust manufacturing processes. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, utilizing our rigorous QC labs to verify enantiomeric excess and impurity profiles for every batch. Our infrastructure is designed to handle complex asymmetric syntheses with the precision and reliability that global partners demand.
We invite you to collaborate with us to leverage these advanced catalytic methods for your specific project needs. Whether you are looking to optimize an existing route or develop a new process from scratch, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can drive value for your supply chain.
