Unlocking Superior Catalytic Activity with Novel Bis(Amphos) Tetrabromo Dipalladium Complexes for Pharmaceutical Synthesis
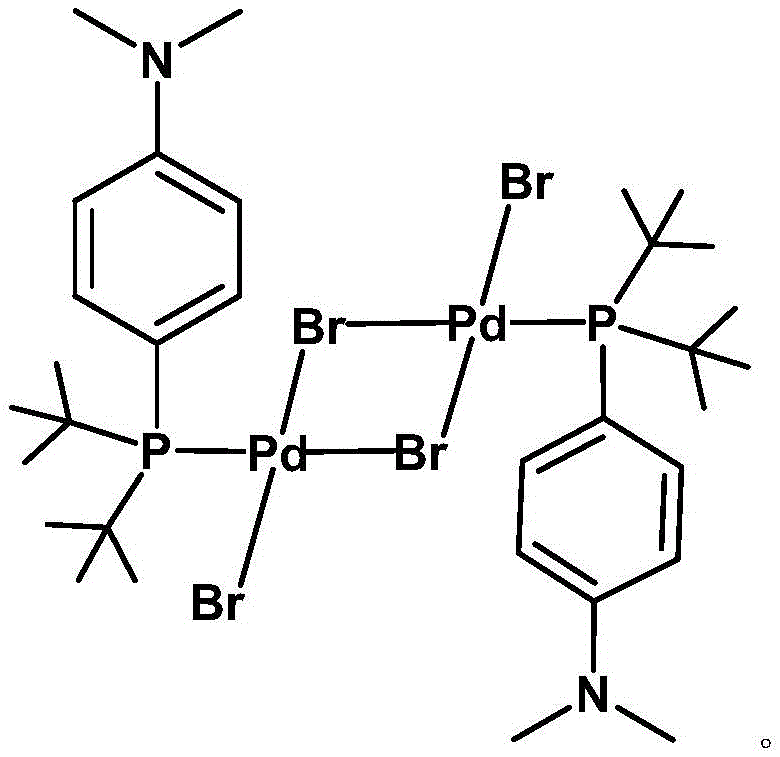
The landscape of transition metal catalysis is undergoing a significant transformation with the introduction of novel binuclear palladium complexes designed to overcome the inherent limitations of traditional mononuclear systems. As detailed in patent CN111909217A, a groundbreaking class of bis(di-tert-butyl-4-dimethylaminophenylphosphine)tetrabromobispalladium(II) compounds has emerged, offering a robust alternative for constructing complex organic frameworks. This innovation addresses critical pain points in the synthesis of pharmaceutical intermediates and agrochemical active ingredients, where catalyst stability and turnover frequency are paramount. The core breakthrough lies in the strategic replacement of chloride ligands with bromide ions within a binuclear architecture, creating a unique [Pd2Br2] four-membered ring structure that fundamentally alters the kinetics of the catalytic cycle. For R&D directors and process chemists, this represents a tangible opportunity to enhance reaction efficiency without compromising on the rigorous purity standards required for GMP manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
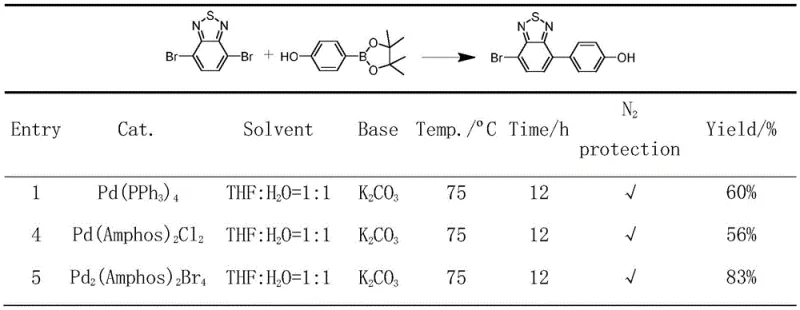
Historically, the industry has relied heavily on tetrakis(triphenylphosphine)palladium [Pd(PPh3)4] as the workhorse for Suzuki-Miyaura cross-coupling reactions. However, this Pd(0) species suffers from notorious instability, rapidly decomposing upon exposure to atmospheric oxygen, which necessitates stringent inert gas protocols and complicates logistics for large-scale operations. Furthermore, conventional mononuclear Pd(II) catalysts utilizing chloride ligands, such as [Pd(Amphos)2Cl2], often encounter kinetic bottlenecks where the dissociation of the chloride ion becomes the rate-determining step, sluggishness that limits overall throughput. These legacy systems frequently require elevated temperatures or excessive catalyst loading to achieve acceptable conversion rates, leading to increased production costs and challenging downstream purification processes due to residual metal contamination. The inability of these traditional catalysts to maintain high activity in sterically hindered or electron-deficient substrates further restricts their utility in synthesizing advanced functional materials and complex drug candidates.
The Novel Approach
The novel binuclear palladium complex described in the patent data introduces a paradigm shift by leveraging the distinct electronic and steric properties of the Amphos ligand combined with bridging bromide atoms. By forming a stable [Pd2Br2] core, the catalyst achieves a delicate balance between thermodynamic stability and kinetic lability. Experimental data demonstrates that this new architecture delivers a conversion rate of 83% in model Suzuki coupling reactions, significantly outperforming the 60% yield of Pd(PPh3)4 and the 56% yield of the mononuclear chloride analogue under identical conditions. This performance leap is attributed to the faster dissociation kinetics of the Pd-Br bond compared to Pd-Cl, facilitating quicker entry into the active catalytic cycle. The result is a highly efficient system that operates effectively at moderate temperatures, reducing energy consumption and minimizing side reactions that typically plague less selective catalytic systems.
Mechanistic Insights into Binuclear Pd-Br Catalysis
The superior performance of this catalyst can be mechanistically traced to the unique coordination environment surrounding the palladium centers. In the catalytic cycle, the initial activation step involves the dissociation of a ligand to generate the coordinatively unsaturated active species. While the Pd-Br bond is thermodynamically stronger than the Pd-Cl bond, the specific geometry of the binuclear [Pd2Br2] ring lowers the activation energy for bromide dissociation kinetically. This counter-intuitive phenomenon allows the catalyst to remain stable during storage yet become highly reactive under process conditions. The bulky tert-butyl groups on the phosphine ligands provide a protective cone that prevents the formation of inactive palladium black aggregates, a common deactivation pathway in homogeneous catalysis. This steric bulk also enhances selectivity by favoring the desired cross-coupling pathway over homocoupling side reactions, ensuring a cleaner impurity profile for the final API intermediate.
Structural analysis confirms that the palladium atoms adopt a planar tetracoordinate geometry, with the Amphos ligands and bromine atoms occupying the remaining coordination sites in a precise arrangement. This rigid structural framework prevents ligand scrambling and ensures consistent catalytic behavior batch after batch. The electron-rich nature of the dimethylamino group on the phenyl ring further donates electron density to the phosphorus and subsequently to the palladium center, facilitating the oxidative addition step which is often critical for activating aryl bromides and chlorides. For process chemists, understanding this electronic modulation is key to predicting substrate scope and optimizing reaction parameters for diverse chemical libraries. The combination of electronic enrichment and steric protection creates a catalyst that is both robust and highly tunable for specific synthetic challenges.
How to Synthesize Bis(di-tert-butyl-4-dimethylaminophenylphosphine)tetrabromobispalladium(II) Efficiently
The preparation of this high-performance catalyst is remarkably straightforward, utilizing readily available starting materials and standard laboratory equipment, which facilitates seamless technology transfer from R&D to pilot plant scales. The synthesis avoids exotic reagents or extreme conditions, relying instead on controlled reflux in acetonitrile to ensure complete dissolution of the palladium source prior to ligand coordination. This simplicity is a major advantage for supply chain managers, as it reduces the dependency on specialized precursors that might face availability constraints. The process yields an orange-red powder with greater than 98% isolated yield and purity exceeding 99% based on palladium content, eliminating the need for complex chromatographic purification steps that would otherwise drive up manufacturing costs. Detailed standardized operating procedures for this synthesis are outlined below to ensure reproducibility and safety.
- Dissolve palladium bromide (PdBr2) in acetonitrile solvent by heating to reflux at 80°C for 3-4 hours under nitrogen protection until a clear dark red solution forms.
- Cool the solution to room temperature and slowly add di-tert-butyl-4-dimethylaminophenylphosphine (Amphos) dropwise while maintaining an inert nitrogen atmosphere.
- Stir the reaction mixture for 2 hours to precipitate the orange-red product, then filter, wash with acetonitrile, and dry to obtain the high-purity catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel catalyst offers compelling economic and operational benefits that extend beyond simple reaction yields. The enhanced stability of the Pd(II) binuclear complex compared to air-sensitive Pd(0) alternatives translates directly into reduced waste and lower inventory shrinkage, as the material maintains its potency over extended storage periods without requiring specialized cold chain logistics. This robustness simplifies warehouse management and reduces the risk of batch failures due to catalyst degradation, ensuring consistent production schedules and reliable delivery timelines for downstream customers. Furthermore, the higher catalytic activity means that lower loadings may be achievable in optimized processes, directly reducing the bill of materials cost per kilogram of finished product while simultaneously lowering the burden on metal scavenging units during purification.
- Cost Reduction in Manufacturing: The elimination of expensive and unstable Pd(0) precursors significantly lowers raw material procurement costs, while the high turnover number reduces the total amount of precious metal required per batch. By avoiding the need for rigorous exclusion of oxygen during storage and handling, facilities can save substantially on inert gas consumption and specialized containment equipment. The simplified downstream processing, resulting from fewer side products and easier metal removal, further drives down operational expenditures associated with solvent recovery and waste treatment. These cumulative efficiencies create a leaner manufacturing process that improves overall margin potential for high-volume API production.
- Enhanced Supply Chain Reliability: The synthesis relies on commodity chemicals like palladium bromide and acetonitrile, which are widely available from multiple global suppliers, mitigating the risk of single-source bottlenecks. The robust nature of the final catalyst product ensures that it can withstand standard shipping conditions without degradation, guaranteeing that the material arrives at the manufacturing site with full activity. This reliability is crucial for maintaining continuous operation in just-in-time manufacturing environments where catalyst failure could lead to costly production stoppages. Additionally, the scalability of the preparation method ensures that supply can be rapidly ramped up to meet surging demand without compromising on quality or purity specifications.
- Scalability and Environmental Compliance: The preparation method generates minimal hazardous waste, as the primary byproduct is easily managed and the solvent system is amenable to recycling and reuse. The high atom economy of the catalytic reaction itself aligns with green chemistry principles, reducing the environmental footprint of the synthesis process. This compliance with increasingly stringent environmental regulations future-proofs the manufacturing process against potential regulatory changes. The ability to run reactions at moderate temperatures also reduces energy consumption, contributing to a more sustainable and cost-effective production lifecycle that appeals to environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this binuclear palladium catalyst in industrial settings. These insights are derived directly from the patent specifications and experimental data to provide accurate guidance for process development teams. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing production lines.
Q: How does the binuclear structure of Pd2(Amphos)2Br4 improve catalytic activity compared to mononuclear analogues?
A: The binuclear structure features a unique [Pd2Br2] four-membered ring where bridging bromine atoms facilitate faster kinetic dissociation compared to chloride bridges, accelerating the rate-determining step in the catalytic cycle.
Q: What are the stability advantages of this Pd(II) catalyst over traditional Pd(0) catalysts like Pd(PPh3)4?
A: Unlike air-sensitive Pd(0) species that degrade rapidly upon exposure, this Pd(II) complex exhibits exceptional thermal and oxidative stability, allowing for easier handling, longer shelf life, and reduced storage costs.
Q: Can this catalyst be used for large-scale Suzuki coupling reactions in API manufacturing?
A: Yes, the synthesis method utilizes common solvents like acetonitrile and achieves yields greater than 98%, making it highly suitable for commercial scale-up and robust process chemistry applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis(Amphos) Palladium Catalyst Supplier
As the demand for high-efficiency coupling catalysts grows in the pharmaceutical and agrochemical sectors, NINGBO INNO PHARMCHEM stands ready to support your production needs with premium-grade catalytic solutions. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistent quality. We maintain stringent purity specifications and operate rigorous QC labs to verify that every batch of catalyst meets the exacting standards required for GMP synthesis. Our technical team is well-versed in the nuances of organometallic chemistry and can provide expert guidance on integrating this novel binuclear system into your specific process workflows.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current manufacturing challenges. By partnering with us, you gain access to specific COA data and comprehensive route feasibility assessments that demonstrate the tangible value of switching to this advanced catalyst system. Let us help you optimize your synthesis strategy, reduce your cost of goods sold, and accelerate your time to market with reliable, high-performance chemical solutions.
