Advanced Manufacturing of 8-Hydroxyjulolidine Derivatives for High-Performance Laser Dyes
Introduction to Advanced 8-Hydroxyjulolidine Manufacturing
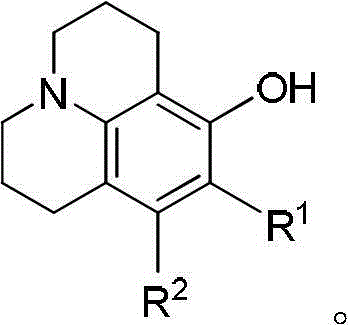
The demand for high-performance fluorescent materials in the fields of biotechnology and optoelectronics has driven significant innovation in the synthesis of rigidified amino-phenol scaffolds. Specifically, 8-hydroxyjulolidine and its derivatives serve as critical precursors for coumarin-based laser dyes and fluorescent probes, offering superior quantum yields and wavelength tunability due to their restricted molecular rotation. As detailed in the groundbreaking patent CN102731498B, a novel preparation method has been developed that overcomes the historical limitations of low yield and hazardous reagent usage associated with traditional synthetic routes. This technology represents a paradigm shift in the manufacturing of reliable electronic chemical intermediates, providing a pathway to high-purity products essential for sensitive bioassay applications. By implementing a controlled two-step alkylation strategy, manufacturers can now achieve substantial improvements in atom economy while adhering to stricter environmental regulations regarding toxic waste disposal.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
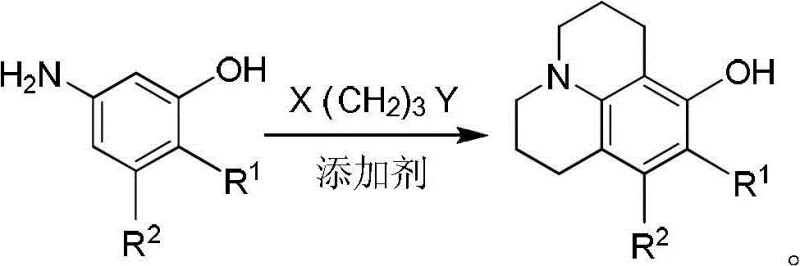
Historically, the synthesis of 8-hydroxyjulolidine has been plagued by inefficient multi-step sequences or low-yielding one-pot reactions that are economically unsustainable for large-scale production. Traditional approaches, such as those cited in US patents 4005092 and 4471041, rely on a protection-deprotection strategy involving the methylation of 3-aminophenol using toxic dimethyl sulfate, followed by cyclization and subsequent harsh demethylation with corrosive acids like hydroiodic acid or boron tribromide. These methods not only introduce significant safety hazards due to the handling of carcinogenic methylating agents but also generate substantial acidic waste streams that require costly neutralization and treatment. Furthermore, attempts to streamline this process into a single step, as seen in US6433176, have historically failed to gain traction due to the prevalence of competitive intermolecular side reactions. In these one-step scenarios, the dialkylating agent preferentially reacts with two molecules of the amine rather than cyclizing, leading to the formation of 1,3-disubstituted propane byproducts and resulting in target product yields of less than 5%, which is commercially unacceptable for a reliable agrochemical intermediate supplier or pharma partner.
The Novel Approach
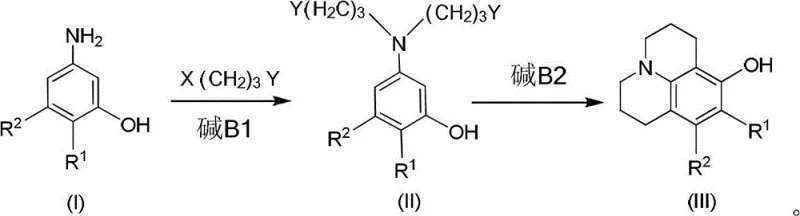
In stark contrast to these legacy methods, the innovative process disclosed in CN102731498B decouples the alkylation and cyclization events into two distinct, controlled stages, thereby effectively suppressing the formation of bis-alkylated impurities. This novel approach utilizes an excess of the dihaloalkane linker, such as 1-bromo-3-chloropropane, in the first stage to ensure that the primary amine of the 3-aminophenol substrate undergoes mono-alkylation exclusively, forming a stable linear intermediate. By isolating or monitoring this intermediate before proceeding to the second stage, the process prevents the statistical probability of double substitution that plagues the one-pot methods. The subsequent intramolecular cyclization is then triggered under specific thermal conditions with a secondary base addition, forcing the remaining halide to react with the phenolic oxygen to close the ring. This strategic separation of reaction phases not only boosts the yield of the target high-purity OLED material precursor significantly but also eliminates the need for hazardous methyl protection groups, thereby simplifying the workflow and reducing the overall environmental footprint of the manufacturing process.
Mechanistic Insights into Controlled Stepwise Cyclization
The core mechanistic advantage of this synthesis lies in the kinetic control of nucleophilic substitution reactions, leveraging the difference in nucleophilicity between the aromatic amine and the phenolic hydroxyl group. In the first intermolecular reaction stage, the primary amine acts as a stronger nucleophile than the phenol, attacking the dihaloalkane linker to form a secondary amine intermediate. The use of a molar excess of the linker, typically in a ratio greater than 2:1 relative to the aminophenol, ensures that the concentration of the electrophile remains high enough to favor mono-substitution over the cross-linking of two amine molecules. This step is typically conducted at moderate temperatures between 40-80°C in polar aprotic solvents like DMF or DMSO, which stabilize the transition state without promoting premature elimination reactions. The addition of a mild base such as sodium bicarbonate or triethylamine serves to scavenge the hydrogen halide byproduct, driving the equilibrium forward without causing unwanted side reactions on the sensitive phenolic ring.
Following the formation of the linear intermediate, the second stage involves an intramolecular nucleophilic attack by the phenolic oxygen on the remaining terminal halide of the propyl chain. This cyclization step requires slightly more vigorous conditions, typically heating to 50-120°C, to overcome the entropic barrier of ring closure. The presence of a second base equivalent is crucial here to deprotonate the phenol, generating a phenoxide ion that is sufficiently nucleophilic to displace the halide and form the ether linkage characteristic of the julolidine core. Crucially, the entire process is preferably conducted under an inert atmosphere, such as nitrogen or argon, to prevent the oxidation of the electron-rich aminophenol substrate and the intermediate, which could otherwise lead to colored impurities and reduced product quality. This meticulous control over reaction parameters ensures that the final commercial scale-up of complex polymer additives or dye precursors maintains a consistent and high-quality impurity profile.
How to Synthesize 8-Hydroxyjulolidine Efficiently
The implementation of this two-step synthetic route offers a robust framework for laboratory and pilot-scale production, providing clear operational milestones that ensure reproducibility and safety. The process begins with the careful selection of solvents and bases that are compatible with large-scale handling, avoiding the extreme hazards associated with traditional demethylation reagents. Operators are advised to monitor the conversion of the starting aminophenol closely using thin-layer chromatography (TLC) to determine the precise endpoint of the first alkylation stage, ensuring complete consumption of the starting material before initiating the cyclization. This attention to detail in the intermediate stage is the key differentiator that allows for the high yields reported in the patent examples, ranging from 50% to over 80% depending on the specific substrate and conditions employed. For a comprehensive understanding of the specific stoichiometric ratios, temperature profiles, and workup procedures required to replicate these results, please refer to the standardized synthesis guide provided below.
- Dissolve the 3-aminophenol compound and excess dihalopropane (X(CH2)3Y) in an organic solvent like DMF, add a base such as sodium bicarbonate, and stir under inert gas at 40-80°C until the starting material is fully converted to the mono-alkylated intermediate.
- Isolate the intermediate through aqueous workup and extraction, removing the solvent to prepare the crude material for the subsequent cyclization step without further purification if purity allows.
- Dissolve the isolated intermediate in a fresh organic solvent, add a second equivalent of base, and heat the mixture to 50-120°C under nitrogen protection to facilitate the intramolecular ring closure, followed by purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the adoption of this novel synthesis method offers transformative benefits that directly impact the bottom line and operational resilience of chemical manufacturing operations. By eliminating the need for specialized corrosion-resistant equipment required for handling strong mineral acids like hydroiodic acid, facilities can utilize standard glass-lined or stainless steel reactors, representing a significant capital expenditure saving. Furthermore, the removal of toxic methylating agents from the supply chain reduces the regulatory burden and insurance costs associated with storing and transporting hazardous Schedule 1 chemicals, thereby streamlining the logistics of raw material acquisition. The simplified workup procedure, which relies on standard aqueous extraction and crystallization rather than complex distillation or hazardous quenching steps, also translates to reduced cycle times and lower energy consumption per kilogram of product produced. These factors collectively contribute to a more agile and cost-effective supply chain capable of responding rapidly to market fluctuations in the demand for laser dye intermediates.
- Cost Reduction in Manufacturing: The economic viability of this process is largely driven by the elimination of expensive and hazardous reagents such as dimethyl sulfate and boron tribromide, which not only carry high purchase prices but also incur significant disposal costs. By utilizing commodity chemicals like 1-bromo-3-chloropropane and common inorganic bases, the raw material cost profile is drastically optimized, allowing for more competitive pricing in the global market. Additionally, the improved yield efficiency means that less starting material is wasted on byproducts, effectively lowering the cost of goods sold (COGS) and maximizing the output from existing reactor capacity without the need for immediate infrastructure expansion.
- Enhanced Supply Chain Reliability: Sourcing high-purity 3-aminophenol and simple dihaloalkanes is significantly more reliable than securing specialized demethylation agents, which are often subject to strict regulatory controls and supply volatility. This shift to widely available feedstocks mitigates the risk of production stoppages due to raw material shortages, ensuring a continuous flow of intermediates to downstream customers in the pharmaceutical and electronic sectors. Moreover, the robustness of the reaction conditions, which tolerate a range of temperatures and solvents, provides manufacturing flexibility that allows producers to adapt quickly to regional availability of solvents like DMF or DMSO, further strengthening supply chain continuity.
- Scalability and Environmental Compliance: The process is inherently designed for green chemistry principles, avoiding the generation of heavy metal waste or persistent organic pollutants that complicate wastewater treatment. The absence of strong acid waste streams simplifies the effluent treatment process, reducing the environmental compliance costs and facilitating easier permitting for facility expansions. This environmental compatibility makes the technology highly scalable, allowing manufacturers to increase production volumes from pilot batches to multi-ton campaigns with minimal additional investment in waste management infrastructure, aligning perfectly with the sustainability goals of modern chemical enterprises.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this technology into their existing portfolios, we have compiled answers to common inquiries regarding the reaction specifics and scalability. These insights are derived directly from the experimental data and technical disclosures within the patent literature, ensuring that the information provided is accurate and actionable for process development scientists. Understanding the nuances of the intermediate isolation and the specific role of the inert atmosphere is critical for successfully transferring this chemistry from the laboratory bench to the production floor. We encourage potential partners to review these details carefully to appreciate the depth of optimization involved in this novel synthetic route.
Q: Why does the conventional one-step synthesis of 8-hydroxyjulolidine result in low yields?
A: Conventional one-step methods often suffer from severe intermolecular side reactions, specifically the formation of 1,3-disubstituted propane byproducts, which drastically reduces the yield of the target cyclic product to less than 5% in many cases.
Q: What are the safety advantages of this new preparation method compared to traditional routes?
A: This novel method eliminates the need for highly toxic methylating agents like dimethyl sulfate and avoids the use of strong corrosive acids such as hydroiodic acid or boron tribromide required for demethylation in older processes, significantly improving operational safety and environmental compliance.
Q: Can this synthesis process be scaled for industrial production of laser dye intermediates?
A: Yes, the process is designed for industrialization as it utilizes commercially available solvents and bases, operates at moderate temperatures, and avoids complex protection-deprotection sequences, making it highly suitable for commercial scale-up of complex fine chemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 8-Hydroxyjulolidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation fluorescent probes and laser dyes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 8-hydroxyjulolidine meets the exacting standards required for sensitive optoelectronic and bioassay applications. Our state-of-the-art facilities are equipped to handle the specific solvent and temperature requirements of this two-step cyclization process, delivering consistent quality that supports your R&D and manufacturing timelines.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your supply chain efficiency and reduce your overall production costs.
