Scalable Visible-Light Mediated Oxidative Decarboxylation for Commercial Aryl Aldehyde Production
The pharmaceutical and fine chemical industries are constantly seeking sustainable methodologies to construct high-value building blocks, and the recent disclosure in patent CN110963900A presents a transformative approach to synthesizing aryl aldehyde compounds. This intellectual property details a novel oxidative decarboxylation strategy that leverages visible light photocatalysis to convert readily available aryl acetic acids directly into valuable aryl aldehydes. Unlike traditional thermal processes that often demand harsh conditions, this technology operates efficiently at room temperature under an open air atmosphere, utilizing molecular oxygen as the sole oxidant. For R&D directors and process chemists, this represents a significant paradigm shift towards greener synthesis, eliminating the reliance on stoichiometric heavy metal oxidants and transition metal catalysts that plague conventional routes. The method employs 2,4,5,6-tetrakis(9-carbazolyl)-isophthalonitrile (4CzIPN) as a robust organic photocatalyst, which, when paired with a specific organic base, facilitates a smooth single-electron transfer process. This innovation not only simplifies the operational workflow but also aligns perfectly with modern regulatory demands for reduced heavy metal residues in active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl aldehydes from aryl acetic acids has been fraught with significant chemical and economic challenges that hinder large-scale manufacturing efficiency. Conventional protocols typically necessitate the use of transition metal catalysts, such as palladium or copper complexes, which introduce severe complications regarding product purity and downstream processing costs. Furthermore, these traditional oxidative decarboxylation reactions often require stoichiometric amounts of strong chemical oxidants, leading to the generation of substantial quantities of inorganic waste salts that are environmentally burdensome and expensive to dispose of. The requirement for elevated temperatures and inert atmospheres in many of these legacy methods further exacerbates energy consumption and operational complexity, making them less attractive for cost-sensitive commercial production. Additionally, the presence of residual heavy metals in the final product often mandates rigorous and costly purification steps, such as scavenging or recrystallization, to meet the stringent purity specifications required by global health authorities. These cumulative factors create a bottleneck in the supply chain, increasing lead times and reducing the overall sustainability profile of the manufacturing process for these critical intermediates.
The Novel Approach
In stark contrast to these legacy issues, the technology described in CN110963900A introduces a streamlined, metal-free pathway that utilizes the abundant energy of visible light to drive the transformation. By employing 4CzIPN as a metal-free organic photocatalyst and DBU as a proton-transfer co-catalyst, the reaction proceeds smoothly under mild conditions without the need for external heating or inert gas protection. The use of air as the oxidant is a particularly brilliant aspect of this design, as it replaces hazardous chemical oxidants with a free, inexhaustible resource, thereby drastically simplifying the reagent profile and waste stream. This method achieves high yields, often exceeding 80-90% for electron-rich substrates, demonstrating that green chemistry principles do not have to come at the expense of reaction efficiency. The operational simplicity of stirring the reaction mixture under blue LED irradiation at room temperature makes this process highly amenable to scale-up in flow chemistry reactors or large batch vessels. Ultimately, this approach offers a reliable aryl aldehyde supplier with a distinct competitive advantage by merging high performance with environmental stewardship.
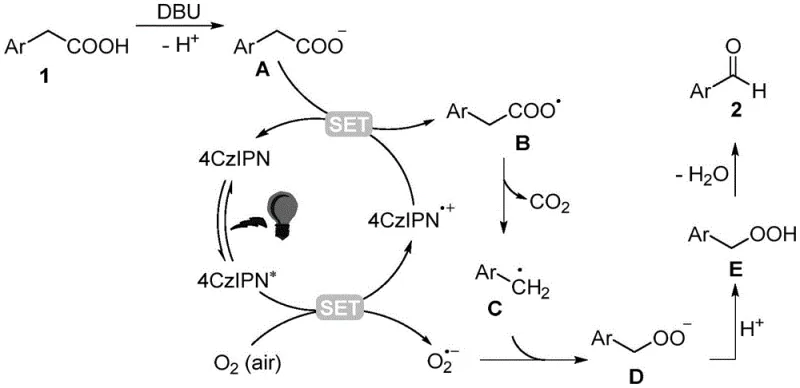
Mechanistic Insights into 4CzIPN-Catalyzed Oxidative Decarboxylation
To fully appreciate the robustness of this synthetic route, one must delve into the intricate photoredox catalytic cycle that drives the conversion of aryl acetic acids to aldehydes. The process initiates when the 4CzIPN photocatalyst absorbs photons from the blue LED source, promoting it to an excited state capable of engaging in single-electron transfer (SET) events. In the presence of the organic base DBU, the aryl acetic acid substrate is deprotonated to form a carboxylate anion, which then interacts with the excited photocatalyst. This interaction triggers an oxidative quenching cycle where the carboxylate donates an electron to the excited catalyst, generating a carboxyl radical intermediate and the reduced form of the photocatalyst. This carboxyl radical is inherently unstable and rapidly undergoes decarboxylation to release carbon dioxide, yielding a reactive benzyl radical species that is pivotal for the subsequent oxidation steps. The reduced photocatalyst then transfers an electron to molecular oxygen dissolved in the reaction medium, regenerating the ground-state catalyst and producing a superoxide radical anion, thus closing the catalytic loop.
The fate of the benzyl radical determines the success of the aldehyde formation, as it must be selectively oxidized without over-oxidation to the corresponding carboxylic acid. The generated superoxide radical anion couples with the benzyl radical to form a peroxide intermediate, which subsequently undergoes protonation and dehydration to furnish the target aryl aldehyde product. This mechanistic pathway is highly advantageous for impurity control because the mild reaction conditions prevent the thermal degradation of sensitive functional groups that might occur under harsher thermal oxidation regimes. The specificity of the radical coupling ensures that side reactions are minimized, leading to a cleaner crude reaction profile that simplifies downstream purification. Understanding this mechanism allows process chemists to fine-tune parameters such as light intensity and oxygen flow to maximize throughput while maintaining the integrity of complex molecular architectures. The elegance of this cycle lies in its atom economy and the seamless integration of renewable energy inputs to drive chemical bond formation.
How to Synthesize Aryl Aldehydes Efficiently
Implementing this photocatalytic protocol in a laboratory or pilot plant setting requires careful attention to the specific reaction parameters outlined in the patent data to ensure reproducibility and high yield. The standard procedure involves dissolving the aryl acetic acid substrate in acetonitrile, which has been identified as the optimal solvent for stabilizing the radical intermediates and solubilizing the organic catalyst. A precise molar ratio of the 4CzIPN catalyst and DBU base is added to the solution, and the mixture is subjected to irradiation from blue LEDs with a wavelength range of 400nm to 480nm. The reaction is conducted in an open vessel to allow continuous exchange with atmospheric oxygen, which is consumed stoichiometrically during the oxidation process. While the general concept is straightforward, the detailed标准化 synthesis steps见下方的指南 ensure that operators can replicate the high yields reported in the optimization studies, avoiding common pitfalls related to light penetration or oxygen mass transfer limitations.
- Prepare the reaction mixture by combining aryl acetic acid, 4CzIPN photocatalyst, and DBU base in acetonitrile solvent.
- Expose the reaction vessel to blue LED light (400-480nm) while stirring under an open air atmosphere at room temperature.
- Monitor the reaction progress and isolate the final aryl aldehyde product via silica gel column chromatography after completion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology translates into tangible strategic benefits that extend far beyond simple chemical curiosity. The elimination of transition metal catalysts removes a major cost center associated with both the purchase of precious metals and the extensive validation required to prove their removal from the final API. By switching to an organic photocatalyst like 4CzIPN, manufacturers can achieve significant cost reduction in pharmaceutical intermediates manufacturing through simplified supply chains and reduced dependency on volatile metal markets. Furthermore, the use of air as the oxidant eliminates the need to procure, store, and handle hazardous chemical oxidants, thereby lowering safety compliance costs and insurance premiums associated with dangerous goods. The mild room temperature conditions also imply a drastic reduction in energy consumption compared to thermal processes that require heating to reflux, contributing to lower utility bills and a smaller carbon footprint for the production facility.
- Cost Reduction in Manufacturing: The removal of expensive transition metals and stoichiometric oxidants fundamentally alters the cost structure of producing aryl aldehydes, allowing for more competitive pricing in the global market. Without the need for specialized metal scavenging resins or complex filtration systems to remove trace palladium or copper, the downstream processing becomes significantly cheaper and faster. This streamlined workflow reduces the overall manufacturing cycle time, enabling facilities to turn over batches more quickly and respond agilely to market demand fluctuations. Additionally, the high atom efficiency of using oxygen from air means that fewer raw materials are wasted as byproducts, maximizing the value derived from every kilogram of starting material purchased.
- Enhanced Supply Chain Reliability: Relying on abundant and non-toxic reagents like air and organic bases mitigates the risk of supply disruptions that often plague the sourcing of specialized metal catalysts. The robustness of the reaction conditions ensures that production can continue consistently without the sensitivity to moisture or oxygen levels that characterizes many organometallic processes. This stability is crucial for maintaining continuous supply to downstream customers, reducing the likelihood of stockouts caused by failed batches or extended purification times. Moreover, the use of common solvents like acetonitrile ensures that raw material procurement remains straightforward and unaffected by niche supply chain bottlenecks.
- Scalability and Environmental Compliance: The green nature of this synthesis aligns perfectly with increasingly stringent environmental regulations, reducing the burden of waste treatment and disposal permits. Scaling this reaction is facilitated by the availability of high-power LED arrays and flow chemistry setups that can handle large volumes of gas-liquid mixtures efficiently. The absence of heavy metal waste streams simplifies the environmental impact assessment and lowers the cost of wastewater treatment, making the process economically viable even in regions with strict ecological standards. This future-proofs the manufacturing asset against tightening regulatory landscapes, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this photocatalytic oxidative decarboxylation technology. These insights are derived directly from the experimental data and optimization studies presented in the patent documentation to assist technical teams in evaluating feasibility. Understanding these nuances is critical for successful technology transfer and process validation.
Q: What are the primary advantages of this photocatalytic method over traditional transition metal catalysis?
A: This method eliminates the need for expensive and toxic transition metal catalysts such as palladium or copper, thereby removing the costly and complex heavy metal removal steps required in downstream processing. Additionally, it utilizes molecular oxygen from air as the terminal oxidant instead of hazardous stoichiometric chemical oxidants, significantly reducing waste salt generation and environmental impact.
Q: Can this synthesis protocol accommodate diverse substituent groups on the aromatic ring?
A: Yes, the protocol demonstrates excellent substrate scope, successfully converting aryl acetic acids with electron-donating groups like methoxy and alkyl, as well as electron-withdrawing halogens, into their corresponding aldehydes with high efficiency. The reaction is particularly effective for para-substituted substrates, yielding up to 90% conversion for electron-rich systems.
Q: Is the reaction sensitive to atmospheric conditions or light wavelength?
A: The reaction strictly requires an aerobic environment as molecular oxygen acts as the essential oxidant in the catalytic cycle; inert atmospheres like nitrogen inhibit the process. Furthermore, optimal efficiency is achieved specifically under blue light irradiation (400-480nm), whereas red or green light sources fail to provide sufficient energy to excite the 4CzIPN photocatalyst effectively.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Aldehydes Supplier
As the global demand for high-quality pharmaceutical intermediates continues to rise, partnering with a manufacturer that possesses advanced synthetic capabilities is essential for maintaining a competitive edge. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging cutting-edge technologies like the visible-light mediated oxidative decarboxylation described in CN110963900A to deliver superior products. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of aryl aldehydes meets the exacting standards required for drug substance manufacturing. Our commitment to green chemistry and process innovation allows us to offer solutions that are not only chemically robust but also economically and environmentally sustainable.
We invite you to engage with our technical procurement team to discuss how this novel synthetic route can be integrated into your supply chain to drive efficiency and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential financial benefits of switching to this metal-free methodology for your specific projects. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique molecular targets. Let us collaborate to build a more resilient and efficient supply chain for the next generation of life-saving medicines.
