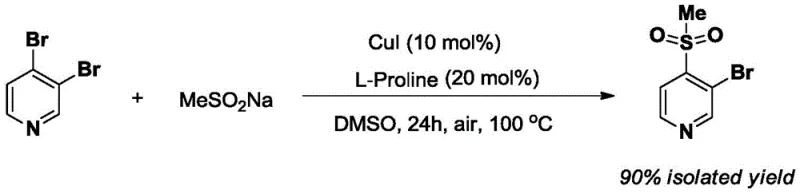
Revolutionizing Pyridine Sulfonation: A High-Yield Route for Pharmaceutical Intermediates
The landscape of fine chemical manufacturing is constantly evolving, driven by the urgent need for more efficient, sustainable, and high-yielding synthetic routes for critical pharmaceutical intermediates. A groundbreaking development in this sector is documented in Chinese Patent CN110407742B, which discloses a highly innovative method for preparing 3-bromo-4-methanesulfonylpyridine. This compound serves as a vital building block in the synthesis of various bioactive molecules and therapeutic agents, making its production efficiency a matter of significant industrial interest. The patent details a transformative one-step protocol that utilizes 3,4-dibromopyridine and sodium methanesulfinate as key starting materials, mediated by a robust copper catalytic system. By operating in dimethyl sulfoxide at an elevated temperature of 100 °C for 24 hours, this methodology achieves an exceptional isolated yield of 90%, marking a substantial leap forward compared to conventional multi-step approaches. For R&D directors and procurement specialists alike, this technology represents a paradigm shift towards streamlined manufacturing that minimizes waste and maximizes output.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-bromo-4-methanesulfonylpyridine has been plagued by inefficiencies inherent in multi-step sequences that rely on harsh reagents and offer suboptimal yields. As highlighted in the background art of the patent, prior methods, such as those referenced in PCT international application 2016180537, typically necessitate a two-stage process involving initial thiolation followed by a separate oxidation step. In the first stage, 3,4-dibromopyridine reacts with sodium methyl mercaptide to generate a thioether intermediate, which must then be isolated or processed further. The second stage involves the oxidation of this 4-methylthio-3-bromopyridine using strong oxidizing agents like m-chloroperoxybenzoic acid (m-CPBA). This traditional pathway suffers from severe drawbacks, including the generation of stoichiometric amounts of chlorobenzoic acid waste, safety hazards associated with peroxide handling, and critically, a very low yield in the oxidation step, reported to be merely 41 percent. These factors collectively result in high production costs, complex purification requirements, and a significant environmental footprint, rendering the conventional route less attractive for modern, green chemistry-compliant manufacturing facilities.
The Novel Approach
In stark contrast to the cumbersome legacy protocols, the novel approach disclosed in CN110407742B introduces a direct, catalytic C-S bond formation strategy that bypasses the need for discrete thiolation and oxidation stages. This method leverages the synergistic effect of Copper(I) Iodide (CuI) as the catalyst and L-Proline as a bidentate ligand to facilitate the direct coupling of the sulfinate salt with the bromopyridine substrate. By employing sodium methanesulfinate directly, the process installs the sulfonyl group in a single operation, effectively merging what were previously two distinct chemical transformations into one seamless reaction. The operational simplicity is further enhanced by the use of DMSO as a polar aprotic solvent, which stabilizes the ionic intermediates and allows the reaction to proceed smoothly at 100 °C under aerobic conditions. This consolidation of steps not only drastically reduces the total processing time and labor but also eliminates the accumulation of intermediates that often lead to yield losses, thereby delivering the target molecule with a remarkable 90% isolated yield.
Mechanistic Insights into CuI/L-Proline Catalyzed Sulfination
The success of this transformation hinges on the sophisticated interplay between the copper catalyst and the amino acid ligand, which together create a highly active catalytic species capable of activating the carbon-halogen bond. Mechanistically, the reaction is believed to proceed through a Cu(I)/Cu(III) catalytic cycle where the L-Proline ligand coordinates to the copper center, enhancing its solubility and stability in the DMSO medium. The sodium methanesulfinate acts as the nucleophilic sulfur source, undergoing transmetallation or coordination with the activated copper complex. Subsequently, the oxidative addition of the 3,4-dibromopyridine into the copper-sulfur bond occurs, preferentially at the 4-position due to electronic and steric factors dictated by the pyridine nitrogen and the adjacent 3-bromo substituent. This regioselectivity is crucial, as it ensures the preservation of the 3-bromo handle, which is often required for subsequent cross-coupling reactions in downstream drug synthesis. The final reductive elimination step releases the desired 3-bromo-4-methanesulfonylpyridine and regenerates the active Cu(I) catalyst, allowing the cycle to continue with high turnover efficiency.
Furthermore, the choice of L-Proline as a ligand plays a pivotal role in suppressing side reactions and controlling the impurity profile of the final product. In many transition-metal catalyzed processes, the formation of homocoupling byproducts or desulfinated species can compromise purity, necessitating expensive chromatographic purification. However, the specific coordination environment provided by the proline ligand appears to favor the desired cross-coupling pathway, minimizing the formation of bis-sulfonated byproducts or debrominated impurities. This high level of chemoselectivity is particularly valuable for pharmaceutical applications where strict impurity thresholds must be met. The reaction conditions, specifically the use of air rather than an inert atmosphere, suggest that the catalytic system is robust enough to tolerate oxygen, which simplifies the engineering requirements for scale-up. This mechanistic robustness translates directly into process reliability, ensuring that batch-to-batch consistency can be maintained even when transitioning from laboratory glassware to industrial-scale reactors.
How to Synthesize 3-Bromo-4-Methanesulfonylpyridine Efficiently
Implementing this synthesis in a production environment requires careful attention to reagent quality and thermal control to maximize the benefits of the patented protocol. The procedure is designed to be operationally simple, requiring standard equipment capable of heating to 100 °C and handling polar solvents. The molar ratios specified in the patent, specifically a 1:1.2 ratio of dibromopyridine to sulfinate salt, ensure complete conversion of the valuable halide substrate while keeping reagent costs in check. Detailed standardized operating procedures regarding mixing speeds, addition rates, and workup protocols are essential for reproducibility. For a comprehensive guide on the exact execution of this synthesis, please refer to the step-by-step instructions provided below.
- Charge a pressure-resistant reactor with dimethyl sulfoxide (DMSO) as the solvent system.
- Sequentially add 3,4-dibromopyridine, sodium methanesulfinate, CuI catalyst (10 mol%), and L-Proline ligand (20 mol%).
- Maintain vigorous stirring at 100 °C for 24 hours under air atmosphere, followed by concentration and chromatographic purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers compelling economic and logistical benefits that extend far beyond simple yield improvements. The transition from a two-step sequence involving hazardous oxidants to a single-step catalytic process fundamentally alters the cost structure of manufacturing this key intermediate. By eliminating the need for m-CPBA, a relatively expensive and potentially dangerous reagent, and removing an entire unit operation (the oxidation step), the process significantly reduces raw material consumption and utility usage. Furthermore, the simplification of the workflow means reduced labor hours and lower equipment occupancy time, allowing manufacturing facilities to increase throughput without capital expansion. These efficiencies culminate in a drastic reduction in the cost of goods sold (COGS), providing a competitive edge in pricing strategies for downstream API production.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the elimination of the low-yielding oxidation step which previously wasted over half of the intermediate material. By achieving a 90% yield in a single pot, the effective consumption of the starting 3,4-dibromopyridine is nearly halved compared to the cumulative yield of the traditional two-step method. Additionally, the replacement of stoichiometric oxidants with a catalytic amount of inexpensive copper salts and proline removes a major cost driver and waste disposal burden. The avoidance of chlorobenzoic acid byproducts further lowers waste treatment costs, contributing to substantial overall cost savings in pharma intermediate manufacturing.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this one-pot reaction enhances reliability by reducing the number of potential failure points in the production line. Traditional multi-step syntheses are vulnerable to delays at each isolation and purification stage; consolidating these into a single reaction minimizes the risk of bottlenecks. Moreover, the raw materials required—3,4-dibromopyridine and sodium methanesulfinate—are commodity chemicals with stable global supply chains, reducing the risk of raw material shortages. The ability to run the reaction under air without stringent inert gas protection further simplifies facility requirements, making it easier to qualify multiple manufacturing sites and ensure continuous supply continuity for clients.
- Scalability and Environmental Compliance: The scalability of this process is supported by the use of DMSO, a high-boiling solvent that is well-suited for large-scale batch operations, and moderate reaction temperatures that do not require specialized cryogenic or high-pressure equipment. The environmental profile is significantly improved by adhering to green chemistry principles, specifically atom economy and waste prevention. By avoiding the generation of heavy metal waste from stoichiometric oxidants and reducing solvent usage through telescoped processing, the method aligns with increasingly stringent environmental regulations. This compliance reduces the regulatory burden on manufacturers and ensures long-term viability of the supply chain in a tightening regulatory landscape.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new manufacturing route is essential for stakeholders evaluating its integration into their supply networks. The following questions address common inquiries regarding the practical implementation, safety, and quality aspects of this copper-catalyzed sulfination technology. These answers are derived directly from the experimental data and technical disclosures found within the patent literature to ensure accuracy and relevance for technical decision-makers.
Q: What are the primary advantages of this Cu-catalyzed method over traditional oxidation routes?
A: This method eliminates the need for hazardous oxidants like m-CPBA and reduces the process from two steps to a single direct sulfination, significantly improving overall yield from roughly 41% in legacy steps to 90%.
Q: Is the catalyst system cost-effective for large-scale production?
A: Yes, the use of inexpensive CuI (10 mol%) and L-Proline (20 mol%) in DMSO offers a highly economical alternative to precious metal catalysts, facilitating easier downstream processing and cost reduction.
Q: What is the regioselectivity profile of this reaction on the pyridine ring?
A: The reaction demonstrates excellent regioselectivity, specifically targeting the 4-position of the 3,4-dibromopyridine scaffold to install the methanesulfonyl group while retaining the 3-bromo substituent.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Bromo-4-Methanesulfonylpyridine Supplier
As the demand for high-quality pyridine derivatives continues to grow within the pharmaceutical and agrochemical sectors, partnering with an experienced CDMO partner is critical for securing a stable supply of key intermediates. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the clinical trial phase or full commercial launch. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 3-bromo-4-methanesulfonylpyridine meets the highest standards required for GMP manufacturing. We understand that consistency is key, and our process engineers are dedicated to optimizing reaction parameters to maintain the high yields and selectivity demonstrated in the patent data.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your current procurement volumes, demonstrating exactly how switching to this efficient route can improve your bottom line. Please contact our technical procurement team today to request specific COA data for our pilot batches and to discuss route feasibility assessments for your downstream applications. Let us help you streamline your supply chain and accelerate your time to market with our superior manufacturing capabilities.
