Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Production
Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Production
The 2,3-dihydroquinolone scaffold represents a privileged structure in medicinal chemistry, serving as the core backbone for numerous bioactive molecules with potent therapeutic properties. As illustrated in the structural diversity of known pharmacophores, these compounds exhibit significant antitumor activity against human cell lines and possess powerful analgesic capabilities, making them highly sought-after targets for drug discovery programs. 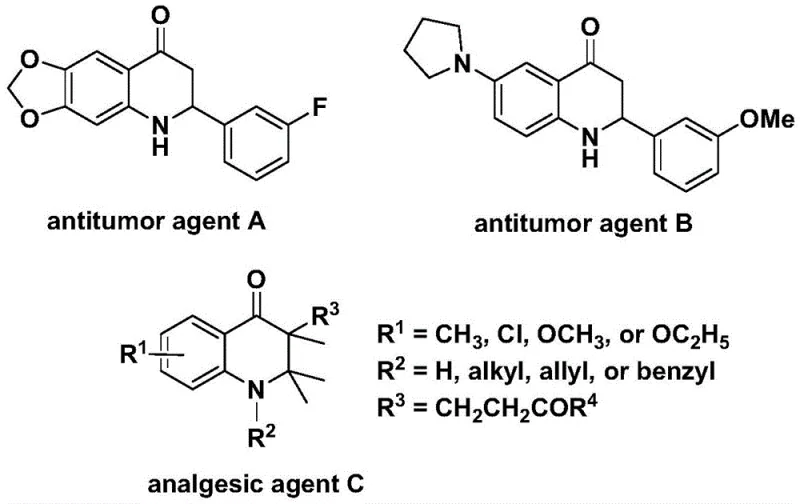 . Recognizing the critical demand for efficient access to these heterocycles, recent intellectual property disclosures, specifically patent CN112239456B, have introduced a transformative preparation method. This novel approach leverages a transition metal palladium-catalyzed carbonylation reaction, utilizing N-pyridine sulfonyl-o-iodoaniline and various olefins as starting materials. By replacing hazardous gaseous carbon monoxide with a solid surrogate, this technology not only addresses significant safety concerns but also streamlines the synthetic workflow, offering a robust pathway for the reliable pharmaceutical intermediate supplier seeking to optimize their production capabilities.
. Recognizing the critical demand for efficient access to these heterocycles, recent intellectual property disclosures, specifically patent CN112239456B, have introduced a transformative preparation method. This novel approach leverages a transition metal palladium-catalyzed carbonylation reaction, utilizing N-pyridine sulfonyl-o-iodoaniline and various olefins as starting materials. By replacing hazardous gaseous carbon monoxide with a solid surrogate, this technology not only addresses significant safety concerns but also streamlines the synthetic workflow, offering a robust pathway for the reliable pharmaceutical intermediate supplier seeking to optimize their production capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 2,3-dihydroquinolone skeleton via carbonylation has been fraught with substantial operational challenges and safety hazards that hinder widespread industrial adoption. Traditional protocols typically rely on the direct use of carbon monoxide gas, which is highly toxic, flammable, and requires specialized high-pressure equipment and rigorous safety infrastructure to handle safely. Furthermore, conventional catalytic systems often suffer from limited substrate scope, struggling to accommodate sterically hindered olefins or sensitive functional groups without significant degradation in yield or selectivity. The reliance on harsh reaction conditions and expensive, air-sensitive catalysts frequently leads to complex purification processes and increased waste generation, driving up the overall cost of goods. These factors collectively create a bottleneck for procurement managers and supply chain heads who require consistent, high-volume supplies of high-purity OLED material or API precursors without the logistical burden of managing hazardous gas supplies.
The Novel Approach
In stark contrast to legacy techniques, the methodology disclosed in CN112239456B introduces a paradigm shift by employing 1,3,5-trimesic acid phenol ester as a solid, stable carbon monoxide substitute. This strategic substitution eliminates the need for high-pressure gas cylinders, thereby drastically simplifying the reactor setup and enhancing workplace safety. The reaction operates under relatively mild thermal conditions, typically between 100-120 °C, using a palladium bis(acetylacetonate) catalyst system paired with a dppp ligand. This new route demonstrates exceptional versatility, capable of synthesizing both 2-aryl and 3-alkyl substituted derivatives through simple substrate design modifications. The use of readily available starting materials, such as N-pyridine sulfonyl-o-iodoaniline and commercial olefins, ensures a streamlined supply chain. Moreover, the process exhibits high atom economy and functional group tolerance, allowing for the direct introduction of diverse substituents like halogens, alkyl chains, and silyl groups, which is crucial for the rapid iteration required in modern drug discovery.
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cyclization
The catalytic cycle underpinning this transformation is a sophisticated sequence of organometallic steps that ensures high efficiency and selectivity. The reaction initiates with the oxidative addition of the palladium(0) species into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate, generating a reactive aryl-palladium intermediate. Subsequently, carbon monoxide, which is released in situ from the thermal decomposition of the 1,3,5-trimesic acid phenol ester, inserts into the palladium-carbon bond to form an acyl-palladium species. This step is critical as it avoids the handling of external CO gas while maintaining the necessary concentration of the carbonyl source at the catalytic center. Following this, the olefin substrate coordinates to the metal center and undergoes migratory insertion into the acyl-palladium bond, creating a new carbon-carbon bond and extending the molecular framework. 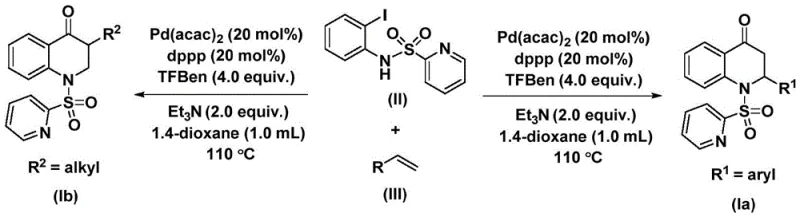 . The cycle concludes with an intramolecular reductive elimination that closes the six-membered heterocyclic ring, releasing the final substituted 2,3-dihydroquinolone product and regenerating the active palladium catalyst for the next turnover.
. The cycle concludes with an intramolecular reductive elimination that closes the six-membered heterocyclic ring, releasing the final substituted 2,3-dihydroquinolone product and regenerating the active palladium catalyst for the next turnover.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or high-temperature thermal cyclizations. The well-defined coordination sphere of the palladium catalyst directs the regioselectivity of the olefin insertion, minimizing the formation of branched isomers or polymerization byproducts that often plague free-radical processes. The use of triethylamine as an additive helps to neutralize acidic byproducts generated during the reaction, preventing catalyst deactivation and ensuring a cleaner reaction profile. Furthermore, the choice of dioxane as the preferred organic solvent facilitates the solubility of both the polar sulfonamide starting materials and the non-polar olefins, promoting homogeneous catalysis which is essential for consistent batch-to-batch reproducibility. This precise control over the reaction pathway translates directly to a simplified downstream processing workflow, where standard silica gel chromatography is sufficient to achieve the stringent purity specifications required for pharmaceutical applications.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
The practical execution of this synthesis is designed for scalability and ease of operation, making it highly attractive for process chemistry teams aiming to transition from milligram-scale discovery to kilogram-scale production. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, solid CO source, base, and substrates in an aprotic solvent, followed by heating to promote the carbonylative cyclization. The robustness of the system allows for a wide range of olefin inputs, from simple styrenes to complex aliphatic alkenes, yielding diverse products such as compounds I-1 through I-5 with good to excellent yields. 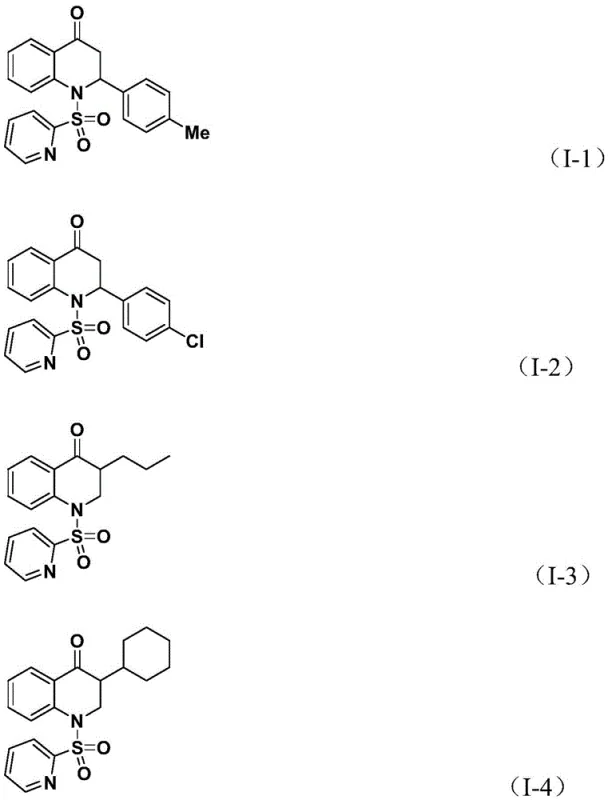 . For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below which outlines the exact molar ratios and temperature profiles optimized in the patent examples.
. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below which outlines the exact molar ratios and temperature profiles optimized in the patent examples.
- Combine palladium bis(acetylacetonate), dppp ligand, triethylamine, 1,3,5-trimesic acid phenol ester, N-pyridine sulfonyl-o-iodoaniline, and olefin in an organic solvent such as dioxane.
- Heat the reaction mixture to a temperature between 100-120 °C and maintain stirring for a duration of 24 to 48 hours to ensure complete conversion.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the target substituted 2,3-dihydroquinolone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers compelling strategic benefits that extend beyond mere chemical novelty. The primary value proposition lies in the drastic simplification of the manufacturing infrastructure; by eliminating the requirement for high-pressure carbon monoxide gas lines and specialized containment systems, facilities can repurpose existing standard glass-lined reactors for this chemistry. This flexibility significantly reduces capital expenditure barriers and accelerates the timeline for technology transfer from R&D to commercial manufacturing. Additionally, the reliance on shelf-stable solid reagents rather than compressed gases mitigates supply chain risks associated with the transportation and storage of hazardous materials, ensuring greater continuity of supply even in regions with strict regulatory oversight on toxic gases.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the utilization of inexpensive, commercially available starting materials and the avoidance of costly safety infrastructure. The solid carbon monoxide surrogate, 1,3,5-trimesic acid phenol ester, is a stable commodity chemical that eliminates the need for specialized gas delivery contracts and monitoring equipment. Furthermore, the high reaction efficiency and broad substrate tolerance reduce the need for extensive optimization campaigns for each new derivative, lowering the overall R&D burn rate. The simplified post-treatment process, which relies on standard filtration and chromatography rather than complex distillation or crystallization steps to remove metal residues, further contributes to substantial cost savings in the final isolation of the active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of robust, non-hazardous reagents that are not subject to the same stringent shipping restrictions as toxic gases. The ability to source key components like palladium catalysts and phosphine ligands from multiple global vendors ensures that production is not bottlenecked by single-source dependencies. Moreover, the reaction's tolerance to various functional groups means that a single platform technology can be used to produce a wide library of intermediates, allowing manufacturers to respond rapidly to changing market demands without retooling entire production lines. This agility is critical for maintaining competitive lead times in the fast-paced pharmaceutical sector.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) standpoint, this method aligns perfectly with modern green chemistry principles by reducing the risk of accidental releases and minimizing waste generation. The absence of high-pressure gas operations lowers the facility's risk profile, potentially reducing insurance premiums and regulatory compliance costs. The process generates minimal hazardous waste compared to traditional stoichiometric methods, and the use of recyclable solvents like dioxane further enhances the sustainability profile. This makes the technology highly scalable, capable of moving seamlessly from gram-scale laboratory synthesis to multi-ton annual commercial production without encountering the safety bottlenecks typical of gas-phase carbonylations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed carbonylation technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities and limitations for potential partners.
Q: What is the primary safety advantage of this carbonylation method?
A: Unlike traditional methods that require handling toxic and flammable carbon monoxide gas cylinders, this protocol utilizes 1,3,5-trimesic acid phenol ester as a solid, stable carbon monoxide substitute, significantly enhancing operational safety in industrial settings.
Q: What types of substrates are compatible with this synthesis route?
A: The method demonstrates excellent substrate compatibility, successfully accommodating both aryl-substituted olefins (yielding 2-aryl derivatives) and alkyl-substituted olefins (yielding 3-alkyl derivatives), including those with sensitive functional groups like halogens and silyl groups.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent explicitly states that the method is simple to operate, uses cheap and easily obtainable raw materials, and has been validated for gram-level synthesis, indicating strong potential for industrial scale-up and commercial application.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted 2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced carbonylation technology in accelerating the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market launch is seamless and efficient. Our state-of-the-art facilities are equipped to handle complex organometallic reactions with the highest standards of safety and precision, supported by rigorous QC labs that guarantee stringent purity specifications for every batch of high-purity pharmaceutical intermediate we deliver.
We invite you to collaborate with our technical team to explore how this innovative synthesis route can optimize your specific project requirements. By leveraging our expertise in process development, we can provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how this method can lower your overall cost of goods. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss custom route feasibility assessments for your proprietary targets.
