Advanced Catalyst-Free Synthesis of 1,3-Cyclohexadiene Compounds for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing complex cyclic scaffolds, particularly 1,3-cyclohexadiene derivatives which serve as critical building blocks for numerous bioactive molecules and natural products. Patent CN108395380B introduces a groundbreaking synthetic strategy that addresses long-standing challenges in this domain by utilizing a catalyst-free, thermal multi-component tandem reaction. This innovative approach combines allenone or allenoate compounds, primary amines, and enones in a single pot to directly access highly functionalized 1,3-cyclohexadiene structures. The significance of this technology lies in its ability to bypass the need for expensive transition metal catalysts and harsh reagents, thereby offering a greener, more economically viable pathway for the production of high-purity pharmaceutical intermediates. By leveraging simple thermal activation under air or nitrogen atmospheres, this method not only simplifies the operational workflow but also enhances the safety profile of the manufacturing process, making it an attractive candidate for industrial adoption by forward-thinking chemical enterprises.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 1,3-cyclohexadiene cores has relied on a variety of sophisticated yet often cumbersome synthetic routes that pose significant hurdles for large-scale manufacturing. Traditional strategies frequently involve intermolecular enyne cross-metathesis reactions requiring specialized ruthenium or molybdenum catalysts, which are not only costly but also sensitive to air and moisture, necessitating rigorous exclusion protocols that drive up operational expenses. Other established methods, such as the Michael/Wittig tandem reactions involving phosphorus salts, generate substantial amounts of phosphine oxide waste, creating severe environmental disposal issues and complicating downstream purification processes. Furthermore, cobalt-catalyzed [2+2+2] cycloadditions, while effective, introduce the risk of heavy metal contamination in the final active pharmaceutical ingredient (API), mandating expensive and time-consuming metal scavenging steps to meet stringent regulatory limits. These conventional pathways often suffer from limited substrate scope, poor atom economy, and multi-step sequences that erode overall yield and increase the carbon footprint of the synthesis, rendering them less ideal for the cost-sensitive and sustainability-focused landscape of modern chemical production.
The Novel Approach
In stark contrast to these legacy techniques, the methodology disclosed in CN108395380B represents a paradigm shift towards simplicity and efficiency through a direct three-component coupling strategy. This novel approach utilizes readily available allenones, primary amines, and enones as starting materials, which undergo a seamless thermal cascade to form the target 1,3-cyclohexadiene ring system without the intervention of any external catalyst or base. The reaction conditions are remarkably mild, typically proceeding at temperatures between 80°C and 140°C in common organic solvents like 1,4-dioxane or 1,2-dichloroethane, and tolerates both air and nitrogen atmospheres, significantly reducing the engineering controls required for reactor setup. As illustrated in the general reaction scheme below, this one-pot protocol achieves high atomic economy by incorporating all atoms of the reactants into the final product structure, minimizing waste generation and maximizing resource utilization.
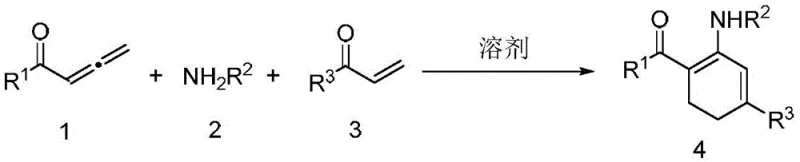
Furthermore, the versatility of this method is evidenced by its compatibility with a wide array of functional groups, including halogens, ethers, and heterocycles, allowing for the rapid diversification of the chemical library without the need for protecting group strategies. This streamlined process eliminates the logistical complexities associated with catalyst handling and removal, thereby offering a distinct advantage in terms of both time-to-market and overall process robustness for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Thermal Multi-component Tandem Cyclization
The underlying mechanism of this transformation is a fascinating example of thermal pericyclic and nucleophilic processes working in concert to build molecular complexity from simple precursors. Although the reaction proceeds without an added catalyst, the inherent reactivity of the allenone moiety serves as the driving force, acting as both an electrophile and a dipole in the sequential bond-forming events. Initially, the primary amine likely engages in a nucleophilic attack on the electron-deficient allene system or the enone, generating a reactive zwitterionic or neutral intermediate that sets the stage for subsequent cyclization. The thermal energy supplied at 120°C provides the necessary activation barrier crossing for the intramolecular ring closure, effectively orchestrating the formation of the six-membered 1,3-cyclohexadiene ring through a concerted or stepwise cascade that ensures high regioselectivity. This catalyst-free nature implies that the reaction trajectory is governed purely by the electronic and steric properties of the substrates, leading to a cleaner impurity profile compared to metal-catalyzed variants where side reactions like homocoupling or catalyst decomposition can occur.
From an impurity control perspective, the absence of transition metals removes a major source of potential contamination, simplifying the purification workflow to standard techniques like silica gel chromatography or recrystallization. The high selectivity observed across various substrates, such as those bearing electron-withdrawing fluorine or chlorine atoms, suggests that the transition state is well-defined and tolerant of electronic perturbations, ensuring consistent product quality. For R&D teams focused on process chemistry, understanding that the reaction relies on thermal activation rather than sensitive catalytic cycles means that scale-up risks related to catalyst deactivation or exothermic runaway are significantly mitigated. The robustness of this mechanism allows for a broader operating window, facilitating the production of high-purity OLED material precursors or drug candidates with minimal batch-to-batch variability, which is a critical parameter for maintaining supply chain reliability in regulated industries.
How to Synthesize 1,3-Cyclohexadiene Derivatives Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting involves a straightforward procedure that emphasizes operational simplicity and safety. The process begins with the precise weighing and dissolution of the allenone, primary amine, and enone components in a selected solvent, followed by heating the mixture to the optimized temperature range. The reaction progress can be monitored via standard analytical techniques such as TLC or HPLC, with typical reaction times around 12 hours ensuring complete conversion of the starting materials. Upon completion, the workup involves the removal of the solvent under reduced pressure, yielding a crude residue that can be purified to obtain the desired 1,3-cyclohexadiene derivative in high purity. The detailed standardized synthesis steps, including specific molar ratios and purification parameters derived from the patent examples, are outlined in the guide below to assist technical teams in replicating these results.
- Dissolve allenone or allenoate compounds, primary amine compounds, and enone compounds in a suitable organic solvent such as 1,4-dioxane or 1,2-dichloroethane.
- Heat the reaction mixture to a temperature between 80°C and 140°C under an air or nitrogen atmosphere for approximately 12 hours to facilitate the tandem cyclization.
- Remove the solvent under reduced pressure and purify the resulting crude 1,3-cyclohexadiene product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalyst-free synthetic route offers compelling economic and logistical benefits that directly impact the bottom line. By eliminating the requirement for precious metal catalysts such as palladium, ruthenium, or cobalt, the raw material costs are drastically simplified, removing the volatility associated with the pricing of these scarce resources. Additionally, the removal of metal scavenging resins and the associated filtration equipment reduces capital expenditure and consumable costs, contributing to substantial cost savings in pharmaceutical intermediate manufacturing. The use of commodity solvents like 1,4-dioxane and the tolerance to air atmosphere further lower the barrier to entry, allowing for production in standard glass-lined reactors without the need for specialized inert gas infrastructure, thereby enhancing supply chain reliability and reducing lead time for high-purity intermediates.
- Cost Reduction in Manufacturing: The most significant financial advantage stems from the complete avoidance of expensive transition metal catalysts and ligands, which often account for a disproportionate share of the bill of materials in traditional cross-coupling or cycloaddition reactions. Without the need for rigorous metal removal steps to meet ppm-level specifications, the downstream processing becomes faster and less resource-intensive, leading to a more streamlined production cycle. This efficiency translates into a lower cost of goods sold (COGS), enabling competitive pricing strategies for bulk chemical supplies while maintaining healthy profit margins. Furthermore, the high atom economy of the multi-component reaction ensures that raw materials are utilized effectively, minimizing waste disposal fees and maximizing the yield per kilogram of input, which is a key driver for long-term economic sustainability.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as substituted allenones, benzylamines, and chalcones ensures a stable and resilient supply chain that is less susceptible to geopolitical disruptions or single-source bottlenecks. Since the reaction does not depend on specialized catalysts that may have long lead times or limited global suppliers, procurement teams can secure raw materials more easily and maintain consistent inventory levels. The robustness of the reaction conditions, which tolerate air and moderate temperatures, also reduces the risk of batch failures due to minor deviations in atmospheric control, ensuring a steady flow of finished goods to meet customer demand. This reliability is crucial for maintaining just-in-time delivery schedules and fostering strong partnerships with downstream API manufacturers who depend on uninterrupted material flow.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this green chemistry approach aligns perfectly with increasingly stringent global standards for waste reduction and solvent usage. The absence of heavy metals eliminates the generation of hazardous metal-containing waste streams, simplifying effluent treatment and reducing the environmental footprint of the manufacturing facility. The process is inherently scalable, as demonstrated by the successful execution of reactions on millimole scales in the patent examples, suggesting a smooth translation to kilogram and tonne scales with minimal re-optimization. This scalability, combined with the use of common solvents and simple workup procedures, facilitates rapid commercial scale-up of complex polymer additives or drug intermediates, allowing companies to respond quickly to market opportunities while adhering to eco-friendly manufacturing principles.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthetic methodology, providing clarity for technical teams evaluating its feasibility for their specific projects. These answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for process development discussions. Understanding these nuances is essential for making informed decisions about integrating this technology into existing production pipelines or new product development roadmaps.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the method described in patent CN108395380B operates without the need for external bases or transition metal catalysts, relying instead on thermal activation to drive the multi-component tandem reaction.
Q: What is the optimal temperature range for this reaction?
A: The reaction proceeds efficiently within a heating temperature range of 80°C to 140°C, with 120°C identified as a preferred condition for maximizing yield in solvents like 1,4-dioxane.
Q: Is the substrate scope limited to specific aromatic groups?
A: The method demonstrates broad substrate tolerance, successfully accommodating phenyl, substituted phenyl (with fluoro, chloro, bromo, methyl, methoxy groups), naphthyl, and thiophen-2-yl groups on the starting materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Cyclohexadiene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalyst-free thermal synthesis described in CN108395380B for producing high-value 1,3-cyclohexadiene derivatives. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch, meeting the exacting standards required by the global pharmaceutical and fine chemical sectors. We are committed to delivering not just a product, but a comprehensive solution that optimizes your supply chain and enhances your competitive edge in the market.
We invite you to collaborate with our expert technical procurement team to explore how this innovative synthesis route can be tailored to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this catalyst-free protocol for your specific application. We encourage you to contact us today to discuss your project requirements,索取 specific COA data, and receive detailed route feasibility assessments that will empower you to make strategic sourcing decisions with confidence.
