Revolutionizing Organic Electronics with Efficient Boron Nitrogen Benzocarbazole Synthesis
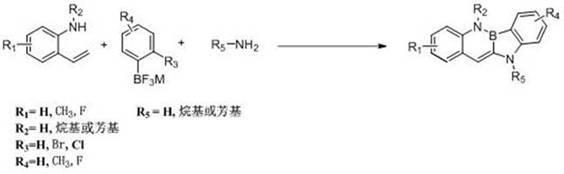
The landscape of organic photoelectric materials is undergoing a significant transformation with the introduction of novel synthetic methodologies described in patent CN112778347A. This groundbreaking intellectual property details a robust and efficient method for synthesizing boron nitrogen benzocarbazole derivatives, a class of compounds that has garnered immense attention for their potential applications in Organic Light Emitting Diodes (OLEDs), Organic Field Effect Transistors (OFETs), and organic solar cells. The core innovation lies in the strategic replacement of carbon-carbon double bonds within a pi-conjugated system with boron-nitrogen units, thereby constructing a unique boron-nitrogen aromatic system that exhibits superior electronic properties compared to traditional all-carbon analogues. By leveraging a high-efficiency transition metal catalytic series reaction, this technology overcomes the historical difficulties associated with boronization reactions, offering a pathway to precise molecular construction that was previously unattainable with conventional techniques. For R&D directors and procurement specialists alike, this patent represents a pivotal shift towards more accessible and scalable production of high-performance electronic materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of boron-nitrogen aromatic compounds has been fraught with significant technical hurdles that have impeded their widespread commercial adoption. Traditional approaches, such as electrophilic borylation, often necessitate the use of highly sensitive boron-bromine compounds which are not only difficult to handle but also pose substantial safety risks in a manufacturing environment. These legacy methods typically demand harsh reaction conditions, including extreme temperatures or pressures, which can degrade sensitive functional groups and lead to poor substrate compatibility. Furthermore, the synthesis of precursors for these reactions is often complex and multi-step, resulting in low overall yields and generating substantial chemical waste. The post-treatment processes are equally cumbersome, frequently requiring complicated purification steps to remove toxic byproducts and residual metals, which drastically increases the cost of goods sold and complicates supply chain logistics for fine chemical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN112778347A introduces a paradigm shift by utilizing a one-pot, three-component reaction strategy that is both operationally simple and chemically elegant. This novel approach employs commercially available and inexpensive starting materials, such as o-aminostyrene derivatives and aryl fluoroborate derivatives, which are stable and easy to source globally. The reaction proceeds under remarkably mild conditions, typically ranging from 25°C to 100°C, eliminating the need for energy-intensive heating or high-pressure equipment. By avoiding air-sensitive toxic substances and utilizing a composite catalyst system, the process ensures high reaction specificity and minimizes the formation of unwanted side products. This streamlined workflow not only enhances the overall yield, which has been demonstrated to reach up to 88% in specific embodiments, but also simplifies the downstream processing to basic operations like extraction and recrystallization, thereby offering a compelling value proposition for cost reduction in electronic chemical manufacturing.
Mechanistic Insights into Pd-Catalyzed Cyclization
The success of this synthetic route hinges on the sophisticated interplay between the palladium catalyst, phosphine ligands, and the specific substrates involved in the cascade reaction. The mechanism likely initiates with the oxidative addition of the aryl fluoroborate derivative to the palladium center, facilitated by the electron-rich nature of the specialized phosphine ligands such as 2-dicyclohexylphosphino-2',4',6'-triisopropylbiphenyl. This activation step is crucial for enabling the subsequent cross-coupling with the o-aminostyrene derivative, forming a key intermediate that sets the stage for ring closure. The presence of a base, such as sodium tert-butoxide or potassium carbonate, plays a vital role in neutralizing acidic byproducts and promoting the deprotonation steps necessary for the cyclization to occur. The careful tuning of the catalyst loading, typically between 0.5% to 10% molar percentage, ensures that the reaction kinetics are optimized for rapid conversion while minimizing the residual metal content in the final product, a critical factor for electronic applications.
Furthermore, the impurity control mechanism inherent in this design is exceptionally robust, addressing a major concern for R&D teams focused on purity specifications. The high selectivity of the palladium-catalyzed system ensures that the boron-nitrogen bond formation occurs exclusively at the desired positions, preventing the generation of regio-isomers that are notoriously difficult to separate. The use of fluoroborate salts instead of more reactive halides reduces the likelihood of homocoupling side reactions, which are common pitfalls in traditional cross-coupling chemistry. Additionally, the mild thermal profile of the reaction prevents thermal degradation of the sensitive boron-nitrogen core, preserving the integrity of the conjugated system. This level of control over the reaction pathway results in a crude product of such high quality that extensive chromatographic purification is often unnecessary, allowing for simple recrystallization to achieve the stringent purity levels required for high-purity OLED material production.
How to Synthesize Boron Nitrogen Benzocarbazole Derivatives Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must adhere to a precise protocol that balances reagent stoichiometry with environmental controls. The process begins with the rigorous exclusion of oxygen and moisture, typically achieved by vacuumizing and exchanging the reaction vessel with nitrogen or argon multiple times, as the palladium catalyst is sensitive to oxidation. Following the addition of the o-aminostyrene derivative, aryl fluoroborate, amine, and the composite catalyst system into an anhydrous solvent like toluene or DMF, the mixture is heated to the specified temperature range. Detailed standardized synthesis steps regarding exact weighing, addition rates, and quenching procedures are provided in the guide below to ensure reproducibility and safety.
- Combine o-aminostyrene derivatives, aryl fluoroborate derivatives, amines, and a composite palladium catalyst system in a solvent under nitrogen atmosphere.
- Heat the reaction mixture to a temperature between 25°C and 100°C and maintain stirring for a duration of 1 to 24 hours to ensure complete conversion.
- Upon completion, perform standard workup procedures including extraction with ethyl acetate, drying over anhydrous sodium sulfate, and recrystallization to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic methodology offers profound advantages that directly address the pain points of procurement managers and supply chain heads. The transition from complex, multi-step legacy processes to this streamlined one-pot reaction significantly reduces the operational overhead associated with manufacturing. By eliminating the need for exotic or hazardous reagents, companies can mitigate regulatory compliance risks and lower the costs associated with waste disposal and safety protocols. The reliance on commodity chemicals like aniline and simple inorganic bases ensures a stable and resilient supply chain, reducing the vulnerability to raw material shortages that often plague the specialty chemical sector. This stability translates into more predictable lead times and the ability to scale production volumes rapidly in response to market demand without compromising on quality or consistency.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the reaction workflow and the use of low-cost starting materials. By removing the requirement for expensive and sensitive boron-halide reagents, the direct material costs are substantially lowered. Furthermore, the high yields and minimal side reactions mean that less raw material is wasted, improving the overall atom economy of the process. The simplified post-treatment, which avoids complex chromatography in favor of crystallization, drastically reduces solvent consumption and labor hours, leading to significant operational expenditure savings. These factors combine to create a highly competitive cost structure that allows for better margin management in the production of complex electronic chemical intermediates.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of widely available and stable reagents that do not require specialized storage or handling conditions. Unlike air-sensitive precursors used in older methods, the fluoroborate derivatives and amines utilized here can be sourced from multiple global suppliers, reducing dependency on single-source vendors. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by equipment failures or environmental fluctuations. This reliability ensures that customers can maintain consistent inventory levels and meet their own production schedules without the fear of unexpected delays, making this a dependable solution for long-term procurement strategies in the fast-paced electronics industry.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or tonne scale is straightforward due to the absence of exothermic hazards and the use of common solvents. The mild reaction temperatures reduce the energy load on manufacturing facilities, contributing to a lower carbon footprint and aligning with modern sustainability goals. Additionally, the reduction in toxic waste generation simplifies environmental compliance, as the effluent streams are easier to treat and dispose of according to international regulations. This environmental friendliness not only protects the company from regulatory fines but also enhances its corporate social responsibility profile, which is increasingly important for partnerships with major multinational corporations committed to green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on the practical aspects of adopting this new method. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary limitations of traditional electrophilic borylation methods?
A: Traditional methods often rely on sensitive boron-bromine compounds, require harsh reaction conditions, and suffer from poor substrate compatibility and complex post-treatment processes, limiting their scalability for industrial applications.
Q: How does the new Pd-catalyzed method improve reaction specificity?
A: By utilizing a composite catalyst system with optimized phosphine ligands and mild temperatures (25-100°C), the new method achieves high reaction specificity with minimal side reactions, resulting in yields ranging from 76% to 88%.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the process uses commercially available and inexpensive raw materials like aniline and avoids air-sensitive toxic substances, making it highly suitable for scalable manufacturing with simplified environmental compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Boron Nitrogen Benzocarbazole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technology described in CN112778347A and are fully equipped to bring these advanced materials to the global market. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from R&D to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are designed to handle complex organometallic chemistry with the utmost precision, adhering to stringent purity specifications and operating rigorous QC labs to guarantee that every batch meets the highest industry standards. We understand that in the competitive landscape of organic electronics, consistency and quality are paramount, and our dedicated team is committed to delivering products that empower your next generation of devices.
We invite you to collaborate with us to unlock the full commercial potential of these boron nitrogen benzocarbazole derivatives. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this novel synthesis can optimize your bottom line. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions with confidence. Let us be your partner in innovation, driving efficiency and excellence in your supply chain for high-purity electronic materials.
