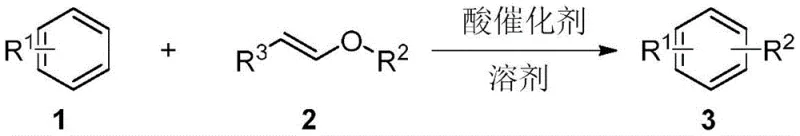
Advanced Synthesis of Alkyl Aromatic Intermediates via Alkenyl Ether Friedel-Crafts Alkylation
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways to access critical building blocks, particularly alkyl-substituted aromatic compounds which serve as foundational scaffolds for countless active pharmaceutical ingredients (APIs) and advanced materials. A significant breakthrough in this domain is detailed in Chinese Patent CN111410598B, which discloses a novel preparation method for alkyl aromatic compounds based on a Friedel-Crafts reaction utilizing alkenyl ethers. This technology represents a paradigm shift from classical alkylation strategies, offering a green, highly efficient synthetic route that operates under remarkably mild conditions. By leveraging the unique reactivity of alkenyl ethers in the presence of Lewis or protonic acid catalysts, this method achieves exceptional selectivity and yield while minimizing the environmental footprint typically associated with traditional electrophilic aromatic substitutions. For R&D directors and process chemists, this patent offers a robust solution to long-standing challenges in impurity control and reaction scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alkyl-substituted aromatic compounds has relied heavily on the Friedel-Crafts alkylation of aromatic rings using alkyl halides, alcohols, or olefins as the alkylating agents. While these classical methods are well-documented, they suffer from inherent and often insurmountable drawbacks that complicate manufacturing and purification processes. The primary issue lies in the generation of unstable carbocation intermediates, which are prone to skeletal rearrangements and disproportionation reactions, leading to a complex mixture of regioisomers and by-products that are difficult to separate. Furthermore, these traditional protocols frequently require harsh reaction conditions, including elevated temperatures and the use of stoichiometric or excessive amounts of strong Lewis acids like aluminum chloride, which generates substantial amounts of corrosive waste and complicates downstream processing. The poor atom economy and the necessity for rigorous quenching and neutralization steps significantly increase the operational costs and safety risks associated with these legacy processes.
The Novel Approach
In stark contrast to these conventional limitations, the methodology described in patent CN111410598B introduces alkenyl ethers as a superior class of alkylating reagents that fundamentally alter the reaction landscape. By employing alkenyl ethers in conjunction with aromatic compounds under acid catalysis, the process effectively bypasses the problematic carbocation rearrangements that plague traditional methods. The reaction proceeds with high specificity, ensuring that the alkyl group is transferred to the aromatic ring without skeletal modification, thereby delivering products with exceptional purity profiles. This novel approach not only simplifies the reaction workflow by utilizing mild temperatures ranging from 25°C to 130°C but also drastically reduces the catalyst loading requirements, often operating efficiently with catalytic amounts of inexpensive Lewis or protic acids. The result is a streamlined synthesis that aligns perfectly with the principles of green chemistry, offering a reliable pharmaceutical intermediate supplier with a distinct competitive advantage in terms of process efficiency and environmental compliance.
Mechanistic Insights into Alkenyl Ether Mediated Friedel-Crafts Alkylation
The mechanistic elegance of this transformation lies in the unique activation mode of the alkenyl ether functionality by the acid catalyst. Upon exposure to a Lewis acid or protonic acid, the electron-rich double bond of the alkenyl ether is activated, facilitating the formation of a stabilized oxocarbenium ion intermediate or a transient carbocation species that is resonance-stabilized by the adjacent oxygen atom. This stabilization is crucial as it prevents the uncontrolled rearrangement of the carbon skeleton, a common pitfall in standard alkyl halide alkylations. The aromatic nucleophile then attacks this activated species at the appropriate position, driven by the electronic properties of the substituents on the ring, leading to the formation of a new carbon-carbon bond. Subsequent elimination or hydrolysis steps release the alkylated aromatic product and a benign carbonyl by-product, such as an aldehyde or ketone, which can be easily removed during workup. This mechanism ensures that the reaction pathway is kinetically controlled towards the desired mono-alkylated product, minimizing polyalkylation and other side reactions.
From an impurity control perspective, this mechanism offers significant advantages for the production of high-purity OLED material or pharmaceutical intermediates. The avoidance of free, unrestrained carbocations means that isomeric impurities arising from hydride or alkyl shifts are virtually eliminated. Additionally, the compatibility of this catalytic system with a wide range of functional groups—including amino, hydroxyl, and alkoxy groups—allows for the direct functionalization of complex substrates without the need for extensive protecting group strategies. This functional group tolerance is particularly valuable when synthesizing complex heterocycles or polysubstituted benzenes, as it preserves the integrity of sensitive moieties that might otherwise degrade under the harsh acidic conditions required by traditional Friedel-Crafts protocols. The ability to tune the acidity of the catalyst further allows process chemists to optimize the reaction rate versus selectivity balance, ensuring consistent quality across different batches.
How to Synthesize Diphenylmethane Efficiently
The practical implementation of this technology is straightforward and amenable to standard laboratory and pilot plant equipment. The general procedure involves the sequential addition of the aromatic substrate, the chosen acid catalyst, the alkenyl ether reagent, and a suitable organic solvent into a reaction vessel. The mixture is then heated to the optimal temperature, typically between 60°C and 120°C, and stirred for a period ranging from 8 to 36 hours depending on the reactivity of the specific substrates involved. Following the reaction, the solvent is removed under reduced pressure, and the crude product is purified via silica gel column chromatography to afford the target alkyl aromatic compound in high yield. For a detailed breakdown of the specific molar ratios, solvent choices, and purification parameters tailored to your specific target molecule, please refer to the standardized synthesis guide below.
- Charge the reactor with the aromatic compound, acid catalyst (Lewis or protic), alkenyl ether, and organic solvent sequentially.
- Heat the reaction mixture to a temperature between 25°C and 130°C and maintain stirring for 8 to 36 hours to ensure complete conversion.
- Upon completion, remove the solvent via reduced pressure distillation and purify the crude product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this alkenyl ether-based alkylation technology translates into tangible strategic benefits that extend beyond mere chemical efficiency. The shift away from hazardous alkyl halides and stoichiometric metal halides towards stable alkenyl ethers and catalytic acid systems significantly de-risks the supply chain by utilizing reagents that are easier to store, transport, and handle. This reduction in hazard classification lowers logistics costs and simplifies regulatory compliance, ensuring a more resilient supply of critical intermediates. Furthermore, the simplified workup procedure, which eliminates the need for complex aqueous quenches and extensive washing steps required to remove metal salts, results in a drastic reduction in wastewater generation and treatment costs. These operational efficiencies contribute to a leaner manufacturing process that is both cost-effective and environmentally sustainable.
- Cost Reduction in Manufacturing: The economic impact of this method is profound, primarily driven by the replacement of expensive and wasteful stoichiometric reagents with low-loading, recoverable acid catalysts. By eliminating the need for excessive amounts of Lewis acids like aluminum chloride, the process removes the costly and time-consuming steps associated with the disposal of aluminum sludge and the neutralization of acidic waste streams. Additionally, the high selectivity of the reaction minimizes the formation of by-products, which directly improves the overall yield and reduces the volume of raw materials required per kilogram of final product. This atom-economic approach ensures that cost reduction in fine chemical manufacturing is achieved through fundamental process intensification rather than simple price negotiation.
- Enhanced Supply Chain Reliability: The reliance on alkenyl ethers, which are commercially available and stable liquid reagents, enhances the reliability of the raw material supply compared to alkyl halides that may be subject to stricter transportation regulations due to their lachrymatory or corrosive nature. The mild reaction conditions also reduce the stress on reactor equipment, extending the lifespan of capital assets and reducing maintenance downtime. This robustness ensures that production schedules can be maintained consistently, reducing lead time for high-purity pharmaceutical intermediates and allowing for more responsive fulfillment of customer orders even during periods of high demand.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the exothermic nature of the reaction being manageable under the specified temperature ranges, preventing thermal runaways that are common in traditional Friedel-Crafts reactions. The environmental profile is significantly improved due to the absence of heavy metal waste and the generation of benign organic by-products, aligning with increasingly stringent global environmental regulations. This ease of commercial scale-up of complex alkyl aromatics ensures that the technology remains viable and compliant as production volumes increase to meet market needs.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this alkenyl ether Friedel-Crafts technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the method's capabilities and limitations for potential adopters. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing manufacturing pipelines.
Q: Why are alkenyl ethers superior to traditional alkyl halides for Friedel-Crafts alkylation?
A: Traditional alkyl halides often suffer from carbocation rearrangements and disproportionation reactions, leading to complex product mixtures and poor selectivity. The use of alkenyl ethers in this novel protocol avoids these side reactions, providing high specificity and yield under much milder conditions.
Q: What types of catalysts are compatible with this alkenyl ether alkylation method?
A: The process is highly versatile and works effectively with a broad range of Lewis acids (such as Zinc Chloride, Indium Chloride) and protic acids (such as Trifluoroacetic acid, p-Toluenesulfonic acid), allowing for optimization based on substrate sensitivity.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method features mild reaction temperatures (25-130°C), simple post-reaction processing (distillation and chromatography), and high atom economy, making it environmentally friendly and economically viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkyl Aromatic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the alkenyl ether Friedel-Crafts alkylation technology in modernizing the production of essential chemical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant manufacturing environment. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of alkyl aromatic compounds meets the exacting standards required by the global pharmaceutical and electronic materials industries. We are equipped to handle the nuanced requirements of this chemistry, from catalyst screening to solvent optimization, delivering a product that is both high-quality and cost-competitive.
We invite you to collaborate with us to leverage this advanced synthetic methodology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis that demonstrates how switching to this greener alkylation route can optimize your specific bill of materials. Please contact our technical procurement team today to request specific COA data for our catalog of alkyl aromatics or to discuss route feasibility assessments for your proprietary targets, ensuring a seamless transition to a more efficient and sustainable supply chain.
