Advanced Nickel-Catalyzed Synthesis of Aromatic Acids for Commercial Scale-Up
The chemical industry is currently witnessing a paradigm shift towards sustainable manufacturing processes, particularly in the synthesis of high-value aromatic carboxylic acids which serve as critical building blocks for numerous active pharmaceutical ingredients and agrochemical products. Patent CN111018691B discloses a groundbreaking green synthesis method that replaces traditional palladium-catalyzed carbonylation with a more economical and environmentally benign nickel-catalyzed system. This innovation addresses the longstanding challenges associated with the use of toxic carbon monoxide gas and expensive noble metal catalysts, offering a viable pathway for the efficient conversion of aryl iodides into aromatic acids. By utilizing formate salts as a solid carbon monoxide surrogate in the presence of an acid anhydride, this technology ensures mild reaction conditions and exceptional operational safety. For global procurement and technical teams, this represents a significant opportunity to optimize supply chain resilience while adhering to increasingly stringent environmental regulations regarding heavy metal residues and hazardous gas handling in fine chemical manufacturing facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of aromatic carboxylic acids from aryl halides has relied heavily on palladium-catalyzed carbonylation reactions that require the direct use of carbon monoxide gas. This conventional approach presents severe logistical and safety hurdles, as carbon monoxide is a highly toxic and flammable gas that necessitates specialized high-pressure equipment and rigorous safety protocols to prevent leakage and exposure. Furthermore, the reliance on palladium, a precious noble metal, introduces substantial volatility in raw material costs and creates significant economic pressure on the final product pricing structure. The harsh reaction conditions often required for these traditional methods can also lead to poor functional group tolerance, resulting in complex impurity profiles that demand extensive and costly downstream purification steps. These factors collectively contribute to higher production costs, extended lead times, and increased environmental compliance burdens for manufacturers attempting to scale these processes for commercial pharmaceutical intermediate production.
The Novel Approach
The novel approach detailed in the patent data fundamentally reengineers the carbonylation process by substituting gaseous carbon monoxide with a safe, solid mixture of formate salts and acid anhydrides. This ingenious strategy allows for the in-situ generation of carbon monoxide directly within the reaction medium, thereby eliminating the need for external gas cylinders and high-pressure infrastructure. By switching from palladium to a cheap nickel catalyst system, the method drastically reduces the raw material cost base while maintaining high catalytic efficiency and selectivity. The reaction proceeds under mild thermal conditions, typically between 80°C and 110°C, which significantly lowers energy consumption and enhances the compatibility with sensitive functional groups commonly found in complex drug molecules. This technological leap not only simplifies the operational workflow but also aligns perfectly with modern green chemistry principles, offering a robust solution for the sustainable manufacturing of high-purity aromatic acid intermediates.
Mechanistic Insights into Nickel-Catalyzed Carbonyl Insertion
The core of this technological advancement lies in the sophisticated nickel-catalyzed catalytic cycle that facilitates the insertion of a carbonyl group into the carbon-iodine bond of the aryl iodide substrate. The mechanism initiates with the oxidative addition of the aryl iodide to the active nickel(0) species, forming an aryl-nickel(II) intermediate that is primed for subsequent transformation. In the presence of the formate salt and acid anhydride, carbon monoxide is generated in situ and immediately coordinates to the nickel center, setting the stage for the migratory insertion step that forms the acyl-nickel bond. This precise control over the carbonyl source ensures that the concentration of reactive CO remains optimal throughout the reaction, minimizing side reactions such as homocoupling or decarbonylation that often plague traditional methods. The cycle concludes with a reductive elimination step that releases the desired aromatic acid product and regenerates the active nickel catalyst, allowing the process to continue efficiently with minimal catalyst loading.
From an impurity control perspective, this mechanism offers distinct advantages by avoiding the use of harsh oxidizing agents that typically degrade sensitive substituents on the aromatic ring. The mild nature of the nickel catalytic system ensures excellent tolerance for a wide range of functional groups, including esters, aldehydes, ethers, and even halogens, which might otherwise be compromised under more aggressive conditions. The use of phosphine ligands further stabilizes the nickel center, preventing the formation of inactive nickel black precipitates and ensuring consistent reaction performance over extended periods. This high level of chemoselectivity results in a cleaner crude reaction mixture, significantly reducing the burden on purification units and enabling the production of intermediates with stringent purity specifications required for pharmaceutical applications. The ability to maintain structural integrity while introducing the carboxylic acid moiety is crucial for the synthesis of complex API intermediates where multiple synthetic steps follow.

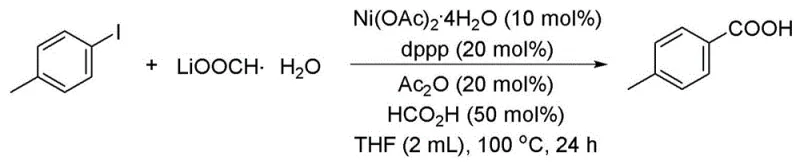
How to Synthesize 4-Methylbenzoic Acid Efficiently
To demonstrate the practical application of this green synthesis technology, we examine the specific preparation of 4-methylbenzoic acid, a key intermediate used in various pharmaceutical and agrochemical formulations. The process involves charging a reaction vessel with 4-iodotoluene, lithium formate monohydrate, a nickel catalyst such as nickel(II) acetate tetrahydrate, and a phosphine ligand like dppp in a solvent system comprising tetrahydrofuran. The addition of acetic anhydride and formic acid triggers the in-situ release of carbon monoxide, which then undergoes insertion into the aryl-iodide bond under heated conditions of approximately 100°C. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining aryl iodide, lithium formate monohydrate, nickel catalyst, and phosphine ligand in an organic solvent under inert atmosphere.
- Add acid anhydride and formic acid to facilitate the in-situ generation of carbon monoxide from the formate salt within the reaction vessel.
- Heat the reaction system to 80-110°C for 10-30 hours to complete the carbonyl insertion, followed by standard workup and purification to isolate the aromatic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this nickel-catalyzed synthesis route offers transformative benefits that extend far beyond simple chemical conversion metrics. The elimination of palladium catalysts removes a major cost driver and supply risk associated with precious metal price fluctuations, leading to a more stable and predictable cost structure for long-term contracts. Additionally, the replacement of toxic carbon monoxide gas with solid formate salts simplifies logistics, storage, and handling requirements, thereby reducing the regulatory overhead and insurance costs associated with hazardous material management. The mild reaction conditions also translate to lower energy consumption and reduced wear on reactor equipment, contributing to overall operational efficiency and sustainability goals. These combined factors create a compelling value proposition for companies seeking to optimize their manufacturing spend while enhancing their environmental, social, and governance (ESG) performance profiles in the global market.
- Cost Reduction in Manufacturing: The substitution of expensive palladium catalysts with abundant and cheap nickel salts results in a direct and significant reduction in raw material expenditure for every batch produced. Furthermore, the avoidance of high-pressure carbon monoxide infrastructure eliminates the capital investment and maintenance costs associated with specialized gas handling systems and safety monitoring equipment. The simplified workup procedure, driven by cleaner reaction profiles, reduces solvent usage and waste disposal costs, contributing to substantial overall cost savings in the manufacturing process. This economic efficiency allows for more competitive pricing strategies without compromising on the quality or purity of the final aromatic acid products supplied to downstream customers.
- Enhanced Supply Chain Reliability: Utilizing solid reagents like lithium formate and acetic anhydride instead of compressed gases mitigates the risks associated with transportation delays and regulatory restrictions on hazardous materials. The widespread availability of nickel catalysts and common organic solvents ensures a robust supply base that is less susceptible to geopolitical disruptions or single-source bottlenecks. This reliability is critical for maintaining continuous production schedules and meeting strict delivery deadlines for pharmaceutical clients who depend on just-in-time inventory models. By securing a more resilient raw material base, manufacturers can guarantee consistent supply continuity even in volatile market conditions.
- Scalability and Environmental Compliance: The mild thermal conditions and absence of toxic gas emissions make this process inherently safer and easier to scale from pilot plant to full commercial production volumes. The reduction in heavy metal residues simplifies the purification process, ensuring that the final product meets stringent regulatory limits for elemental impurities without requiring complex scavenging steps. This alignment with green chemistry principles facilitates smoother regulatory approvals and enhances the corporate reputation regarding environmental stewardship. Consequently, the technology supports sustainable growth and long-term viability in an increasingly regulated global chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed aromatic acid synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and advantages. Understanding these details is essential for technical teams evaluating the feasibility of integrating this method into existing manufacturing workflows. We encourage stakeholders to review these answers to gain a comprehensive understanding of the operational benefits and quality assurances provided by this innovative approach.
Q: What are the primary advantages of using nickel over palladium for aromatic acid synthesis?
A: Nickel catalysts are significantly more cost-effective than noble metal palladium catalysts and offer comparable efficiency for carbonyl insertion reactions. Furthermore, the nickel-based system described in patent CN111018691B eliminates the need for toxic carbon monoxide gas by using solid formate salts, enhancing operational safety and reducing environmental compliance burdens.
Q: How does this method improve impurity profiles compared to traditional oxidation routes?
A: Traditional multi-step oxidation routes often generate complex byproduct mixtures due to over-oxidation or harsh conditions. This nickel-catalyzed carbonylation proceeds under milder temperatures (80-110°C) with high functional group tolerance, minimizing side reactions and simplifying downstream purification processes for high-purity API intermediates.
Q: Is this synthesis method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the method utilizes commercially available reagents like lithium formate and acetic anhydride, avoiding the logistical challenges of handling high-pressure CO gas. The mild reaction conditions and robust catalyst system support reliable commercial scale-up from kilogram to multi-ton annual production capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical and fine chemical industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the nickel-catalyzed carbonylation described in CN111018691B can be successfully translated into robust industrial operations. We are committed to delivering high-purity aromatic acid intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to quality and consistency makes us a trusted partner for companies seeking reliable sources of complex chemical building blocks.
We invite you to collaborate with us to explore how this green synthesis technology can optimize your supply chain and reduce manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can support your project goals. Let us help you achieve greater efficiency and sustainability in your aromatic acid sourcing strategy.
