Advanced Osmium Vinylidene Complexes: Revolutionizing Catalytic Hydrosilylation for Pharmaceutical Intermediates
Advanced Osmium Vinylidene Complexes: Revolutionizing Catalytic Hydrosilylation for Pharmaceutical Intermediates
The landscape of organometallic chemistry is continually evolving with the discovery of novel structures that bridge the gap between fundamental research and industrial application. A significant breakthrough in this domain is detailed in patent CN115960142A, which introduces a brand-new class of metal heterocyclic compounds containing an intracyclic osmium vinylidene bond. These compounds represent a substantial advancement over previously known isoosmium benzenes and pyridines, offering unique structural features that translate into superior catalytic performance. The invention addresses the historical challenge of synthesizing high-ring-tension metalloheterocycles by providing a robust, one-pot methodology that utilizes readily available starting materials. This development is particularly critical for the fine chemical industry, where the demand for efficient, selective, and stable catalysts for C-H and Si-H activation is ever-increasing. By leveraging the unique electronic properties of the osmium center within a rigid heterocyclic framework, researchers have unlocked new pathways for transforming simple alkynes into valuable intermediates.
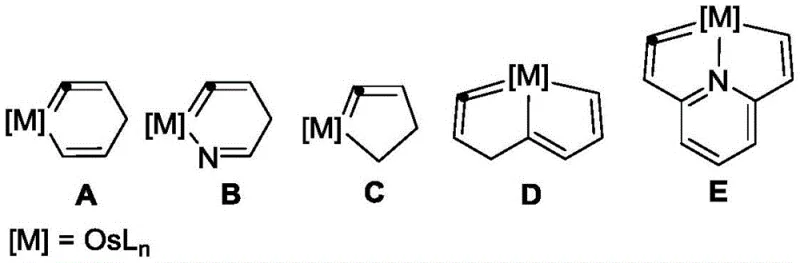
The limitations of conventional methods for preparing metal-carbon multiple bond compounds often stem from the inherent instability of the target structures and the complexity of the synthetic routes required to access them. Traditional approaches to synthesizing osmium vinylidene species frequently involve multi-step sequences, harsh reaction conditions, or the use of highly sensitive precursors that degrade upon exposure to air or moisture. Furthermore, existing compounds like isoosmium benzene A or osmium hetero-five-membered rings C and D often suffer from high ring strain that predisposes them to unwanted ring-opening reactions with nucleophiles, limiting their utility in practical synthesis. The lack of a general method to introduce diverse substituents at key positions on the heterocyclic ring has further restricted the tunability of these catalysts for specific industrial applications. Consequently, the scarcity of stable, modifiable osmium vinylidene complexes has hindered their widespread adoption in large-scale manufacturing processes where reliability and reproducibility are paramount.
In stark contrast to these legacy challenges, the novel approach disclosed in the patent utilizes a streamlined one-pot strategy that dramatically simplifies the construction of the osmium-vinylidene core. By reacting 2,6-bis(ethynyl)pyridine derivatives with an osmium source such as OsCl2(PPh3)3 followed by the addition of an organic halogen electrophile like N-halosuccinimide, the target metalloheterocycles are formed with high efficiency. This method not only avoids the isolation of unstable intermediates but also allows for the direct introduction of various substituents at the 9-position of the ring in a single operational step. The reaction proceeds under mild conditions, typically at room temperature in dichloromethane, and tolerates a wide range of functional groups on the pyridine ligand. This versatility enables the rapid generation of a library of catalysts with tailored steric and electronic properties, facilitating the optimization of catalytic performance for specific transformations such as hydrosilylation. The simplicity of the workup, involving basic filtration and chromatography, further enhances the practical appeal of this methodology for both academic research and industrial scale-up.
Mechanistic Insights into One-Pot Cyclization and Catalytic Activity
The formation of the intracyclic osmium vinylidene bond involves a sophisticated interplay of coordination chemistry and electrophilic activation that warrants detailed examination. The process initiates with the coordination of the bis-alkyne ligand to the osmium center, followed by an intramolecular cyclization that is triggered by the electrophilic attack of the N-halosuccinimide reagent. This step is crucial as it establishes the rigid heterocyclic framework that stabilizes the otherwise reactive osmium-carbon double bond character. The resulting structure features a unique electronic environment where the metal center is locked within a planar or near-planar conjugated system, enhancing its ability to activate small molecules like silanes. The presence of phosphine ligands and the specific geometry of the pyridine backbone contribute to the overall stability of the complex, preventing decomposition pathways that typically plague similar high-valent metal species. Understanding this mechanistic pathway is essential for chemists aiming to replicate the synthesis or modify the ligand sphere to create next-generation catalysts with even higher turnover frequencies.
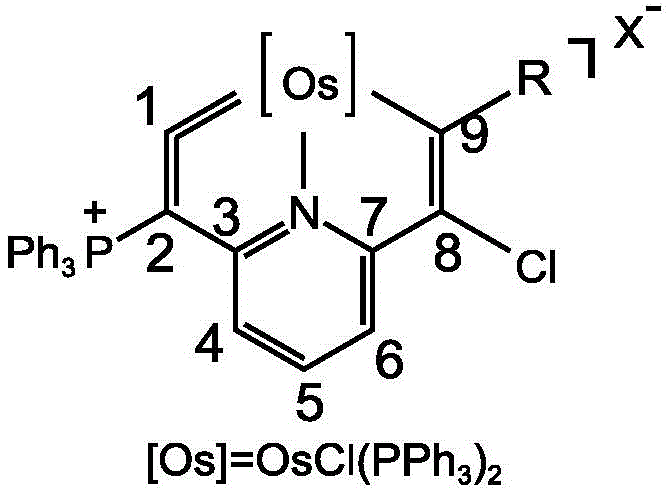
Impurity control in the synthesis of these complex organometallic species is managed through the high regioselectivity of the cyclization reaction and the robustness of the final product. Although the starting materials possess multiple potential reaction sites, the interaction between the osmium center and the specific alkyne moieties directs the reaction towards the formation of the desired seven-membered metallacycle with minimal side products. The use of N-halosuccinimides as oxidants ensures a clean transformation, as the succinimide byproduct is easily removed during the aqueous workup or column chromatography. Furthermore, the distinct NMR signatures of the osmium-bound carbons, which appear at characteristic downfield shifts due to the metal's influence, allow for precise monitoring of reaction completion and purity. This level of control over the impurity profile is vital for pharmaceutical applications, where trace metal contaminants or organic byproducts can compromise the safety and efficacy of the final drug substance. The ability to produce these catalysts as stable green or yellow powders with yields exceeding 80% underscores the reliability of the process.
How to Synthesize Osmium Vinylidene Complexes Efficiently
The synthesis of these high-value osmium catalysts is designed to be accessible to laboratories equipped with standard Schlenk techniques, although the final products exhibit surprising stability towards air. The protocol begins by dissolving the osmium precursor and the diethynylpyridine ligand in dry dichloromethane under an inert atmosphere to prevent premature oxidation. After a brief stirring period to ensure complete coordination, the electrophilic halogen source is introduced to drive the cyclization to completion. Detailed standardized synthesis steps for preparing specific analogues with varying substituents are provided in the guide below, ensuring reproducibility across different batches and scales. This straightforward procedure eliminates the need for specialized high-pressure equipment or cryogenic conditions, making it an attractive option for process chemistry teams looking to integrate novel catalytic technologies into their workflows.
- Mix 2,6-bis(ethynyl)pyridine derivatives with OsCl2(PPh3)3 in dichloromethane under inert gas at room temperature for 10 minutes.
- Add N-halosuccinimide (NIS, NBS, or NCS) as the electrophilic reagent and stir for an additional 30 minutes to complete the cyclization.
- Filter the reaction mixture, concentrate the filtrate, and purify the resulting green or yellow powder via neutral alumina column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this new osmium vinylidene technology offers compelling advantages rooted in process efficiency and material stability. The ability to conduct catalytic reactions under ambient air atmosphere represents a paradigm shift from traditional organometallic catalysis, which often requires rigorous exclusion of oxygen and moisture using expensive gloveboxes or specialized reactors. This tolerance to air significantly reduces the capital expenditure associated with reactor infrastructure and lowers the operational costs related to inert gas consumption and maintenance of anaerobic conditions. Moreover, the high selectivity observed in the hydrosilylation of phenylacetylene, favoring the alpha-isomer with a ratio of 98/2, minimizes the need for downstream purification steps such as distillation or recrystallization to separate isomeric byproducts. This reduction in processing complexity translates directly into shorter production cycles and lower energy consumption, aligning with modern sustainability goals and cost-reduction strategies in fine chemical manufacturing.
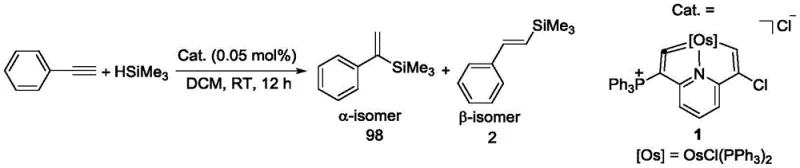
Enhanced supply chain reliability is another critical benefit derived from the robust nature of these catalysts and the simplicity of their synthesis. The starting materials, including OsCl2(PPh3)3 and various N-halosuccinimides, are commercially available from stable global suppliers, reducing the risk of raw material shortages that can disrupt production schedules. The one-pot synthesis method itself is highly scalable, as demonstrated by the consistent yields obtained in laboratory examples, suggesting a smooth transition from gram-scale development to kilogram or ton-scale commercial production without significant re-optimization. Additionally, the long shelf-life of the isolated catalyst powders allows for inventory buffering, enabling manufacturers to stockpile critical catalytic components without fear of degradation. This inventory flexibility provides a strategic buffer against market volatility and ensures continuous supply continuity for downstream customers relying on these intermediates for their own synthesis campaigns.
Scalability and environmental compliance are further strengthened by the benign nature of the reaction conditions and the manageable waste profile. The use of dichloromethane as a solvent is standard in the industry, and established protocols exist for its recovery and recycling, minimizing environmental impact. The high atom economy of the cyclization reaction, combined with the low catalyst loading required for effective hydrosilylation (as low as 0.5 mol%), reduces the overall mass intensity of the process. This efficiency is crucial for meeting increasingly stringent regulatory standards regarding heavy metal discharge and solvent emissions. By implementing this technology, companies can achieve substantial cost savings through reduced waste disposal fees and lower raw material usage, while simultaneously enhancing their corporate social responsibility profiles through greener manufacturing practices. The combination of high performance, operational simplicity, and environmental compatibility makes this osmium vinylidene platform a highly attractive investment for forward-thinking chemical enterprises.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the stability, application, and customization of the osmium vinylidene catalysts described in the patent documentation. These insights are derived directly from the experimental data and mechanistic studies presented in the intellectual property, providing a reliable foundation for decision-making. Understanding these nuances helps R&D and procurement teams evaluate the fit of this technology within their existing process portfolios and identify opportunities for innovation.
Q: What are the stability conditions for these osmium vinylidene catalysts?
A: Unlike many sensitive organometallic species, these intracyclic osmium vinylidene complexes demonstrate remarkable stability, allowing catalytic reactions to proceed efficiently under ambient air atmosphere at room temperature.
Q: How does the selectivity compare to traditional hydrosilylation methods?
A: The patented catalysts achieve exceptional Markovnikov selectivity (alpha-isomer) exceeding 98%, significantly outperforming conventional systems that often produce mixtures of isomers requiring difficult separation.
Q: Can the substituent at the 9-position be modified?
A: Yes, the one-pot synthesis method allows for versatile functionalization at the 9-position by selecting different N-halosuccinimide reagents or substituted pyridine ligands, enabling tailored catalyst design.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Osmium Vinylidene Complex Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercially viable chemical solutions, and the osmium vinylidene complex platform exemplifies our commitment to innovation. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of catalyst meets the exacting standards required for pharmaceutical and fine chemical synthesis. We understand the critical importance of consistency in catalytic performance, and our quality management systems are designed to deliver products with reproducible activity and selectivity profiles.
We invite potential partners to engage with our technical procurement team to discuss how these novel osmium catalysts can optimize your specific hydrosilylation processes. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits associated with switching to this air-stable, high-selectivity catalytic system. We are prepared to provide specific COA data and route feasibility assessments tailored to your project requirements, helping you mitigate risk and accelerate your time-to-market. Let us collaborate to harness the power of cutting-edge organometallic chemistry for your next breakthrough in chemical manufacturing.
