Advanced Biomimetic Catalysis for Scalable Chiral Trifluoromethyl Amino Acid Production
Introduction to Breakthrough Biomimetic Catalysis Technology
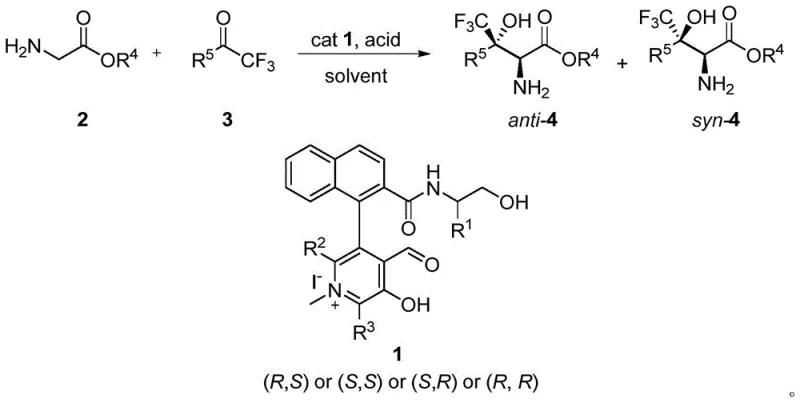
The pharmaceutical industry continuously seeks robust methodologies for incorporating fluorine atoms into bioactive molecules, particularly the trifluoromethyl group, due to its profound impact on metabolic stability, lipophilicity, and bioavailability. Patent CN111269132B introduces a transformative approach to synthesizing chiral β-trifluoromethyl-β-hydroxy-α-amino acids and their derivatives, which are critical building blocks for next-generation therapeutics. This technology leverages a novel biaryl-structured chiral N-methylpyridoxal catalyst to facilitate an asymmetric aldol reaction that mimics biological enzymatic processes. Unlike conventional chemical synthesis which often struggles with stereocontrol and harsh reaction environments, this biomimetic strategy operates under remarkably mild conditions, achieving high yields and exceptional enantioselectivity. For R&D directors and procurement specialists, this represents a paradigm shift towards more sustainable and cost-effective manufacturing of complex fluorinated intermediates.
The core innovation lies in the catalyst design, which enables the direct participation of glycine derivatives without the need for cumbersome protecting groups on the amino functionality. This not only streamlines the synthetic route but also aligns with green chemistry principles by reducing step count and waste generation. The patent details a comprehensive library of substrates, demonstrating the versatility of the catalyst across various trifluoromethyl ketones, ranging from aromatic to aliphatic and heteroatom-containing systems. As a reliable pharmaceutical intermediate supplier, understanding such foundational IP is crucial for securing supply chains for high-value API precursors. The ability to produce these chiral scaffolds with high optical purity directly impacts the efficacy and safety profiles of the final drug products, making this technology a cornerstone for modern medicinal chemistry programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral β-hydroxy-α-amino acids containing trifluoromethyl groups has been fraught with significant technical and economic challenges. Traditional routes often rely on stoichiometric chiral auxiliaries or transition metal catalysts that require strict anhydrous conditions and expensive ligands. A major bottleneck in these conventional methods is the necessity for amino protection and subsequent deprotection steps. The amino group in glycine is highly nucleophilic and reactive, necessitating protection to prevent side reactions during carbon-carbon bond formation. This adds at least two synthetic steps, increasing material costs, solvent consumption, and overall processing time. Furthermore, many existing methods suffer from moderate to poor diastereoselectivity and enantioselectivity, requiring costly and yield-loss-inducing purification steps like chiral HPLC or recrystallization to meet pharmaceutical grade standards.
Additionally, conventional metal-catalyzed approaches often face issues with residual metal contamination, which is a critical quality attribute for pharmaceutical ingredients. Removing trace metals to ppm levels adds further complexity and cost to the downstream processing. The reaction conditions for these older methods can also be quite harsh, involving cryogenic temperatures or strong bases that limit the scope of compatible functional groups. For supply chain heads, these factors translate into longer lead times, higher variability in batch-to-batch consistency, and increased vulnerability to raw material price fluctuations for specialized reagents. The inefficiency of these legacy processes creates a substantial barrier to the commercial scale-up of complex fluorinated amino acids, limiting their availability for drug development pipelines.
The Novel Approach
The methodology disclosed in CN111269132B offers a compelling solution to these longstanding problems by employing a biomimetic organocatalytic system. The novel chiral N-methylpyridoxal catalyst acts as a synthetic enzyme mimic, facilitating the direct aldol addition of tert-butyl glycinate to trifluoromethyl ketones. Crucially, this process eliminates the need for amino protection entirely. The catalyst forms a transient aldimine intermediate with the glycine ester, activating the alpha-proton for deprotonation under mild basic conditions generated in situ. This streamlined approach reduces the synthetic sequence significantly, directly addressing the cost reduction in pharmaceutical intermediate manufacturing. The reaction proceeds efficiently in common solvents like dichloromethane at temperatures as mild as -10°C, avoiding the energy intensity of deep cryogenic processes.
Moreover, the catalyst exhibits remarkable turnover efficiency. The patent examples demonstrate that catalyst loadings as low as 0.1 mol% are sufficient to drive the reaction to completion with high conversion. This ultra-low loading is a game-changer for commercial viability, as the synthesis of the chiral catalyst itself, while multi-step, becomes negligible in cost when amortized over large production batches. The structural versatility of the catalyst, featuring a biaryl backbone derived from BINOL resolution, allows for fine-tuning of the steric environment to optimize selectivity for different substrates. This adaptability ensures that the process remains robust even when scaling up to multi-kilogram or tonne quantities, providing a stable and reliable supply source for high-purity chiral intermediates required by global pharmaceutical partners.
Mechanistic Insights into Biomimetic Aldol Catalysis
The mechanistic elegance of this transformation lies in its simulation of natural enzymatic pathways involving Vitamin B6 derivatives. The chiral N-methylpyridoxal catalyst first condenses with the tert-butyl glycinate to form a chiral aldimine species. This imine formation significantly increases the acidity of the alpha-proton adjacent to the imine nitrogen, allowing for facile deprotonation by a mild base or even the solvent system to generate a nucleophilic carbanion equivalent. This activated species is held in a rigid chiral environment defined by the biaryl structure of the catalyst, which dictates the facial selectivity of the subsequent attack on the electrophilic trifluoromethyl ketone. The spatial arrangement of the bulky aryl groups on the catalyst shields one face of the planar intermediate, forcing the ketone to approach from the less hindered trajectory, thereby establishing the new stereocenters with high fidelity.
Following the carbon-carbon bond formation, the resulting adduct undergoes hydrolysis to release the final chiral β-trifluoromethyl-β-hydroxy-α-amino acid derivative and regenerate the free pyridoxal catalyst, completing the catalytic cycle. This regeneration is key to the low catalyst loading observed in the examples. The presence of the trifluoromethyl group on the ketone enhances its electrophilicity, facilitating the addition step, but also introduces steric bulk that challenges stereocontrol. The catalyst overcomes this through precise non-covalent interactions, potentially including hydrogen bonding or pi-stacking, which stabilize the transition state. Understanding this mechanism is vital for R&D teams aiming to further optimize the process or adapt it to novel substrates, ensuring that impurity profiles remain controlled and that the desired diastereomer (syn or anti) can be accessed selectively by choosing the appropriate catalyst enantiomer.
How to Synthesize Chiral Beta-Trifluoromethyl Amino Acids Efficiently
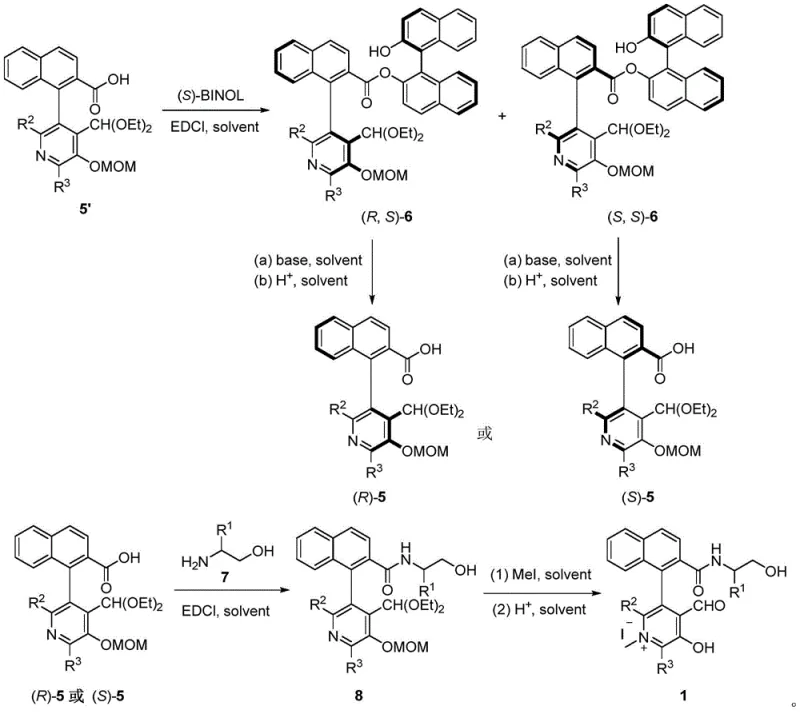
Implementing this synthesis route requires careful attention to the preparation of the chiral catalyst and the optimization of reaction parameters such as temperature and solvent choice. The process begins with the resolution of racemic carboxylic acid precursors using (S)-BINOL to isolate the single-configuration chiral acid, which is then converted into the active pyridoxal catalyst through condensation, methylation, and hydrolysis steps. Once the catalyst is prepared, the core aldol reaction is straightforward, involving the mixing of the glycine ester, trifluoromethyl ketone, acid additive, and catalyst in a suitable solvent. Detailed standard operating procedures for the synthesis steps are provided below to guide process chemists in replicating these high-yielding transformations.
- Resolution of carboxylic acid precursors using (S)-BINOL to obtain single-configuration chiral acid compounds.
- Condensation of the chiral acid with chiral amino alcohol followed by methylation and hydrolysis to generate the active pyridoxal catalyst.
- Catalytic asymmetric aldol reaction between tert-butyl glycinate and trifluoromethyl ketone in dichloromethane at -10°C to yield the target amino acid derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalytic technology offers tangible strategic benefits beyond mere technical novelty. The most significant advantage is the drastic simplification of the manufacturing workflow. By removing the protection and deprotection steps associated with the amino group, the overall process mass intensity (PMI) is significantly reduced. Fewer steps mean less solvent usage, lower energy consumption for heating and cooling, and reduced labor hours for reactor occupancy. This directly translates to substantial cost savings in the final price of the intermediate, making it more competitive in the global market. Furthermore, the use of readily available starting materials like tert-butyl glycinate and various trifluoromethyl ketones ensures a stable supply chain, reducing the risk of bottlenecks associated with exotic or proprietary reagents.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the ultra-low catalyst loading and the reduced step count. Traditional methods often require stoichiometric amounts of chiral reagents or expensive metal catalysts with loadings of 5-10 mol%. In contrast, this pyridoxal system operates effectively at 0.1 mol%, representing a fifty to one-hundred-fold reduction in catalyst cost per unit of product. Additionally, the elimination of protection groups removes the cost of protecting reagents and the solvents required for their removal. The mild reaction conditions (-10°C to room temperature) also lower utility costs compared to processes requiring cryogenic cooling below -78°C or high-temperature reflux, contributing to a leaner and more cost-efficient production model.
- Enhanced Supply Chain Reliability: Supply continuity is critical for pharmaceutical manufacturing, and this process enhances reliability through the use of robust and stable reagents. The chiral catalyst, once synthesized, is stable and can be stored for extended periods, allowing for batch production of the catalyst independent of the final API campaign. The starting materials, such as glycine esters and trifluoromethyl ketones, are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency. The high yields reported in the patent examples, often exceeding 80-90%, ensure that material throughput is maximized, reducing the need for oversized reactors to meet volume targets and allowing for faster turnaround times from order to delivery.
- Scalability and Environmental Compliance: Scaling organocatalytic reactions can sometimes be challenging due to mixing or heat transfer issues, but the mild nature of this aldol reaction facilitates smooth scale-up. The absence of heavy metals simplifies waste treatment and disposal, aligning with increasingly stringent environmental regulations regarding heavy metal discharge in pharmaceutical manufacturing. The solvent system, primarily dichloromethane or similar chlorinated solvents, is well-understood in industrial settings with established recovery and recycling protocols. The high selectivity of the reaction minimizes the formation of difficult-to-separate diastereomers, reducing the burden on purification units and lowering the volume of hazardous waste generated from chromatography or extensive recrystallization efforts, thus supporting a more sustainable and compliant operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral synthesis technology. These answers are derived directly from the experimental data and specifications outlined in the patent documentation, providing clarity on performance metrics and operational parameters. Understanding these details helps stakeholders assess the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the primary advantage of this pyridoxal catalytic method over traditional synthesis?
A: The primary advantage is the elimination of amino protection and deprotection steps. Traditional methods often require harsh conditions and multiple steps to protect the amine group, whereas this biomimetic approach allows glycine derivatives to react directly under mild conditions, significantly simplifying the workflow and reducing waste.
Q: What levels of stereoselectivity can be achieved with this catalyst system?
A: The patent data demonstrates excellent stereoselectivity. For instance, in the synthesis of compound 4b, the syn-isomer achieved an enantiomeric excess (ee) of 97%, while the anti-isomer reached 87%. Diastereomeric ratios (dr) are also favorable, often exceeding 1:5 or 1:6 in favor of the syn-product depending on the substrate.
Q: Is the catalyst loading efficient for large-scale production?
A: Yes, the process is highly efficient regarding catalyst usage. The patent examples utilize a catalyst loading as low as 0.1 mol% (e.g., 0.001 mmol catalyst for 1.0 mmol substrate). This ultra-low loading drastically reduces the cost per kilogram of product, making it economically viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Beta-Trifluoromethyl Amino Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced catalytic technologies play in accelerating drug discovery and development. Our team of expert process chemists has extensively evaluated the methodology described in CN111269132B and possesses the technical capability to implement this biomimetic aldol reaction on a commercial scale. We understand that transitioning a novel laboratory process to industrial production requires rigorous optimization and validation. Our facility is equipped with state-of-the-art reactors capable of maintaining the precise low-temperature conditions required for this synthesis, ensuring that the high enantioselectivity observed in the patent is maintained consistently across large batches. We are committed to delivering high-purity intermediates that meet the stringent quality standards of the global pharmaceutical industry.
We invite potential partners to engage with our technical procurement team to discuss how this technology can be tailored to your specific project needs. Whether you require custom synthesis of novel trifluoromethyl ketone substrates or scale-up of the final amino acid derivatives, we offer a Customized Cost-Saving Analysis to demonstrate the economic benefits of switching to this catalytic route. We encourage you to request specific COA data and route feasibility assessments from our team to verify our capacity and quality control measures. By leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we can help you secure a reliable supply of these valuable chiral building blocks, ensuring your drug development timelines are met with efficiency and confidence.
