Scalable One-Pot Synthesis of Difluoromethylthiochromonothiophene Compounds for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking efficient routes to novel heterocyclic scaffolds that possess significant biological activity. Patent CN110372718B introduces a groundbreaking methodology for the synthesis of difluoromethylthiochromonothiophene compounds, a unique class of fused heterocycles combining chromone and thiophene motifs. This innovation addresses critical bottlenecks in traditional synthetic organic chemistry by enabling the direct construction of these complex architectures through a streamlined one-pot tandem reaction. The significance of this technology lies not only in the novelty of the resulting molecular structures but also in the operational simplicity it affords process chemists. By utilizing inexpensive and readily available starting materials such as elemental sulfur and sodium difluorochloroacetate, the process circumvents the reliance on costly transition metal catalysts or hazardous difluoromethylating reagents that have historically plagued this field. For R&D directors and procurement managers alike, this represents a pivotal shift towards more sustainable and economically viable manufacturing protocols for high-value bioactive intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chromone derivatives and their sulfur-containing analogues has been fraught with significant technical and economic challenges. Traditional approaches often rely on multi-step sequences involving Claisen condensation, Baker-Venkatamaran rearrangement, or Kostanecki-Robinson reactions, which frequently require harsh reaction conditions and generate substantial chemical waste. Furthermore, the introduction of the difluoromethylthio group (-SCF2H) typically necessitates the use of specialized reagents such as PhSO2SCF2H or electrophilic silver complexes, which are not only prohibitively expensive but also pose safety risks due to their instability. These conventional pathways often suffer from poor atom economy and require rigorous purification steps to remove trace metal contaminants, a critical concern for pharmaceutical applications. The cumulative effect of these limitations is a high cost of goods sold (COGS) and extended lead times, making the commercialization of such complex molecules difficult for supply chain managers aiming for cost-effective production.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a convergent one-pot strategy that dramatically simplifies the synthetic landscape. By reacting 2-hydroxychalcone derivatives directly with sodium difluorochloroacetate and elemental sulfur in the presence of potassium methoxide and TEMPO, the process achieves simultaneous ring closure and functionalization. This approach eliminates the need for isolating unstable intermediates, thereby reducing solvent consumption and processing time. The reaction proceeds under mild thermal conditions, typically between 60°C and 80°C, which enhances energy efficiency and safety profiles compared to high-temperature alternatives. As demonstrated in the specific embodiment below, the transformation efficiently constructs the tricyclic core with high regioselectivity.
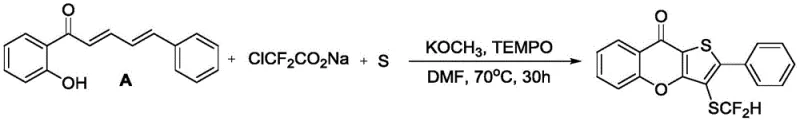
This streamlined workflow not only accelerates the timeline from bench to pilot plant but also ensures a cleaner impurity profile, which is paramount for regulatory compliance in drug substance manufacturing. The ability to access these novel scaffolds in a single operational step represents a substantial advancement in process chemistry, offering a reliable pathway for the reliable pharmaceutical intermediate supplier to meet growing market demands.
Mechanistic Insights into TEMPO-Mediated Tandem Cyclization
The success of this transformation hinges on a sophisticated cascade of mechanistic events initiated by the base-mediated activation of the substrate. Initially, potassium methoxide deprotonates the phenolic hydroxyl group of the 2-hydroxychalcone derivative, generating a nucleophilic phenoxide species. This anion undergoes an intramolecular Michael addition across the conjugated enone system, closing the pyrone ring to form the chromone backbone and generating a transient carbanion at the alpha-position. Simultaneously, elemental sulfur (S8) interacts with the reaction medium to generate polysulfide species, which act as soft nucleophiles. These sulfur species attack the electrophilic centers generated during the cyclization, facilitating the formation of the thiophene ring fusion. The presence of TEMPO (2,2,6,6-tetramethylpiperidine-1-oxyl) is crucial as a mild oxidant; it mediates the dehydrogenation steps required to aromatize the newly formed thiophene ring and stabilizes radical intermediates that might otherwise lead to polymerization or side reactions.
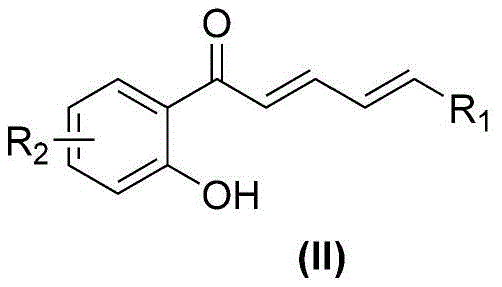
Following the construction of the fused heterocyclic core, the difluoromethylthio group is installed via nucleophilic substitution. The carbanion generated during the cyclization sequence attacks the difluorocarbene species derived from the decomposition of sodium difluorochloroacetate under the basic conditions. This final step installs the pharmacologically important -SCF2H moiety with high fidelity. Understanding this intricate interplay between base catalysis, sulfur insertion, and oxidative aromatization allows process engineers to fine-tune reaction parameters such as stoichiometry and temperature to maximize yield. The robustness of this mechanism against various electronic substituents on the aromatic rings ensures broad substrate applicability, making it a versatile tool for medicinal chemists exploring structure-activity relationships in new drug candidates.
How to Synthesize Difluoromethylthiochromonothiophene Efficiently
To implement this synthesis effectively, precise control over reaction conditions and reagent quality is essential. The protocol dictates the use of anhydrous dimethylformamide (DMF) as the solvent to ensure complete solubility of the inorganic salts and organic substrates. Molecular sieves are often added as an additive to scavenge trace moisture, which can hydrolyze the reactive difluorocarbene intermediates and lower the overall yield. The reaction is typically conducted under an inert atmosphere, such as nitrogen or argon, to prevent the oxidation of sensitive sulfur species by atmospheric oxygen. While the standard procedure involves heating at 70°C for approximately 30 hours, optimization studies suggest that temperatures ranging from 60°C to 80°C can be employed depending on the specific electronic nature of the substrate. Detailed standardized operating procedures for scaling this reaction are provided in the technical guide below.
- Dissolve potassium methoxide, TEMPO, 2-hydroxychalcone derivative, sodium difluorochloroacetate, and sulfur in anhydrous DMF under inert gas protection.
- Heat the reaction mixture to 60-80°C (optimally 70°C) and stir for 20-36 hours to facilitate tandem cyclization and difluoromethylthiolation.
- Cool to room temperature, filter insolubles, extract with ethyl acetate, and purify via silica gel column chromatography using ethyl acetate/petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers compelling advantages that directly impact the bottom line and supply chain resilience. The primary driver for cost reduction is the substitution of exotic, high-cost reagents with commodity chemicals. Elemental sulfur and sodium difluorochloroacetate are produced on a massive industrial scale, ensuring a stable and predictable supply chain that is immune to the volatility often seen with specialized organometallic reagents. Furthermore, the elimination of transition metal catalysts removes the necessity for expensive and time-consuming metal scavenging steps during downstream processing. This simplification of the purification train not only reduces the consumption of silica gel and solvents but also shortens the batch cycle time, allowing for higher throughput in existing manufacturing facilities. For procurement managers, this translates to a significant decrease in the cost of goods sold without compromising the quality of the final active pharmaceutical ingredient.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from the use of low-cost starting materials and the consolidation of multiple synthetic steps into a single pot. By avoiding the use of precious metal catalysts such as palladium or rhodium, the process eliminates a major cost center associated with both reagent purchase and waste disposal. Additionally, the high atom economy of the tandem reaction minimizes the generation of byproduct waste, reducing the environmental fees and treatment costs associated with chemical manufacturing. This lean manufacturing approach ensures that the production of high-purity pharmaceutical intermediates remains financially viable even in competitive markets.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the reliance on globally available bulk chemicals rather than niche reagents sourced from limited suppliers. Sodium difluorochloroacetate and sulfur are standard inventory items for most chemical distributors, mitigating the risk of production stoppages due to raw material shortages. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality or environmental factors, leading to more consistent batch-to-batch performance. This reliability is critical for maintaining just-in-time delivery schedules and meeting the stringent deadlines imposed by downstream drug formulation partners.
- Scalability and Environmental Compliance: The scalability of this one-pot method is evidenced by its operation in common polar aprotic solvents like DMF, which are well-understood in large-scale reactor operations. The absence of cryogenic requirements or high-pressure vessels simplifies the engineering controls needed for scale-up, allowing for seamless transfer from laboratory glassware to industrial steel reactors. Moreover, the reduced waste profile aligns with modern green chemistry principles, facilitating easier compliance with increasingly strict environmental regulations regarding solvent emissions and heavy metal discharge. This environmental stewardship enhances the corporate sustainability profile of manufacturers adopting this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis. These answers are derived directly from the experimental data and mechanistic understanding presented in the patent literature. They serve as a quick reference for technical teams evaluating the feasibility of integrating this route into their existing production pipelines. For more specific data regarding impurity profiles or custom substrate evaluation, please refer to the detailed analytical reports available upon request.
Q: What are the key advantages of the one-pot synthesis method described in CN110372718B?
A: The method eliminates the need for expensive transition metal catalysts and harsh difluoromethylating reagents. It utilizes cheap, commercially available sulfur and sodium difluorochloroacetate, significantly simplifying the supply chain and reducing raw material costs while maintaining high purity standards suitable for API intermediates.
Q: What is the substrate scope for this difluoromethylthiochromonothiophene synthesis?
A: The process demonstrates excellent tolerance for various substituents on the aromatic ring, including electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups like halogens and trifluoromethyl. It is also compatible with heteroaromatic systems such as furan, yielding products with up to 86% isolated yield.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the reaction operates under mild conditions (60-80°C) in a common solvent (DMF) without requiring specialized high-pressure equipment or cryogenic cooling. The simple workup procedure involving filtration and standard extraction makes it highly amenable to scale-up from kilogram to multi-ton production levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Difluoromethylthiochromonothiophene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this novel synthetic methodology for the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from discovery to market is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of sulfur chemistry and fluorination processes, adhering to stringent purity specifications and rigorous QC labs to guarantee product integrity. We understand that the successful commercialization of complex heterocycles requires more than just a recipe; it demands a partner who can navigate the complexities of process safety, regulatory compliance, and supply chain logistics with expertise and precision.
We invite you to collaborate with our technical team to explore how this cost-effective synthesis can enhance your drug development pipeline. By leveraging our capabilities, you can secure a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to contact our technical procurement team to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules. Let us help you accelerate your timeline to market with reliable, high-quality chemical solutions.
