Advanced Photochemical Synthesis of 3-Methylsulfonyl Thioflavonoids for Commercial Scale-Up
Introduction to Next-Generation Thioflavonoid Synthesis
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex heterocyclic scaffolds, particularly those found in bioactive natural products and drug candidates. Patent CN113880800A introduces a groundbreaking synthetic methodology for producing 3-methylsulfonyl substituted thioflavonoid compounds, a class of molecules with significant potential in medicinal chemistry. This innovation leverages a sophisticated sulfur dioxide insertion reaction driven by visible light photocatalysis, marking a distinct departure from traditional thermal cyclization methods. By utilizing a catalytic amount of sodium methanesulfinate as an initiator alongside a photosensitizer, the process achieves high efficiency and excellent product purity under remarkably mild conditions. For R&D directors and process chemists, this represents a pivotal shift towards greener, more sustainable manufacturing protocols that eliminate the need for harsh oxidants while maintaining robust yields across a diverse range of substrates.
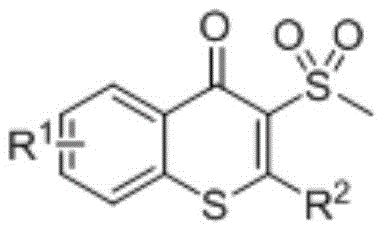
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of thioflavonoid cores has relied on methodologies that pose significant operational and economic challenges for large-scale manufacturing. Traditional approaches, such as those developed by the Larock group, often necessitate the use of highly reactive and difficult-to-handle reagents like iodine chloride at cryogenic temperatures around minus 78 degrees Celsius. Such extreme conditions not only demand specialized equipment capable of sustaining deep freezing but also incur substantial energy costs and safety risks associated with handling corrosive halogenating agents. Furthermore, alternative tandem addition substitution reactions involving sodium sulfide or the use of aryl sulfonyl hydrazides typically require stoichiometric amounts of transition metals or strong oxidizing agents. These additives frequently lead to complex impurity profiles, necessitating rigorous and costly purification steps to remove residual metals or oxidation by-products, thereby complicating the supply chain and increasing the overall cost of goods sold for the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the novel photochemical strategy disclosed in the patent data offers a streamlined, oxidant-free pathway that operates efficiently at ambient temperatures. By employing a sulfur dioxide substitute, such as potassium metabisulfite or sodium metabisulfite, in conjunction with a visible-light-driven photocatalyst, the reaction bypasses the need for hazardous oxidants entirely. This method utilizes a methylthio-substituted alkynone substrate which undergoes a radical-mediated cyclization to form the target thioflavonoid skeleton with high atom economy. The elimination of extreme temperature requirements and toxic reagents significantly simplifies the reactor setup and operational protocol, making it highly attractive for industrial scale-up. The versatility of this approach is further evidenced by its compatibility with a wide array of functional groups, allowing for the synthesis of diverse derivatives without compromising yield or purity, thus providing a superior alternative for the production of high-value chemical intermediates.
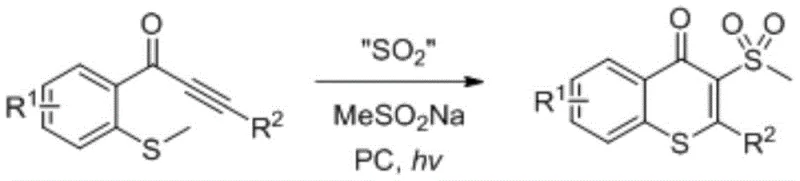
Mechanistic Insights into Photochemical Sulfur Dioxide Insertion
The core of this technological advancement lies in the intricate radical mechanism facilitated by the photosensitizer, typically fac-Ir(2-phenylpyridyl)3 or an organic acridinium-based catalyst. Upon irradiation with visible light, the photosensitizer enters an excited state, initiating a single-electron transfer process that activates the sulfur dioxide surrogate. This activation generates sulfonyl radicals in situ, which then attack the electron-deficient alkyne moiety of the methylthio-substituted alkynone substrate. The subsequent intramolecular cyclization involves the sulfur atom of the methylthio group attacking the vinyl radical intermediate, closing the ring to form the thioflavonoid core. This cascade occurs smoothly at room temperature, driven by the thermodynamic stability of the newly formed aromatic system and the strong sulfur-carbon bonds. The use of sodium methanesulfinate as a catalytic initiator is crucial, as it helps propagate the radical chain reaction, ensuring high conversion rates even with low catalyst loading, which is a critical factor for minimizing metal contamination in the final product.
From an impurity control perspective, the mildness of this photochemical protocol is paramount. Traditional oxidative conditions often lead to over-oxidation of sensitive functional groups or decomposition of the thioether linkage, generating difficult-to-remove side products. However, the neutral and non-oxidative nature of this SO2 insertion reaction preserves the integrity of sensitive substituents such as halogens, trifluoromethyl groups, and amines. The absence of transition metal catalysts in the stoichiometric sense (using only ppm levels of photocatalyst) drastically reduces the burden on downstream purification processes. This results in a cleaner crude reaction mixture, facilitating easier isolation of the target compound via standard chromatographic techniques. For quality assurance teams, this translates to a more consistent impurity profile and a higher likelihood of meeting stringent regulatory specifications for pharmaceutical intermediates without the need for extensive recrystallization or scavenging steps.
How to Synthesize 3-Methylsulfonyl Thioflavonoids Efficiently
The practical implementation of this synthesis route is designed for straightforward execution in standard laboratory and pilot plant settings. The process begins with the precise weighing of the methylthio-substituted alkynone precursor, the sulfur dioxide source, and the catalytic initiators, which are then dissolved in a common organic solvent such as dichloromethane or ethyl acetate. The reaction vessel is purged with an inert gas like nitrogen to prevent oxygen quenching of the excited photocatalyst states, ensuring maximum efficiency of the light-driven cycle. Once the mixture is exposed to a standard white daylight lamp, the transformation proceeds autonomously over a period of approximately 24 hours, requiring minimal operator intervention. Detailed standardized synthetic steps, including specific molar ratios and workup procedures validated by experimental examples, are outlined in the technical guide below to ensure reproducibility and safety during scale-up operations.
- Combine methylthio-substituted alkynone, sulfur dioxide substitute (e.g., K2S2O5), sodium methanesulfinate, and a photosensitizer (e.g., fac-Ir(ppy)3) in an organic solvent like dichloromethane under an inert atmosphere.
- Stir the reaction mixture at room temperature (15-40°C) under illumination from a white daylight lamp (e.g., 32W) for approximately 24 hours to facilitate the radical insertion cyclization.
- Upon completion, concentrate the reaction solution under reduced pressure and purify the crude product via column chromatography using a mixture of ethyl acetate and petroleum ether to isolate the target thioflavonoid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this photochemical methodology offers compelling economic and logistical benefits that directly impact the bottom line. The primary driver for cost optimization is the elimination of expensive and hazardous reagents such as iodine chloride or stoichiometric oxidants, which not only carry high purchase prices but also incur significant disposal costs due to their classification as hazardous waste. By replacing these with inexpensive, commodity-grade chemicals like potassium metabisulfite and sodium methanesulfinate, the raw material cost structure is significantly improved. Furthermore, the ability to run the reaction at room temperature eliminates the energy-intensive requirements for cryogenic cooling or high-temperature heating, leading to substantial reductions in utility consumption and operational overheads. This energy efficiency, combined with the simplified reactor configuration, allows for a more agile and cost-effective manufacturing process that can respond quickly to market demands.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts and strong oxidants from the process workflow fundamentally alters the cost dynamics of production. Without the need for expensive metal scavengers or complex waste treatment protocols for heavy metals, the downstream processing costs are drastically simplified. The use of catalytic amounts of photocatalyst means that the consumption of precious metals is minimized to trace levels, reducing the financial exposure to volatile metal markets. Additionally, the high reaction yields reported in the patent data, ranging up to 92 percent for certain substrates, ensure that raw material utilization is maximized, minimizing waste and improving the overall mass balance of the synthesis. This efficiency translates directly into a lower cost per kilogram of the final intermediate, enhancing competitiveness in the global marketplace.
- Enhanced Supply Chain Reliability: The reliance on readily available, bulk chemical feedstocks enhances the resilience of the supply chain against disruptions. Reagents such as metabisulfites and common organic solvents are produced on a massive industrial scale, ensuring consistent availability and stable pricing compared to specialized, niche reagents required by older methods. The robustness of the reaction conditions, which tolerate a wide variety of functional groups, means that suppliers can source diverse starting materials without fearing batch-to-batch variability caused by minor impurities. This flexibility allows for a more diversified sourcing strategy, reducing the risk of single-supplier dependency. Moreover, the simplified operational requirements mean that production can be easily transferred between different manufacturing sites without the need for highly specialized infrastructure, ensuring continuity of supply even in the face of regional logistical challenges.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been a challenge, but modern flow chemistry and LED technologies have made this increasingly viable for tonnage production. The benign nature of the reagents and the absence of toxic by-products align perfectly with modern environmental, health, and safety (EHS) standards. The process generates minimal hazardous waste, simplifying compliance with stringent environmental regulations and reducing the carbon footprint of the manufacturing operation. The ease of workup, involving simple concentration and chromatography, facilitates rapid scale-up from gram to kilogram quantities without the need for process re-engineering. This scalability ensures that the technology can meet the growing demand for these intermediates in the pharmaceutical and agrochemical sectors while maintaining a commitment to sustainable and responsible chemical manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the practical aspects of the technology. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into their existing production pipelines. The answers highlight the robustness of the chemistry and its alignment with industry standards for quality and efficiency.
Q: Does this synthesis method require hazardous oxidizing agents?
A: No, a key advantage of this patented method (CN113880800A) is that it operates without the need for external oxidants, which simplifies the reaction system and reduces safety hazards associated with strong oxidizers.
Q: What are the optimal reaction conditions for this transformation?
A: The reaction proceeds efficiently at room temperature (preferably 25-35°C) under visible light illumination using a photosensitizer catalyst, avoiding the extreme low temperatures (-78°C) required by traditional methods.
Q: Is the substrate scope limited for this photochemical process?
A: The method demonstrates a wide substrate application range, tolerating various substituents such as halogens, trifluoromethyl, dimethylamino, and methoxyl groups on the phenyl or thiophene rings, ensuring versatility for diverse derivative synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Methylsulfonyl Thioflavonoids Supplier
As a leader in the fine chemical sector, NINGBO INNO PHARMCHEM is uniquely positioned to leverage this advanced photochemical technology for the commercial production of high-purity 3-methylsulfonyl thioflavonoids. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We understand the critical importance of stringent purity specifications and rigorous QC labs in the pharmaceutical supply chain, and our facilities are equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the highest international standards. By partnering with us, clients gain access to a supply chain that is not only reliable and compliant but also optimized for cost and sustainability through the adoption of cutting-edge synthetic methodologies.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to evaluate the feasibility of this route for a new drug candidate, our experts are ready to provide detailed support. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to technological excellence can drive value and efficiency in your upcoming projects.
