Advanced Oxidative Synthesis of Phenyl 2-Pyrimidinyl Ketones for Commercial Herbicide Production
Advanced Oxidative Synthesis of Phenyl 2-Pyrimidinyl Ketones for Commercial Herbicide Production
The global demand for high-efficiency herbicides continues to drive innovation in the synthesis of critical agrochemical intermediates. Patent CN101048387A introduces a transformative methodology for the preparation of phenyl 2-pyrimidinyl ketones, specifically compounds of formula (I), which serve as pivotal precursors for sulfonylurea herbicides. This technology addresses long-standing challenges in oxidative cleavage reactions by replacing hazardous traditional oxidants with a benign hydrogen peroxide and acetic acid system. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, this process represents a significant leap forward in both chemical efficiency and operational safety. The ability to convert sulfide precursors directly into high-value ketone functionalities with excellent yields establishes a new benchmark for sustainable manufacturing in the fine chemicals sector.
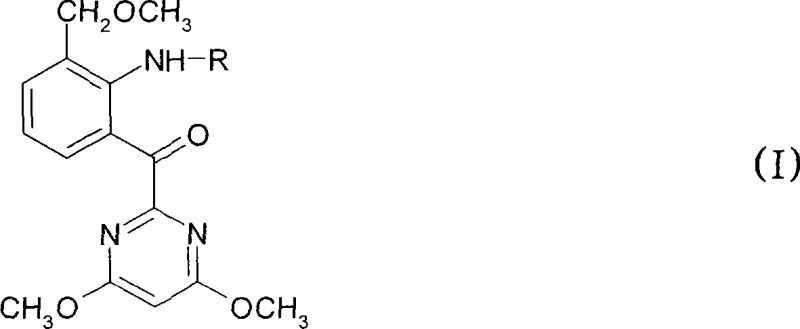
Traditionally, the synthesis of complex heterocyclic ketones often relied on stoichiometric amounts of heavy metal oxidants or multi-step sequences that generated substantial waste streams. Conventional methods frequently suffered from poor atom economy, requiring extensive purification protocols to remove metal residues that could poison downstream catalytic processes. Furthermore, the handling of strong oxidizing agents posed significant safety risks in large-scale reactors, necessitating specialized containment and waste treatment infrastructure. These limitations not only inflated the cost of goods sold but also created bottlenecks in supply chain continuity, as regulatory pressures on heavy metal discharge became increasingly stringent. Consequently, manufacturers faced difficulties in scaling these processes to meet the volumetric demands of the global agrochemical market without compromising on environmental compliance or profit margins.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The historical reliance on chromium-based or manganese-based oxidants for converting thio-ethers to ketones has become untenable in modern green chemistry frameworks. These reagents generate toxic sludge that requires costly disposal procedures, directly impacting the bottom line for any cost reduction in herbicide manufacturing initiative. Additionally, the reaction conditions for these traditional oxidations were often harsh, leading to the degradation of sensitive functional groups on the pyrimidine ring, such as methoxy substituents. This lack of chemoselectivity resulted in complex impurity profiles that were difficult to separate, lowering the overall yield and purity of the final active pharmaceutical or agrochemical ingredient. The operational complexity of managing exothermic reactions with unstable oxidants further restricted the batch sizes, limiting the ability to achieve economies of scale.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a remarkably simple yet effective combination of hydrogen peroxide and acetic acid to effect the oxidative transformation. This method leverages the in situ generation of peracetic acid or activated peroxide species to selectively oxidize the methylthio group, eventually cleaving it to form the desired carbonyl functionality. The use of acetic acid serves a dual purpose as both a solvent and a reaction promoter, simplifying the reaction matrix and reducing the need for additional volatile organic compounds. As illustrated in the reaction scheme below, the conversion of the sulfide precursor (Formula II) to the ketone product (Formula I) proceeds with high fidelity, preserving the integrity of the surrounding molecular architecture.
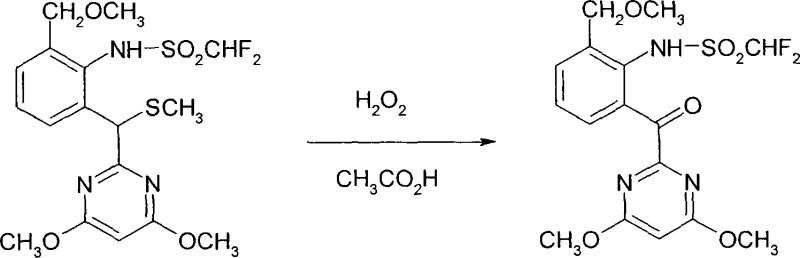
This streamlined process eliminates the need for transition metal catalysts, thereby removing the risk of metal contamination in the final product. The reaction conditions are mild enough to be conducted in standard glass-lined steel reactors, facilitating immediate adoption by existing manufacturing facilities. By shifting to this peroxide-based system, producers can achieve a drastic simplification of the workup procedure, often requiring only dilution with water and extraction to isolate the crude product. This operational simplicity translates directly into reduced cycle times and lower utility consumption, providing a compelling value proposition for supply chain heads focused on efficiency and throughput optimization.
Mechanistic Insights into Peroxide-Mediated Oxidative Cleavage
The mechanistic pathway of this transformation involves the sequential oxidation of the sulfur atom. Initially, the sulfide moiety is oxidized to the corresponding sulfoxide, an intermediate that can be isolated under controlled conditions as shown in Formula (IIc). This step is crucial as it activates the adjacent carbon-sulfur bond towards further oxidative attack. In the presence of excess hydrogen peroxide and the acidic medium provided by acetic acid, the sulfoxide undergoes further oxidation, likely forming a sulfone or a Pummerer-type intermediate that facilitates hydrolysis. The acidic environment protonates the oxygen species, enhancing their electrophilicity and driving the cleavage of the C-S bond to release the carbonyl group. Understanding this progression allows chemists to fine-tune the stoichiometry of hydrogen peroxide to stop at the sulfoxide stage if desired, or push through to the ketone for complete conversion.
Controlling the impurity profile is paramount for high-purity agrochemical intermediate production. The specificity of the hydrogen peroxide/acetic acid system minimizes side reactions such as ring oxidation or demethylation of the methoxy groups, which are common pitfalls with stronger, non-selective oxidants. The patent data indicates that by adjusting the temperature and reaction time, one can maximize the yield of the target ketone while minimizing the formation of over-oxidized byproducts. For instance, maintaining the reaction temperature between 40°C and 100°C ensures sufficient energy for the cleavage step without triggering thermal decomposition of the sensitive pyrimidine core. This level of control is essential for meeting the rigorous quality specifications required by downstream formulators of herbicidal active ingredients.
How to Synthesize Phenyl 2-Pyrimidinyl Ketones Efficiently
The synthesis protocol outlined in the patent provides a robust framework for laboratory and pilot-scale production. The process begins with the dissolution of the sulfide starting material in glacial acetic acid, followed by the controlled addition of aqueous hydrogen peroxide. The reaction is typically initiated at room temperature to manage the initial exotherm and then heated to accelerate the conversion. Workup involves quenching the excess oxidant, often with a reducing agent like sodium sulfite, followed by extraction into an organic solvent such as ethyl acetate. The detailed standardized synthesis steps see the guide below.
- Dissolve the starting sulfide compound (Formula II) in glacial acetic acid or an aqueous acetic acid mixture.
- Add 30-35% aqueous hydrogen peroxide solution slowly while maintaining temperature control between 15°C and 100°C.
- Stir the reaction mixture for 1 to 48 hours, then quench with water and extract the product using organic solvents like ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this oxidative process offers tangible strategic benefits beyond mere chemical yield. The shift away from specialized, hazardous reagents to commodity chemicals like hydrogen peroxide and acetic acid significantly de-risks the supply chain. These raw materials are globally available in bulk quantities, ensuring consistent pricing and availability even during market fluctuations. Furthermore, the elimination of heavy metals simplifies the regulatory compliance landscape, reducing the administrative burden associated with environmental reporting and waste disposal permits. This operational agility allows manufacturers to respond more rapidly to market demand spikes for herbicide intermediates without being constrained by waste treatment capacity.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of expensive, single-use oxidants with low-cost, high-volume commodities. By removing the need for expensive metal scavengers and complex filtration systems required for catalyst removal, the overall processing cost is substantially lowered. The simplified workup procedure reduces solvent consumption and energy usage associated with distillation and drying steps. Additionally, the high selectivity of the reaction minimizes product loss during purification, effectively increasing the mass balance and overall process efficiency. These factors combine to deliver significant cost savings that can be passed down the supply chain or retained as improved margin.
- Enhanced Supply Chain Reliability: Reliance on a diverse portfolio of readily available reagents enhances the resilience of the manufacturing supply chain. Unlike specialized catalysts that may have single-source suppliers and long lead times, hydrogen peroxide and acetic acid are produced by multiple vendors worldwide. This redundancy mitigates the risk of supply disruptions due to geopolitical issues or production outages at a single facility. Moreover, the stability of the reagents allows for safer storage and handling, reducing the likelihood of accidents that could halt production. This reliability is critical for maintaining just-in-time delivery schedules to major agrochemical clients.
- Scalability and Environmental Compliance: The inherent safety of the peroxide-based system facilitates seamless scale-up from kilogram to multi-ton production. The absence of toxic heavy metals means that wastewater treatment is less complex and costly, aligning with increasingly strict global environmental standards. The process generates primarily water and acetic acid as byproducts, which are easier to treat or recycle compared to heavy metal sludge. This environmental compatibility not only reduces disposal costs but also enhances the corporate sustainability profile of the manufacturer, a key factor for modern procurement decisions. The ability to scale safely and cleanly ensures long-term viability and regulatory approval for commercial operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the practical realities of scaling chemical processes while maintaining high quality and safety standards.
Q: What are the key advantages of using hydrogen peroxide in this synthesis?
A: Using hydrogen peroxide eliminates the need for toxic heavy metal oxidants, significantly reducing environmental waste and improving operator safety while maintaining high conversion rates.
Q: Can this process be scaled for industrial production?
A: Yes, the process utilizes inexpensive, commercially available reagents like acetic acid and hydrogen peroxide, making it highly suitable for large-scale manufacturing without complex equipment requirements.
Q: What is the purity profile of the resulting ketones?
A: The method allows for easy purification through standard extraction and crystallization techniques, yielding high-purity intermediates suitable for downstream herbicide synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenyl 2-Pyrimidinyl Ketones Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final agrochemical products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of phenyl 2-pyrimidinyl ketones meets the highest industry standards. Our commitment to technical excellence allows us to navigate complex synthetic challenges, delivering materials that facilitate efficient downstream processing for our partners.
We invite you to engage with our technical procurement team to discuss how this advanced oxidative technology can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your project timelines. Let us collaborate to drive innovation and efficiency in the global agrochemical market together.
