Scalable Enantioselective Hydrogenation of Oximes for High-Purity Agrochemical Intermediates
Introduction to Advanced Enantioselective Hydrogenation Technology
The landscape of chiral intermediate synthesis is undergoing a significant transformation driven by the need for more sustainable and efficient catalytic processes. Patent CN113195460A introduces a groundbreaking methodology for the enantioselective iridium-catalyzed hydrogenation of oximes and oxime ethers. This technology addresses long-standing challenges in producing chiral hydroxylamines, which are critical building blocks for various bioactive molecules. By leveraging novel chiral iridium complexes in combination with acidic additives, this process achieves high levels of stereocontrol that were previously difficult to attain with traditional reduction methods. For industry leaders seeking a reliable agrochemical intermediate supplier, understanding this technological shift is paramount for securing future supply chains.
The core innovation lies in the specific design of the iridium catalyst, which features a chiral cyclopentadienyl ligand and a bidentate C,N-chelating ligand. This unique structural arrangement creates a highly defined chiral environment around the metal center, allowing for precise differentiation between prochiral faces of the oxime substrate. Unlike earlier methods that often suffered from poor catalytic efficiency or over-reduction to primary amines, this new approach ensures that the reaction stops selectively at the hydroxylamine stage. This level of precision is essential for manufacturing high-purity agrochemical intermediates where impurity profiles are strictly regulated.
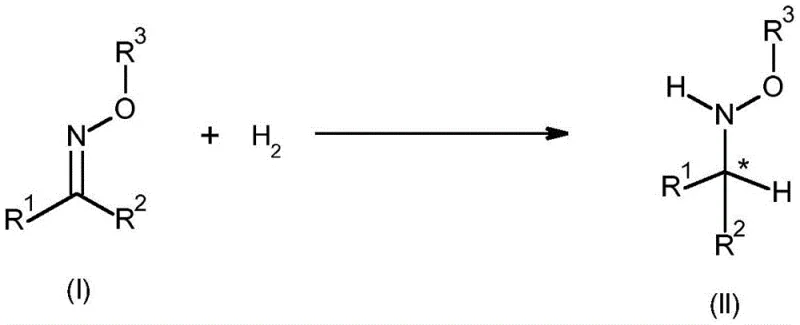
Furthermore, the versatility of this catalytic system allows it to accommodate a wide range of substituents on the oxime substrate, including alkyl, aryl, and heteroaryl groups. This broad substrate scope makes it an invaluable tool for process chemists working on diverse molecular scaffolds. The ability to tune the catalyst structure by modifying the ligands provides an additional layer of optimization, enabling the fine-tuning of enantioselectivity for specific challenging substrates. As we delve deeper into the technical specifics, it becomes clear why this patent represents a significant leap forward in asymmetric synthesis capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral hydroxylamines has relied heavily on stoichiometric reduction methods, particularly using borane complexes in the presence of chiral amino alcohols. While effective in some laboratory settings, these conventional methods present severe drawbacks when considered for industrial application. The primary issue is the poor atom economy associated with using stoichiometric amounts of borane reagents, which generates substantial quantities of boron-containing waste that requires costly disposal and treatment. Additionally, borane reductions are notorious for lacking chemoselectivity, often leading to the over-reduction of the oxime functionality all the way to the corresponding primary amine, thereby destroying the desired hydroxylamine motif.
Another significant limitation of traditional transition metal-catalyzed hydrogenation attempts has been the difficulty in controlling stereoselectivity. Many early catalysts based on cobalt, ruthenium, or rhodium failed to provide sufficient enantiomeric excess or suffered from rapid deactivation under the reaction conditions. The tendency for these systems to reduce carbon-carbon double bonds instead of the nitrogen-oxygen bond further complicates the purification process. For procurement teams focused on cost reduction in agrochemical intermediate manufacturing, these inefficiencies translate directly into higher raw material costs and lower overall yields, making the final product economically unviable at scale.
The Novel Approach
The novel approach disclosed in the patent utilizes a sophisticated iridium catalytic system that overcomes these historical barriers. By employing molecular hydrogen as the reducing agent, the process achieves near-perfect atom economy, with water being the only theoretical byproduct alongside the desired hydroxylamine. The introduction of a strong acid additive, such as methanesulfonic acid, plays a pivotal role in activating the substrate and stabilizing the catalytic cycle, preventing the formation of inactive catalyst species. This synergy between the chiral iridium complex and the acid allows the reaction to proceed with high turnover numbers and exceptional stereoselectivity.
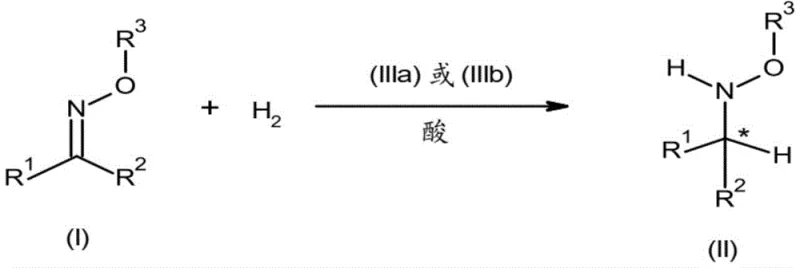
Moreover, this method effectively suppresses the over-reduction pathway that plagues borane chemistry. The specific electronic and steric properties of the iridium catalyst ensure that the reduction stops cleanly at the hydroxylamine stage, preserving the N-O bond integrity. This chemoselectivity simplifies downstream processing, as there is no need for complex separation steps to remove primary amine impurities. For supply chain managers, this reliability means more predictable batch outcomes and reduced risk of production delays caused by off-spec material, facilitating the commercial scale-up of complex agrochemical intermediates.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Hydrogenation
The success of this enantioselective process is rooted in the intricate mechanistic interplay between the iridium center, the chiral ligands, and the substrate. The catalyst typically features a pentamethylcyclopentadienyl (Cp*) or a substituted chiral cyclopentadienyl ligand that acts as an electron donor and provides a steric cap. Coupled with this is a bidentate C,N-ligand, often derived from imines or similar structures, which coordinates to the iridium to form a rigid, chiral pocket. This specific geometry forces the incoming oxime substrate to approach the metal hydride species from a single preferred direction, thereby dictating the stereochemical outcome of the hydrogen addition.
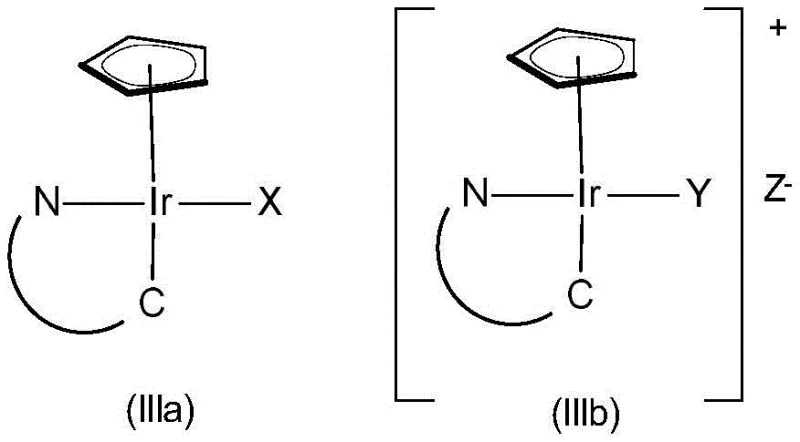
During the catalytic cycle, the acid additive serves to protonate the nitrogen atom of the oxime, increasing its electrophilicity and facilitating coordination to the electron-rich iridium center. The subsequent migratory insertion of the N=O bond into the Ir-H bond is the stereodetermining step. The bulky substituents on the chiral ligands create a steric clash that disfavors one transition state over the other, leading to high enantiomeric ratios. Following the insertion, reductive elimination releases the chiral hydroxylamine product and regenerates the active iridium species. Understanding these mechanistic nuances is critical for R&D directors aiming to optimize reaction parameters such as temperature, pressure, and solvent choice for maximum efficiency.
Impurity control is another area where this mechanism offers distinct advantages. Because the catalyst is highly specific for the N=O bond reduction, side reactions such as the hydrogenation of aromatic rings or other sensitive functional groups are minimized. This inherent selectivity reduces the burden on purification units and ensures that the final high-purity agrochemical intermediates meet stringent regulatory standards. The robustness of the iridium complex also means it can withstand the acidic conditions necessary for the reaction without decomposing, ensuring consistent performance throughout the batch.
How to Synthesize Chiral Hydroxylamines Efficiently
The synthesis of these valuable chiral building blocks follows a streamlined protocol that integrates catalyst preparation, substrate formation, and the key hydrogenation step. The process begins with the generation of the active iridium catalyst, which can be prepared in situ or isolated as a stable salt. The oxime substrate is typically formed via condensation of a ketone with an alkoxyamine, a straightforward transformation that yields the precursor ready for reduction. The core hydrogenation step is then conducted in a pressure vessel using common alcoholic solvents.
- Preparation of the chiral iridium catalyst complex involving cyclometallation of an iridium dimer with a C,N-bidentate ligand followed by halogen abstraction.
- Formation of the oxime ether substrate through condensation of the corresponding ketone with an alkoxyamine hydrochloride salt.
- Execution of the asymmetric hydrogenation reaction under elevated hydrogen pressure (e.g., 50 bar) in the presence of the iridium catalyst and a stoichiometric acid additive.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iridium-catalyzed technology offers compelling strategic benefits beyond mere technical superiority. The shift from stoichiometric reagents to a catalytic hydrogenation process fundamentally alters the cost structure of production. By eliminating the need for expensive and hazardous borane reagents, manufacturers can achieve substantial cost savings in raw material procurement. Furthermore, the use of molecular hydrogen, a commodity chemical, replaces high-cost reducing agents, driving down the variable cost per kilogram of the final product significantly.
- Cost Reduction in Manufacturing: The transition to a catalytic process drastically reduces the mass intensity of the reaction. Since the iridium catalyst is used in minute quantities (often less than 1 mol%), the cost contribution of the precious metal is negligible compared to the savings gained from avoiding stoichiometric waste generation. This efficiency translates directly into a more competitive pricing model for the final intermediate, allowing buyers to negotiate better terms without compromising on quality or supply security.
- Enhanced Supply Chain Reliability: Relying on specialized stoichiometric reagents can introduce supply chain vulnerabilities, especially if those reagents are sourced from limited suppliers. In contrast, the reagents required for this iridium-catalyzed process—hydrogen gas, common acids, and standard solvents—are widely available globally. This ubiquity ensures that production is not held hostage by the availability of niche chemicals, thereby reducing lead time for high-purity agrochemical intermediates and mitigating the risk of disruption.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process is far superior. The reduction in chemical waste simplifies effluent treatment and lowers the environmental footprint of the manufacturing site. This aligns with increasingly strict global regulations on chemical manufacturing and waste disposal. The scalability of high-pressure hydrogenation is well-established in the fine chemical industry, meaning that transferring this process from pilot scale to multi-ton commercial production involves minimal technical risk, ensuring a steady and continuous supply.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enantioselective hydrogenation technology. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and advantages of the method for potential partners and stakeholders.
Q: What are the primary advantages of using iridium catalysis over borane reduction for oximes?
A: Iridium-catalyzed hydrogenation utilizes molecular hydrogen, offering superior atom economy compared to stoichiometric borane reagents. It significantly reduces chemical waste and avoids the formation of stoichiometric boron byproducts, while providing excellent control over stereoselectivity to prevent over-reduction to primary amines.
Q: How does the acid additive influence the enantioselective hydrogenation process?
A: The acid additive, such as methanesulfonic acid, is crucial for partially protonating the oxime substrate and the hydroxylamine product. This protonation facilitates the coordination to the iridium center and stabilizes the intermediate species, ensuring high conversion rates and maintaining the integrity of the chiral center during the reduction.
Q: Is this catalytic process suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. It operates under standard high-pressure hydrogenation conditions using robust iridium complexes. The use of catalytic amounts of metal (typically 0.01 to 1 mol%) and common organic solvents like isopropanol makes it highly adaptable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Hydroxylamine Supplier
The technological advancements detailed in patent CN113195460A represent a significant opportunity for the fine chemical industry to produce chiral hydroxylamines more efficiently and sustainably. At NINGBO INNO PHARMCHEM, we recognize the potential of this iridium-catalyzed route to transform the production of key agrochemical and pharmaceutical intermediates. Our team of expert process chemists is well-versed in handling sensitive organometallic catalysts and high-pressure hydrogenation reactions, ensuring that the transition from laboratory discovery to commercial reality is seamless and robust.
We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, adhering to stringent purity specifications and operating within our rigorous QC labs. Whether you require custom synthesis of the specific iridium catalysts or the full-scale manufacture of the hydroxylamine intermediates, our infrastructure is designed to support your needs. We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis for your specific project. Let us provide you with specific COA data and route feasibility assessments to demonstrate how we can become your trusted partner in delivering high-quality chiral intermediates.
