Advanced Acid-Catalyzed Synthesis of Polysubstituted Triazole Derivatives for Commercial Scale-Up
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for more efficient and cleaner synthetic routes for pharmaceutical intermediates. A significant breakthrough in this domain is documented in Chinese Patent CN109096211B, which details a novel preparation method for polysubstituted triazole derivatives. This technology represents a paradigm shift from traditional metal-catalyzed approaches to a highly efficient, strong acid-catalyzed 1,3-dipolar cycloaddition. By utilizing acrolein derivatives and azide compounds under mild, low-temperature conditions, this method addresses critical pain points in modern organic synthesis, such as regioselectivity control and the elimination of toxic metal residues. For R&D directors and procurement specialists alike, understanding this patented methodology is crucial, as it offers a pathway to high-purity intermediates with streamlined processing, positioning it as a vital tool for the reliable pharmaceutical intermediate supplier seeking to optimize their portfolio.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of 1,2,3-triazole derivatives has heavily relied on the Huisgen 1,3-dipolar cycloaddition between terminal alkynes and azides. While the copper-catalyzed variant (CuAAC) revolutionized the field, it is not without significant drawbacks for large-scale industrial application. The primary concern for any quality-focused organization is the presence of residual copper, a heavy metal that is strictly regulated in active pharmaceutical ingredients (APIs). Removing trace amounts of copper to meet stringent pharmacopeial standards often necessitates additional purification steps, such as specialized scavenging resins or repeated recrystallization, which drastically increases both processing time and operational costs. Furthermore, conventional thermal cycloadditions without catalysts often require harsh conditions, including high temperatures and prolonged reaction times, which can lead to poor selectivity, decomposition of sensitive functional groups, and ultimately, lower overall yields. These inefficiencies create bottlenecks in the supply chain, making the cost reduction in pharmaceutical intermediate manufacturing a challenging objective when relying solely on legacy technologies.
The Novel Approach
In stark contrast, the method disclosed in patent CN109096211B introduces a robust alternative that bypasses these limitations entirely. By employing a strong acid catalyst, such as trifluoromethanesulfonic acid or boron trifluoride etherate, the reaction proceeds efficiently at temperatures below 0°C, often completing within a mere 30 to 120 minutes. This low-temperature protocol not only preserves the integrity of thermally labile substituents but also enhances the regioselectivity of the cycloaddition, ensuring the formation of the desired 1,4-disubstituted or polysubstituted triazole isomers with high fidelity. The absence of transition metals simplifies the downstream processing significantly; there is no need for expensive metal scavengers, allowing for a straightforward workup involving dilution, extraction, and standard chromatographic purification. This streamlined workflow translates directly into enhanced supply chain reliability and substantial cost savings, making it an attractive option for the commercial scale-up of complex polymer additives or drug candidates.
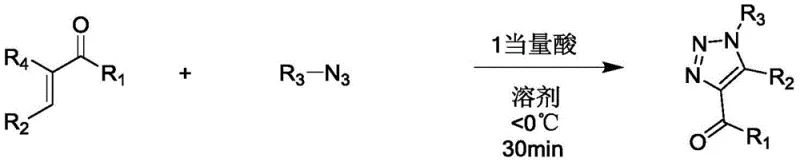
Mechanistic Insights into Acid-Catalyzed 1,3-Dipolar Cycloaddition
To fully appreciate the technical superiority of this method, one must delve into the mechanistic nuances of the acid-catalyzed transformation. The core of this reaction lies in the activation of the electron-deficient double bond of the acrolein derivative by the strong Brønsted or Lewis acid. In the presence of a catalyst like trifluoromethanesulfonic acid, the carbonyl oxygen or the alkene moiety of the acrolein derivative becomes protonated or coordinated, significantly increasing its electrophilicity. This activation lowers the energy barrier for the nucleophilic attack by the azide species, facilitating the 1,3-dipolar cycloaddition under much milder conditions than would otherwise be possible. The reaction mechanism likely proceeds through a concerted or stepwise pathway where the azide adds across the activated alkene, followed by cyclization and subsequent aromatization to form the stable triazole ring. This precise control over the electronic environment of the reactants allows for the accommodation of a wide variety of substituents, including electron-withdrawing groups like nitro or halogen atoms, which might deactivate the system in uncatalyzed scenarios.
Furthermore, the choice of reaction conditions plays a pivotal role in impurity control and product quality. By maintaining the reaction temperature below 0°C, typically around -40°C as exemplified in the patent data, the formation of side products such as polymerization of the acrolein derivative or decomposition of the azide is effectively suppressed. The rapid reaction kinetics, evidenced by completion times as short as 30 minutes, minimize the exposure of the product to the acidic environment, preventing potential acid-catalyzed degradation of the newly formed triazole ring. The quenching step, utilizing triethylamine in a stoichiometric ratio of 2:1 to 3:1 relative to the acid, ensures immediate neutralization, locking in the high purity of the crude product. This rigorous control over the reaction parameters results in yields reaching up to 98%, demonstrating a level of efficiency that is critical for reducing lead time for high-purity pharmaceutical intermediates in a competitive market.
How to Synthesize Polysubstituted Triazole Efficiently
The practical implementation of this synthesis route is designed for ease of operation while maintaining high standards of safety and efficiency. The process begins with the careful preparation of the reaction mixture, where precise stoichiometric ratios of the acrolein derivative, azide, and strong acid catalyst are combined in a suitable solvent such as dichloromethane or acetonitrile. The maintenance of low temperature is critical during this addition phase to manage the exotherm and ensure selectivity. Following the reaction period, the mixture is quenched and subjected to a standard liquid-liquid extraction protocol. The detailed standardized synthesis steps see the guide below for specific operational parameters.
- Combine acrolein derivative, azide compound, strong acid catalyst, and solvent in a reaction vessel maintained below 0°C.
- React the mixture for 30 to 120 minutes, then quench the reaction by adding 2 to 3 equivalents of triethylamine relative to the acid.
- Dilute with dichloromethane, extract the organic phase, wash with water, dry, concentrate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this acid-catalyzed technology offers tangible strategic benefits that extend beyond mere chemical curiosity. The elimination of transition metal catalysts is perhaps the most significant advantage, as it removes a major cost center associated with metal procurement and, more importantly, the extensive purification required to meet regulatory limits. This simplification of the manufacturing process leads to a drastic reduction in cycle times, allowing for faster turnover of batches and improved responsiveness to market demands. Additionally, the use of commercially available and relatively inexpensive strong acids as catalysts, compared to specialized ligand-metal complexes, contributes to a lower bill of materials. The robustness of the reaction across a wide range of substrates means that a single platform technology can be used to produce a diverse library of intermediates, optimizing equipment utilization and reducing the need for specialized training or unique reactor setups for different products.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the removal of expensive noble metal catalysts and the associated downstream purification costs. Without the need for copper scavengers or complex filtration systems to remove metal residues, the operational expenditure per kilogram of product is significantly lowered. Furthermore, the high yields reported, often exceeding 90% and reaching up to 98% in optimized examples, mean that raw material waste is minimized, maximizing the atom economy of the process. The short reaction times also translate to lower energy consumption for heating or prolonged stirring, contributing to a leaner and more cost-effective manufacturing profile that aligns with modern green chemistry principles.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the reliance on readily available starting materials such as acrolein derivatives and organic azides ensures a stable and secure source of inputs. Unlike specialized catalysts that may have long lead times or single-source dependencies, the reagents for this process are commodity chemicals with robust global supply networks. The simplicity of the workup procedure, involving standard solvents like dichloromethane and common bases like triethylamine, further reduces the risk of supply disruptions. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of downstream pharmaceutical clients, thereby strengthening the overall resilience of the supply chain.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by its straightforward engineering requirements. The reaction does not demand extreme pressures or temperatures, allowing it to be run in standard glass-lined or stainless steel reactors commonly found in fine chemical plants. The waste stream is also easier to manage; the absence of heavy metals simplifies wastewater treatment and disposal, reducing the environmental footprint and compliance costs associated with hazardous waste handling. The ability to achieve high purity through simple column chromatography or crystallization means that the final product meets stringent quality specifications with minimal environmental impact, supporting sustainable manufacturing goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this patented synthesis method. These answers are derived directly from the experimental data and technical disclosures found in the patent literature, providing a clear understanding of the process capabilities and limitations for potential partners and stakeholders.
Q: What are the advantages of this acid-catalyzed method over traditional copper-catalyzed click chemistry?
A: This method eliminates the need for transition metal catalysts like copper, thereby avoiding costly and complex heavy metal removal steps required for pharmaceutical grade purity. Additionally, it operates at significantly lower temperatures (below 0°C) compared to traditional thermal methods, enhancing selectivity and reducing energy consumption.
Q: What is the substrate scope for this triazole synthesis?
A: The process demonstrates a broad substrate scope, accommodating various acrolein derivatives with hydrogen, alkyl, aryl, amino, or alkoxy groups, as well as diverse azide compounds including benzyl, phenethyl, and alkyl azides. This versatility allows for the creation of complex libraries of triazole derivatives.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process is highly scalable due to its short reaction times (as low as 30 minutes), simple workup procedures involving standard extraction and chromatography, and the use of commercially available strong acid catalysts. The high yields reported (up to 98%) further support its economic viability for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the acid-catalyzed triazole formation described in CN109096211B. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative laboratory protocols into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity observed in the lab are faithfully reproduced in our pilot and production plants. We operate with stringent purity specifications and utilize rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of polysubstituted triazole meets the exacting standards required by the global pharmaceutical industry.
We invite forward-thinking R&D and procurement leaders to collaborate with us to leverage this technology for their specific projects. Whether you require custom synthesis of novel triazole derivatives or optimization of existing routes for cost and efficiency, our technical team is ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your project needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can accelerate your development timeline and enhance your product's market competitiveness.
